Introduction
What are exerkines?
How does exercise trigger exerkine release?
Major exerkines and their effects
How do exerkines promote wellness and prevent disease?
Exerkines in disease prevention and potential therapeutics
References
Further reading
Discover how the body’s own exercise-triggered signaling molecules orchestrate cross-organ communication to enhance resilience, regulate metabolism, and protect against chronic disease.
 Image Credit: Roman Samborskyi / Shutterstock.com
Image Credit: Roman Samborskyi / Shutterstock.com
Introduction
This article examines how exercise-induced exerkines coordinate inter-organ communication to prevent disease, as well as promote metabolic, cardiovascular, immune, and neurological health. Exerkines are now recognized as key molecular mediators underlying the systemic adaptations to both acute and chronic exercise training. Rather than acting in isolation, exerkines function within an integrated, multi-organ signaling network that underpins exercise-induced resilience and healthspan1,2.
What are exerkines?
Exerkines are a diverse group of bioactive signaling molecules released from various tissues in response to acute or chronic physical exercise. These molecules, which range from proteins, peptides, and lipids to metabolites and nucleic acids, interact through endocrine, paracrine, or autocrine pathways to coordinate communication between skeletal muscle, adipose tissue, liver, heart, and brain. Importantly, their effects are context-dependent and influenced by exercise modality, duration, intensity, and the physiological state of the individual1,2.
When first described following the discovery of exercise-induced interleukin-6 (IL-6) release from contracting skeletal muscle in 20001, the concept centered on myokines. However, the term “exerkines” now encompasses signaling molecules released from multiple organs - including skeletal muscle (myokines), heart (cardiokines), liver (hepatokines), white and brown adipose tissue (adipokines and batokines), and neurons (neurokines). Exerkines also comprise lipids, metabolites, nucleic acids, and micro ribonucleic acids (microRNAs). Current research emphasizes that exerkines extend beyond classical cytokines to include metabolites and lipid mediators that collectively reshape systemic metabolism1,2.
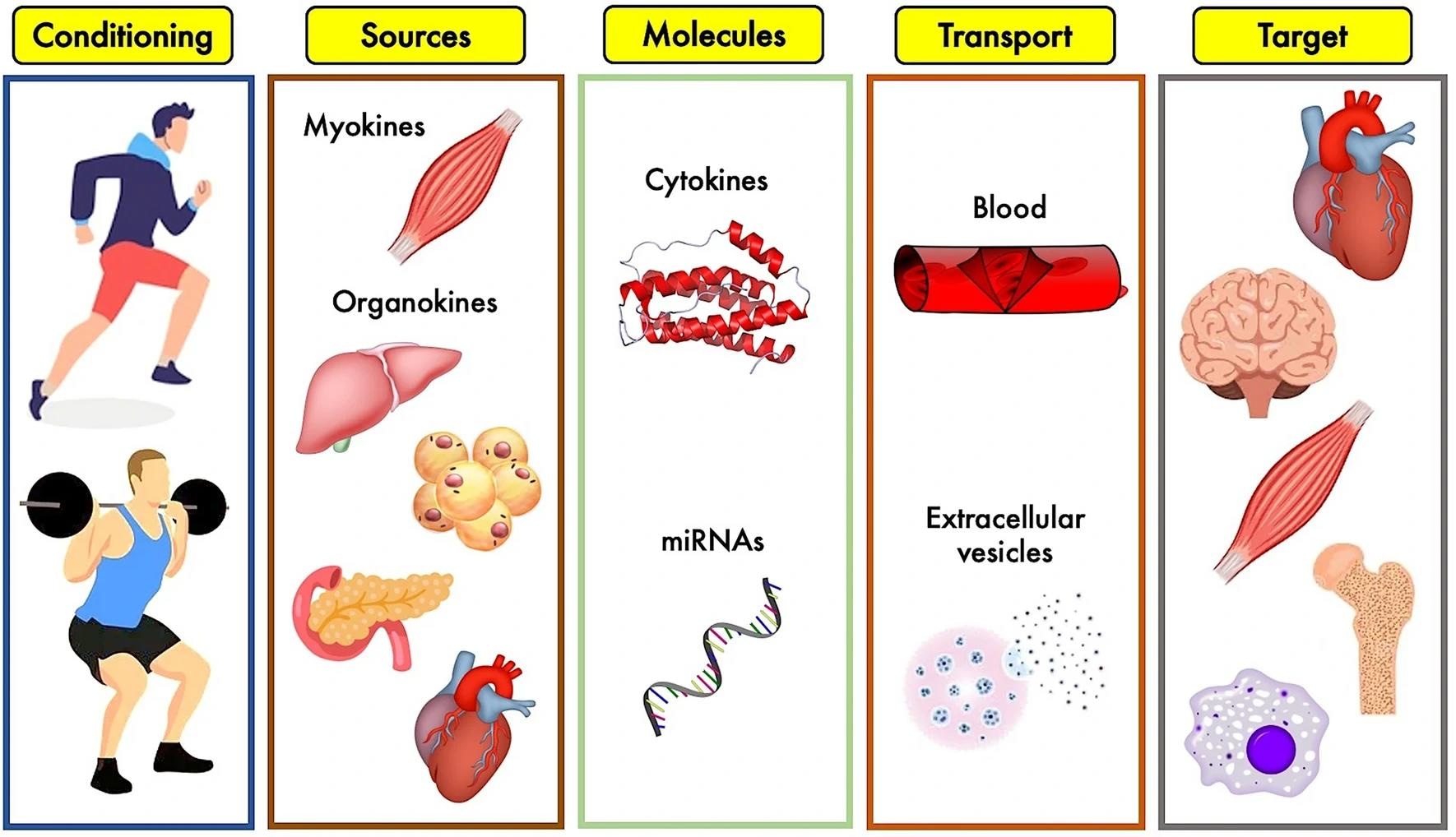
Overview of the exerkines’ system: different types of physical exercise stimulate biological tissues to release a plethora of cytokines and other molecules which, circulating into blood stream or stuffed into extracellular vesicles, reach biological targets to produce specific effects.3
How does exercise trigger exerkine release?
Exercise acts as a potent biological stimulus that induces the release of exerkines through mechanical, metabolic, neural, and hormonal signals generated during physical activity. During exercise, contraction-induced calcium flux, altered cellular energy status (e.g., AMP-activated protein kinase activation), hypoxia-related signaling, and inflammatory cues collectively stimulate exerkine production and secretion1,3. These signals differ between endurance and resistance exercise, leading to partially distinct exerkine signatures1,2.
Exerkines may be released directly into the circulation or packaged within extracellular vesicles (EVs), including exosomes, which facilitate inter-organ communication1,3. Acute exercise leads to rapid and transient rises in circulating exerkines that maintain metabolic homeostasis, regulate inflammation, and adjust substrate utilization during and immediately after exercise2. In contrast, chronic training promotes longer-term adaptations associated with improved metabolic flexibility and reduced systemic inflammation2. Exercise timing and circadian alignment may further modulate exerkine secretion and tissue responsiveness1.
Notably, several factors affect the magnitude and composition of exerkine release, including exercise modality, intensity and duration, nutritional status, circadian timing, age, sex, and an individual’s training status or genetic background1,2. Inter-individual variability is substantial, and some individuals exhibit attenuated or distinct molecular responses (“non-responders”) to similar exercise stimuli1.
 Image Credit: Cheer-J-ane / Shutterstock.com
Image Credit: Cheer-J-ane / Shutterstock.com
Major exerkines and their effects
Muscle-derived exerkines
Skeletal muscle is a major source of exerkines released during contraction, with interleukin-6 (IL-6) the most extensively studied myokine. Acute exercise leads to a rapid increase in IL-6 levels, during which these molecules act as endocrine and paracrine regulators to enhance glucose uptake, stimulate lipid oxidation, and promote anti-inflammatory pathways by inducing cytokines such as IL-10 and the IL-1 receptor antagonist (IL-1RA)1. Although initially considered pro-inflammatory, exercise-induced IL-6 exerts predominantly metabolic and anti-inflammatory effects in this context1,2.
Skeletal muscle produces several other factors, including apelin, an exerkine that increases with exercise and declines with age. Experimental studies in rodents demonstrate that apelin enhances mitochondrial biogenesis, autophagy, and muscle regenerative capacity via activation of the APJ receptor, and may counteract age-associated sarcopenia. However, much of this mechanistic evidence derives from animal models, and translation to human physiology remains under investigation3.
Fat tissue exerkines
Adipose tissue contributes to the exerkine network through adipokines and lipid mediators. Adiponectin, which is released from white adipose tissue, improves insulin sensitivity and reduces inflammation2. Comparatively, brown adipose tissue secretes 12,13-dihydroxy-9Z-octadecenoic acid (12,13-diHOME) in response to exercise, which enhances skeletal muscle fatty acid uptake and oxidation to improve cardiometabolic profiles1,2. Metabolite-derived exerkines such as β-aminoisobutyric acid (BAIBA) further illustrate the metabolic breadth of exercise-induced signaling1.
Irisin, meteorin-like (METRNL), and fibroblast growth factor 21 (FGF-21) are other crucial exerkines that provide a wide range of systemic benefits. FGF21 and METRNL have been implicated in metabolic adaptation, adipose tissue remodeling, and improved insulin sensitivity following endurance exercise1,2. However, circulating FGF21 responses to chronic training appear variable and may depend on metabolic health status1. In addition to facilitating the browning of adipose tissue, irisin promotes cognitive function, bone metabolism, and glucose homeostasis2.
Heart & cardiovascular exerkines
The production of vascular endothelial growth factor (VEGF) by muscle and endothelial cells induces angiogenesis, thereby enhancing tissue perfusion1. Nitric oxide, produced by endothelial cells, improves vascular tone and endothelial function, both of which support cardiovascular health2. Exercise also modulates circulating growth differentiation factor 15 (GDF-15), a stress-responsive cytokine with multi-organ metabolic effects observed following acute endurance exercise in humans. The precise tissue source of exercise-induced GDF-15 remains incompletely defined3.
Liver & metabolic exerkines
The liver secretes numerous hepatokines that regulate systemic metabolism during exercise, including IL-6, FGF-21, fetuin-A, and angiopoietin-like 4 (ANGPTL4). Acute and chronic exercise can elevate circulating FGF21 levels, which modulate glucose and lipid metabolism by enhancing insulin sensitivity and influencing triglyceride handling1,2. ANGPTL family members also regulate tissue-specific lipid trafficking during and after exercise1.
Extracellular vesicles & microRNAs
Exercise alters the release and cargo composition of extracellular vesicles, including exosomes enriched with proteins and microRNAs1,3. Specific exercise-responsive microRNAs, such as miR-1192 (identified in trained mice) and miR-342-5p (observed in young trained adults), have been associated with cardioprotective signaling pathways. These emerging exerkines are promising but should be considered investigational until validated across larger human cohorts3. These vesicles act as carriers of exerkines that enable inter-organ crosstalk involved in metabolic regulation and coordinated cellular adaptation to physical activity.
 Image Credit: PeopleImages / Shutterstock.com
Image Credit: PeopleImages / Shutterstock.com
Exerkines, including IL-6, adiponectin, FGF21, and lipid mediators, promote glucose uptake, fatty acid oxidation, lipid clearance, and insulin sensitivity to reduce the risks of obesity, type II diabetes, and cardiovascular disease. Exerkines also mediate immune modulation by eliciting a transient pro-inflammatory response during acute exercise, followed by enhanced anti-inflammatory signaling, thereby contributing to improved immune surveillance and long-term immune health. These immune-metabolic interactions highlight the integrated nature of exercise-induced adaptations1,2.
In the nervous system, exercise-induced exerkines, including brain-derived neurotrophic factor (BDNF) and circulating microRNAs, support neuroplasticity, cognition, and protection against neurodegeneration. Collectively, these actions contribute to healthy aging, as chronic exercise-associated exerkine profiles are linked to reduced low-grade inflammation, preserved metabolic flexibility, enhanced tissue repair, and improved systemic resilience. Such adaptations likely reflect coordinated signaling across muscle, liver, adipose tissue, vasculature, and immune cells rather than the action of any single molecule1,2.
Exerkines in disease prevention and potential therapeutics
Exercise-induced exerkines enhance glucose regulation, lipid handling, and insulin sensitivity, thereby reducing the risk of metabolic and cardiovascular diseases. Anti-inflammatory exerkines reduce chronic low-grade inflammation, whereas vascular and metabolic exerkines support endothelial function and energy balance. Emerging evidence suggests that exercise-modulated exerkines may influence tumor biology through immune and metabolic mechanisms1,2. For example, preclinical evidence indicates that oxytocin released from muscle cells may exert autocrine and paracrine effects on muscle metabolism, with broader systemic implications that remain under investigation3.
Circulating exerkine profiles may also serve as biomarkers of exercise responsiveness and cardiometabolic health1. Large-scale multi-omics initiatives, such as the Molecular Transducers of Physical Activity Consortium (MoTrPAC), aim to systematically map exercise-induced molecular signatures across tissues to improve precision exercise medicine. Future research must clarify dose-response relationships, temporal dynamics, and inter-individual variability to translate exerkine biology into clinical practice1,2.
References
- Chow, L. S., Gerszten, R. E., Taylor, J. M., et al. (2022). Exerkines in health, resilience and disease. Nature Reviews Endocrinology 18; 273-289. DOI: 10.1038/s41574-022-00641-2. https://www.nature.com/articles/s41574-022-00641-2
- Novelli, G., Calcaterra, G., Casciani, F., et al. (2024). ‘Exerkines’: A Comprehensive Term for the Factors Produced in Response to Exercise. Biomedicines 12(9). DOI: 10.3390/biomedicines12091975. https://www.mdpi.com/2227-9059/12/9/1975
- Magliulo, L., Bondi, D., Pini, N., et al. (2022). The wonder exerkines - novel insights: a critical state-of-the-art review. Molecular and Cellular Biochemistry 477(1); 105-113. DOI: 10.1007/s11010-021-04264-5. https://link.springer.com/article/10.1007/s11010-021-04264-5
Further Reading
Last Updated: Mar 2, 2026