Nanomaterials (NMs) can now be found in a wide range of different products, including electronic devices, food, cosmetic products, and medicine.
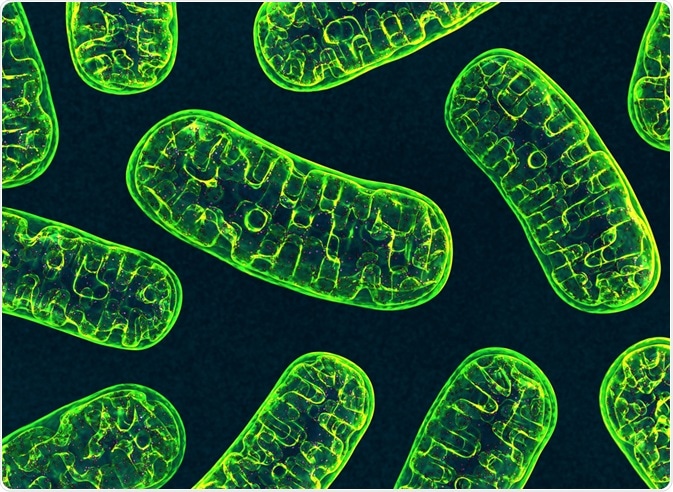 Image Credits: 3d_man / Shutterstock.com
Image Credits: 3d_man / Shutterstock.com
As a result of its growing use and worldwide demand, the growing number of individuals exposed to NMs through their production, processing, transportation, application, disposal, and subsequent release has pushed researchers to gain a deeper understanding as to how these substances might affect human, animal, and environmental health.
What are mitochondria?
Often referred to as the “powerhouse” of the cell, the mitochondrion is a dual membrane-bound organelle that also contains its own genome. Whereas the outer membrane of the mitochondria is responsible for the selective passage of both ions and molecules into and out of this organelle, the inner membrane exhibits a highly restrictive permeability to ensure that both the electron transport chain (ETC) and adenosine triphosphate (ATP) synthesis continue to proceed without any interference from other molecules.
In addition to its role in the production of ATP, the mitochondrial genome, while much smaller than that found in the nucleus, houses several important genes responsible for encoding rRNA and tRNA, as well as several proteins involved in both ETC and ATP synthesis. Mitochondria also play a role in several other biological processes including ion exchange, metabolite transport, calcium signaling, and the initiation of apoptosis.
What is mitochondrial toxicity?
Some of the earliest known chemicals that were found to specifically target the mitochondria and exert toxic effects included oligomycin, 2,4-dinitrophenol, pentachlorophenol, and carbon monoxide. Since then, a significant amount of research has been done to not only understand the specific functions of the mitochondria but also how this highly complex organelle responds to stress.
In fact, most in vitro toxicity screening assays that are performed early on in the chemical discovery process will often initially begin with analyses of possible mitochondrial toxicity effects, particularly via changes in this organelle’s membrane potential.
While numerous, some of the most cutting-edge focuses in mitochondria toxicology research include how chemicals might change the concentration of reactive oxygen species (ROS), most of which are normally produced in the mitochondria.
While mitochondria within the cell exhibit similar defense systems that are present in other organelles, they can be vulnerable following exposure to robust stress. Therefore, researchers are often interested in understanding how altered mitochondrial homeostasis could result in certain toxic endpoints.
How do nanomaterials cause mitochondrial toxicity?
To date, researchers have confirmed the mitochondrial toxicity of some NMs, commonly shown to induce alterations the morphology of this organelle, reducing its membrane potential and respiratory function, as well as by promoting the release of cytochrome C. Aside from these effects, one of the most notable mechanisms of NM-induced mitochondrial toxicity is due to the generation of ROS.
The overproduction of ROS following NM exposure can occur through either direct damage to the mitochondria, which subsequently leads to several toxic effects, or following a reduction in the activity of the mitochondrial respiratory chain complexes, which can lead to the generation of ROS.
ROS are highly reactive chemical agents that can cause a wide range of deleterious damage to cellular macromolecules. In addition to the inherent adverse effects associated with high levels of ROS, the ability of NMs to alter mitochondrial dynamics in this way can ultimately lead to changes in ATP production levels and lowered mitochondrial membrane potential (MMP) levels.
Which nanomaterials induce mitochondrial toxicity?
Several different types of NMs have been thoroughly evaluated for their ability to induce toxic effects on mitochondria. Metal and metal oxide nanoparticles, both of which account for approximately 37% of the products that are made with NMs around the world, have been studied for their ability to cause changes in the mitochondria.
Titanium oxide (TiO2) nanoparticles (NPs), for example, have been found to induce mitochondrial toxicity in various in vitro models through several different mechanisms, some of which include reduced MMP, depolarization of the mitochondrial membrane, increased cytochrome C release, mitochondrial swelling, mitophagy activation, and much more.
Similarly, silver nanoparticles (Ag-NPs) have also been shown to reduce MMP and ATP production, as well as promote mitochondrial fragmentation in several in vitro models.
Conclusion
Clearly, there is a sufficient amount of scientific evidence that suggests the relationship between exposure to NMs and altered mitochondrial function. Although there remains a need for further in vivo studies to confirm the results acquired in the preliminary in vitro studies cited here, it is crucial for these toxic effects to be taken seriously among the industries that produce nanoproducts.
Sources
- “Mitochondria” – Scitable by Nature Education
- Meyer, J. N., Hartman, J. H., & Mello, D. F. (2018). Mitochondrial Toxicity. Toxicological Sciences 162(1); 15-23. DOI: 10.1093/toxsci/kfy008.
- Wu, D., Ma, Y., Cao, Y., & Zhang, T. (2020). Mitochondrial toxicity of nanomaterials. Science of The Total Environment 702(1). DOI: 10/1016/j.scitotenv.2019.134994.
- Natarajan, V., Wilson, C. L., Hayward, S. L., & Kidambi, S. (2015). Titanium Dioxide Nanoparticles Trigger Loss of Function and Pertubation of Mitochondrial Dynamics in Primary Hepatocytes. PLoS one. DOI: 10.1371/journal.pone.0134541.
Further Reading
Last Updated: Mar 9, 2020