Introduction
The Arrangement of the Aorta
Aortic Dissection Pathogenesis
Symptoms
Pre-dispositions
Detection
Treatment
References
Further Reading
The aorta is the main artery of the human body. Its large structure is key to maintaining the vital function of pumping oxygenated blood around the whole body.
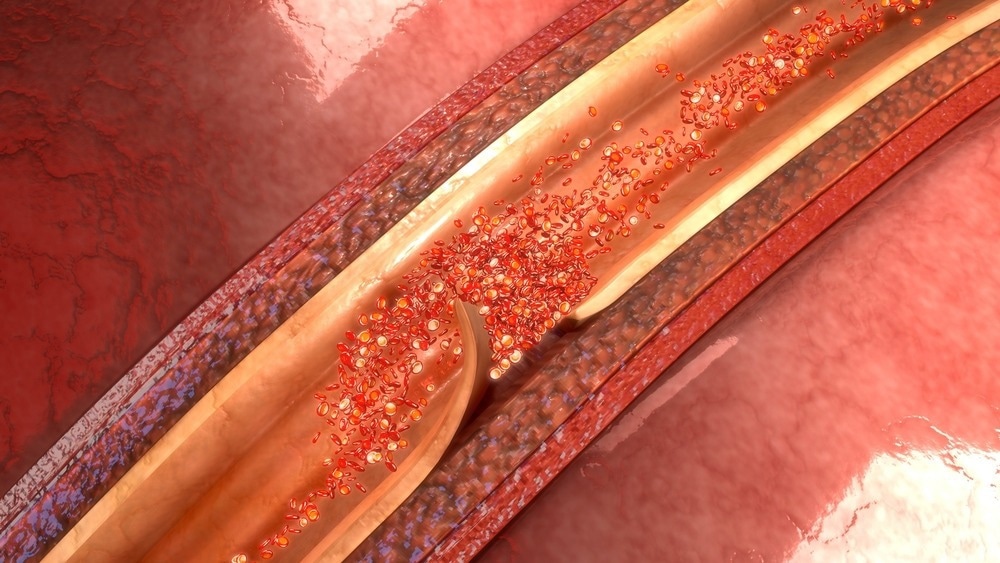
Aortic dissection. Image Credit: sciencepics/Shutterstock.com
Aortic dissection occurs when the wall of the aorta tears, allowing blood to exit the aorta through a false lumen. If the blood escapes through this rupture to the rest of the body, it can prove quickly fatal. The estimated yearly prevalence is 5 to 30 individuals per million.
The arrangement of the aorta
The aortic wall is constructed of the intima, the media and the adventitia. The intima is made of the external endothelium backed by connective tissue. This sublayer mediates between the two layers across the expansion and contraction of the aorta. The media consists of 50 layers of perforated elastin fibers disseminated with smooth muscle and collagenous fibers.
The adventitia is the external collagenous and connective tissue layer that provides strength and support to the vigorous motion of the aorta. The outer layer is intervened with an extensive network of blood vessels that extend into the media, collectively called the vasa vasorum.
Aortic dissection pathogenesis
There are two main systems used to classify the occurrences of aortic dissections. The Stanford system is the most common and differentiates by the locus of the intimal tear, type A in the ascending aorta and type B in the descending aorta.
The DeBakey system classifies into more specific loci: type 1 initiates in the ascending aorta at least until the aortic arch, type 2 is restricted to the ascending aorta and type 3 begins in the descending aorta until above the diaphragm (3a) or below the diaphragm (3b).
Aortic dissections begin with an intimal tear. Blood quickly invades the intimal-media space and causes separation between the layers while widening the entrance. The diversion of blood results in a sudden blood pressure drop; while the growth of the false lumen occludes the true lumen. The accumulation of blood predisposes to further cardiac trauma or even sudden death.
The collection of blood in vessels may lead to an aneurysm. Excess blood flow and blockages may cause ruptures of the ventricles, atria or vessels; ruptures are the most common cause of death. Accumulation of fluid in the pericardial space is known as hemopericardium. This compresses the heart and impairs the ability of the four chambers to properly gather blood – this is called acute cardiac tamponade. The stress on the heart may also result in aortic regurgitation, where blood leaks from the aorta back into the left ventricle.
Symptoms
Patients experiencing aortic dissection usually complain of a sharp ripping pain within the chest and upper back, this pain may extend to the neck and rest of the back. This pain is accompanied by shortness of breath, severe stomach pain and leg pain – this may develop into difficulty in walking. Stroke symptoms have also been identified with this condition; these include sudden vision problems, difficulties with speech, weakness or paralysis on one side and a weaker pulse detection on one side. Aortic dissections progress rapidly and can lead to a loss of consciousness depending on the severity.
Pre-dispositions
The incidence of aortic dissections in males is twice that of females with the former experiencing the disease in their 60s and 70s while the occurrence in the latter is associated with their 80s. Age is a risk factor for aortic dissection. The aorta experiences a loss of elastin and an increase in collagen which impacts flexibility and exertion tolerance; smooth muscle cell impairment and microcalcification within the media. Most ascending aortic dissections have been found to occur when the aortic diameter is less than 5.5cm.
Hypertension and smoking are applicable cardiac disease risk factors that lack a causal link to aortic dissection. Hypertension alters the hemodynamic forces applied to the aortic wall. This may induce cycles of injury and repair, increasing the risk of an intimal tear at these sites. Hypertension is also associated with loss of elastin and smooth muscle cell degradation.
Pre-existing cardiac distress such as aortic ulcers or pre-existing aneurysms weakens defenses against aortic dissections. Atherosclerosis and vasculitis compromise the integrity of blood vessels; inflammatory diseases, congenital cardiovascular defects and drug use are also risk factors. Pregnancy, delivery and iatrogenic aortic injury are other stressors on the heart that create vulnerability to aortic dissections.
There are several genetic diseases that pre-dispose vulnerability to the development of aortic dissection. Marfan’s syndrome results in alterations to the meshwork of aortic elastin fibers and vessels are more prone to aneurysm. An independent study found that 50% of patients with aortic dissection under the age of 40 had Marfan’s syndrome. Defects in the aortic wall are found in Ehlers-Danlos, Loeys–Dietz and Turner syndromes.
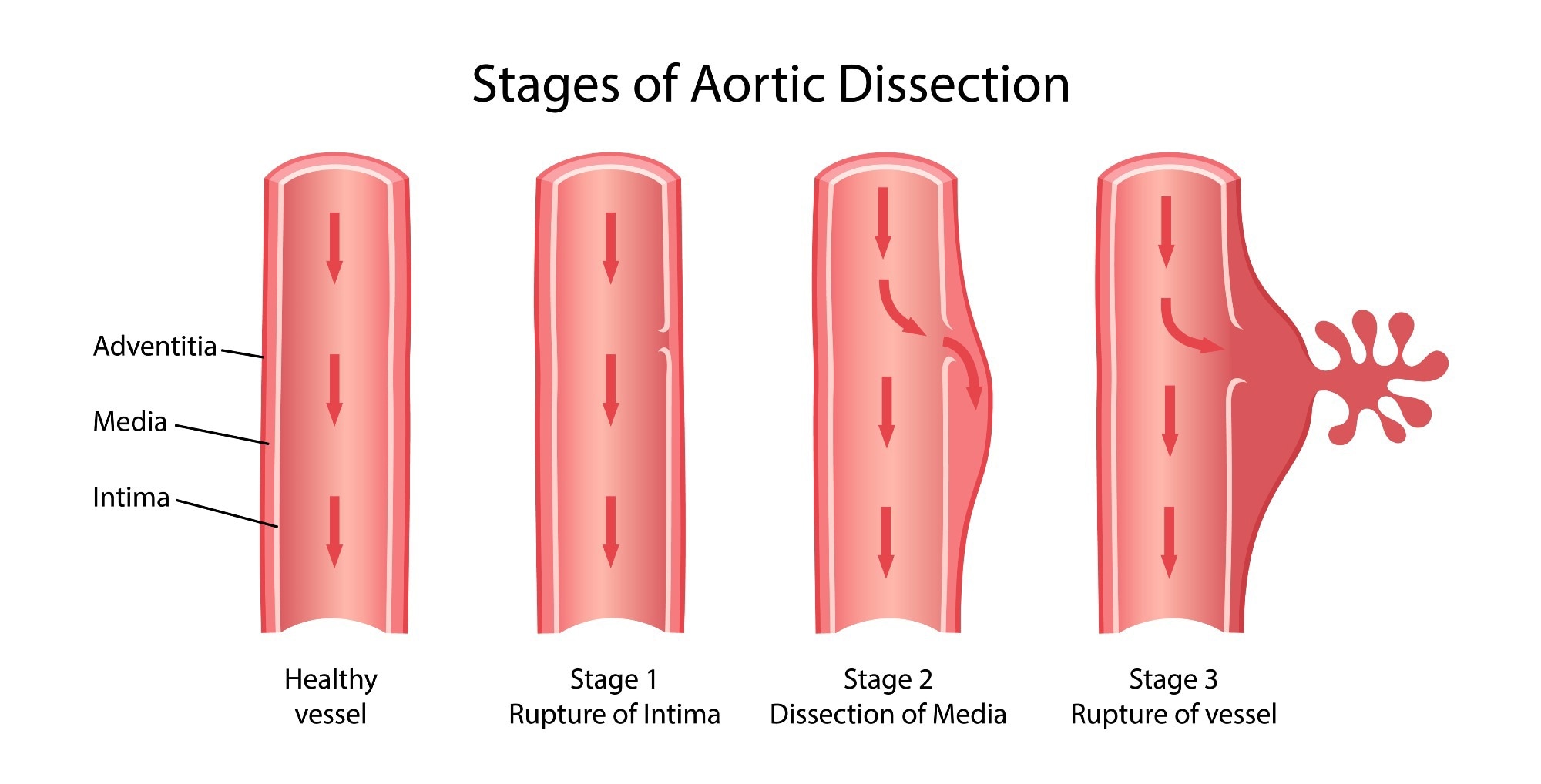
The stages of aortic dissection. Image Credit: Olga Bolbot/Shutterstock.com
Detection
Detection is usually conducted through ECG and X-ray by looking for the widening of the aortic silhouette – this marker is not always present. There is an overlap between the images of aortic dissection and acute myocardial infarction (AMI). 8% of AMI diagnoses are Type A aortic dissection.
Cardiovascular imaging is used to identify the presence of the aortic rupture. The classification systems are then utilized to determine the type of dissection. CT aortography, transesophageal echocardiogram or CT angiography in ED dept can be used to confirm the final diagnosis, CT angiography being the most common.
Treatment
An urgent surgical consultation is arranged upon suspicion of an aortic dissection, ascending aortic dissections require emergency surgeries. Initial effort is made to slow down the dissection and put less pressure on the aortic wall.
Strong analgesic and beta blockers are administered to the patient. Alternatives to beta blockers are used as a result of contraindication or aortic regurgitation, including esmolol, labetalol or diltiazem. Patients with low blood pressure should be given intravenous fluids and vasopressors. Mental activity and urine output should not be affected.
The surgery for a type A consists of closing the intimal tear and inserting a synthetic vascular graft to aid the reconstruction of the aorta. Endovascular stent-grafting has been used as a less invasive method, usually for type B aortic dissections of a more complex nature. The surgical procedure may last from 5 to 10 hours but the recovery period varies per individual.
References
- LeMaire, S., Russell, L. Epidemiology of thoracic aortic dissection. Nat Rev Cardiol 8, 103–113 (2011). https://doi.org/10.1038/nrcardio.2010.187
- Levy D, Goyal A, Grigorova Y, et al. Aortic Dissection. [Updated 2021 Dec 15]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441963/
- Anon, Aortic dissection. Aortic dissection - Symptoms, diagnosis and treatment | BMJ Best Practice. Available at: https://bestpractice.bmj.com/topics/en-gb/3000226.
- Anon, Aortic dissection. Heart Research UK. Available at: https://heartresearch.org.uk/aortic-dissection/.
- Grant & Griffin, 2013. Hemopericardium. In Grainger & Allison's Diagnostic Radiology Essentials. Elsevier. Available at: https://www.sciencedirect.com/topics/medicine-and-dentistry/hemopericardium
- Knott, D.L., 2020. Aortic dissection: Causes, symptoms and treatment. Patient.info. Available at: https://patient.info/heart-health/abdominal-aortic-aneurysm/aortic-dissection.
- Criado, F.J., 2011. Aortic dissection: a 250-year perspective. Texas Heart Institute Journal, 38(6), p.694. Available at: https://www.ncbi.nlm.nih.gov/books/NBK441963/
Further Reading
Last Updated: Jul 27, 2022