Introduction
Symptoms
Obtaining a diagnosis
Treatment
References
Further Reading
Adenomyosis is a condition that affects the uterus and is histologically characterized by ectopic endometrial glands and stroma located in the myometrial, which are surrounded by hypertrophic (a lodgement caused by the increase in the size of cells) and hyperplastic (enlargement caused by an increase in cell proliferation) myometrial changes. The myometrium is the middle layer of the uterine wall, which is comprised of smooth muscle cells and controls the contraction of the uterus.

Image Credit: B-D-S Piotr Marcinski/Shutterstock
Adenomyosis is most common in the 4th and 5th decades of life. Moreover, adenomyosis is a difficult condition to diagnose as it is commonly misdiagnosed as endometriosis. This can be confounded by the high prevalence of co-occurrence with adenomyosis; 80% of women with endometriosis also have adenomyosis. women with endometriosis, however, present symptoms much earlier in life, most commonly in the 3rd and 4th decades. The condition is also more common in multiparous women - those who have had one or more previous births.
Symptoms
The symptomatology of adenomyosis is not well understood due to the late development of technologies that now enable diagnosis. Before the advent of magnetic resonance imaging, or high-quality transvaginal ultrasound, adenomyosis was difficult to identify. In addition, the majority of women who remain asymptomatic are diagnosed with the condition only after being identified incidentally following an imaging procedure.
Adenomyosis also coexists with several conditions other than endometriosis, therefore the overlapping of related symptoms complicates diagnosis. The most common of these other conditions are uterine fibroids.
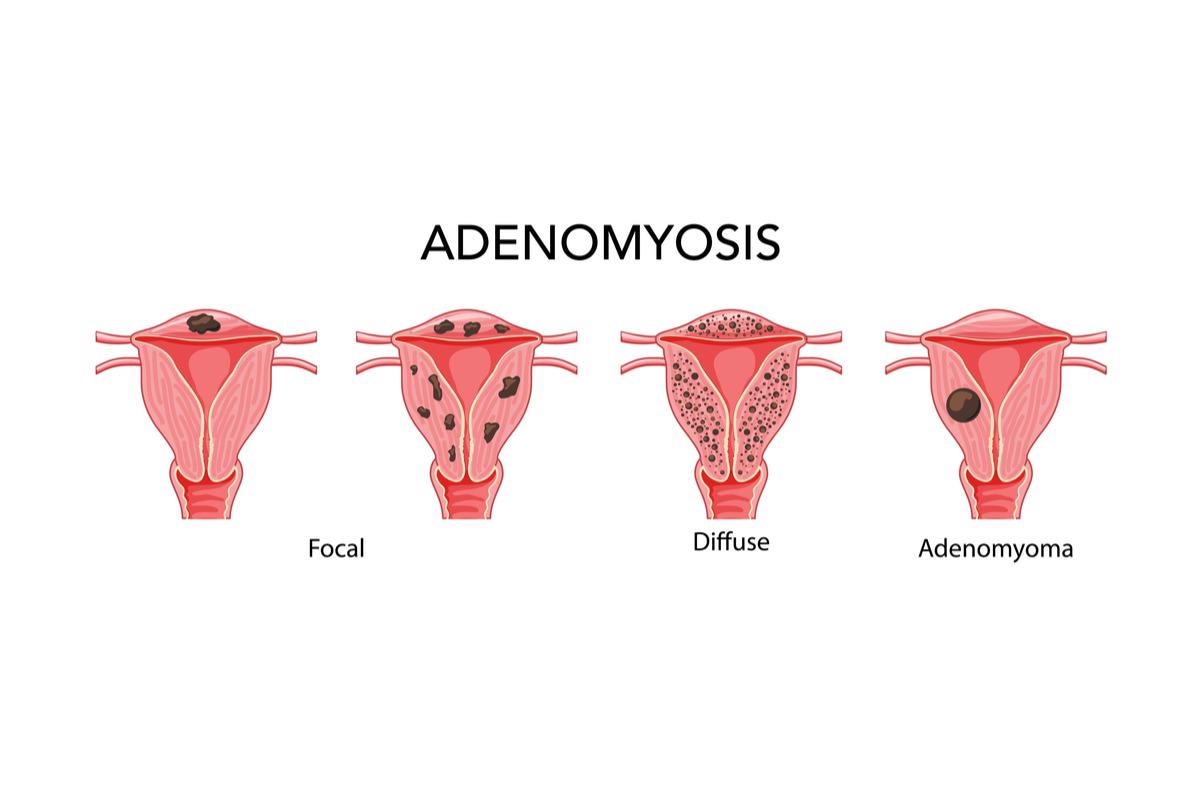
Image Credit: Vectoressa/Shutterstock
Obtaining a diagnosis
A diagnosis is typically made as a result of viewing the following:
- Sub endometrial echogenic striations: invasion of the endometrial glands into the sub endometrial tissue
- Globular shape: ultrasound features of a globular uterus with ill-defined adenomyotic lesions
- Myometrial cysts: cysts present in the myometrium
Subendometrial striations positively predict the presence of adenomyosis in up to 80% of cases. The myometrial cysts present as a result of the endometrial glands cycling. During the reproductive phase, the superficial area of the endometrium, the endometrium functionalis, shifts through cycles of secretion and proliferation. During the menstrual phase, the endometrium undergoes desquamation (shedding) in the absence of any ovum implantation.
Cysts are all between 1 and 5mm in size. Other features, which can be viewed via imaging methods, include asymmetric thickening of the uterine wall, an inability to distinguish the interface of the endometrium and the myometrium, and the formation of adenomyomas. Adenomyomas are benign nodes/lesions comprised of an aggregate of benign endometrial glands. These are poorly defined and have features that overlap with fibroids.
The diagnostic sensitivity ultrasound results in suboptimal imaging, ranging from 50 to 87%; consequently, magnetic resonance is considered to be a superior tool that can show the presence of adenomyomas more clearly.

 Read Next: Endometriosis Treatments
Read Next: Endometriosis Treatments
Treatment
The most definitive cure for adenomyosis is hysterectomy. However, this is typically less desirable as this eliminates fertility.
Medical treatment for adenomyosis follows the same approaches that are used to treat endometriosis; that is to reduce the production of endogenous estrogen or to induce differentiation of the endometrium with progestins. In addition, treatment may also serve to suppress menstruation. The types of therapies used are based on the targeting of the hypothalamic-pituitary-gonadal axis as this governs mammalian reproduction. These include gonadotrophin-releasing hormone agonist (GnRH agonist), oral contraceptives (OCs), progestins, and danazol. More recently, selective progesterone receptor modulators (SPRMs), selective estrogen receptor modulators (SERMs), or aromatase inhibitors (AIs) are being used.
Nonsteroidal anti-inflammatory drugs (NSAIDs) are also one of the primary medical therapies employed to treat adenomyosis. NSAIDs inhibit cyclooxygenase (COX)-1 and/or COX-2 activity, which produce prostaglandins, lipid compounds implicated in the inflammatory processes. They, therefore, serve to alleviate pain.
It is important to note that treatment for adenomyosis is aimed at treating the symptoms, and no reduction in the lesions occurs via the use of any drug, at any dose; after treatment discontinuation, lesions resume their metabolic activity. Moreover, medical treatments are associated with adverse events in the long term. As is the case with endometriosis no treatment is universally effective in the case of adenomyosis.
In the case of failure to respond to medication, several interventional radiologic procedures may be used. MRI-guided and ultrasound-guided high-intensity ultrasound thermal celeb intense elation can be carried out to target the disease. By embolizing the uterine artery, blood flow to the uterus is reduced; this consequently results in tissue necrosis, leading to a decrease in the size of the uterus. While promising, these treatments require additional data on direct treatment comparisons as well as outcomes. In the long-term, these procedures are effective in cases where fertility wants to be preserved.
For patients who do not prioritize future fertility, endometrial ablation may be considered. This procedure is a less invasive alternative relative to hysterectomy. However deeper adenomyotic cysts are not able to be targeted.
The most invasive treatments are myomectomy and partial hysterectomy; these procedures allow fertility to be preserved.
While effective at targeting deeper cysts, there is a risk of future scarring which can cause disease recurrence as these procedures disrupt the interface of the endometrium and myometrium (which is a risk factor for adenomyosis). These procedures also increase the risk of future pregnancy complications combat with an increased risk of rupturing of the membrane, premature labor, and spontaneous abortion.
Adenomyosis continues to remain a challenge to identify and treat due to the lack of research and failure to develop and adopt standardized diagnostic criteria, particularly in patients who wish to retain their fertility. Noninvasive diagnostics such as imaging and biopsies of the myometrial are continuing to improve; consequently, the likelihood of developing standardized criteria for diagnosis is increasing. researchers suggest that standardized histopathologic and imaging diagnoses are an unmet need and present the main barrier to obtaining a full understanding of adenomyosis.
Adenomyosis of the uterus. Image Credit: Lisa Culton/Shutterstock
References
- Taran FA, Stewart EA, Brucker S. (2013) Adenomyosis: Epidemiology, Risk Factors, Clinical Phenotype and Surgical and Interventional Alternatives to Hysterectomy. doi:10.1055/s-0033-1350840.
- Kim MD, Won JW, Lee DY, et al. (2004) Uterine artery embolization for adenomyosis without fibroids. Clin Radiol. doi:10.1016/j.crad.2003.11.018.
- Liang E, Brown B, Kirsop R, et al. (2012) Efficacy of uterine artery embolisation for treatment of symptomatic fibroids and adenomyosis - an interim report on an Australian experience. Aust N Z J Obstet Gynaecol. doi:10.1111/j.1479-828X.2011.01399.x.
- Zhou M, Chen JY, Tang LD, et al. (2011) Ultrasound-guided high-intensity focused ultrasound ablation for adenomyosis: the clinical experience of a single center. Fertil Steril. doi:10.1016/j.fertnstert.2010.10.020.
- Marjoribanks J, Proctor ML, Farquhar C. (2003)Nonsteroidal anti-inflammatory drugs for primary dysmenorrhoea. Cochrane Database Syst Rev. doi:10.1002/14651858.CD001751.
- Takeuchi M, Matsuzaki K. (2011) Adenomyosis: usual and unusual imaging manifestations, pitfalls, and problem-solving MR imaging techniques. Radiographics. doi:10.1148/rg.311105110. PMID: 21257936.
- Reinhold C, McCarthy S, Bret PM, et al. (1996) Diffuse adenomyosis: comparison of endovaginal US and MR imaging with histopathologic correlation. Radiology. doi:10.1148/radiology.199.1.8633139.
- Garcia L, Isaacson K. (2011) Adenomyosis: a review of the literature. J Minim Invasive Gynecol. doi:10.1016/j.jmig.2011.04.004.
Further Reading
Last Updated: May 23, 2022