Making Life’s Reactions go Faster
In living organisms, there are many chemical reactions taking place. These reactions have many different functions, including breaking down the food we eat and getting energy from the broken down food. To facilitate this, there are a group of proteins known as enzymes.
Enzymes speed up (or catalyze) these chemical reactions, working with only one substrate to catalyze one reaction. Many enzymes come together to form pathways, leading to a variety of functions, such as muscle contraction.
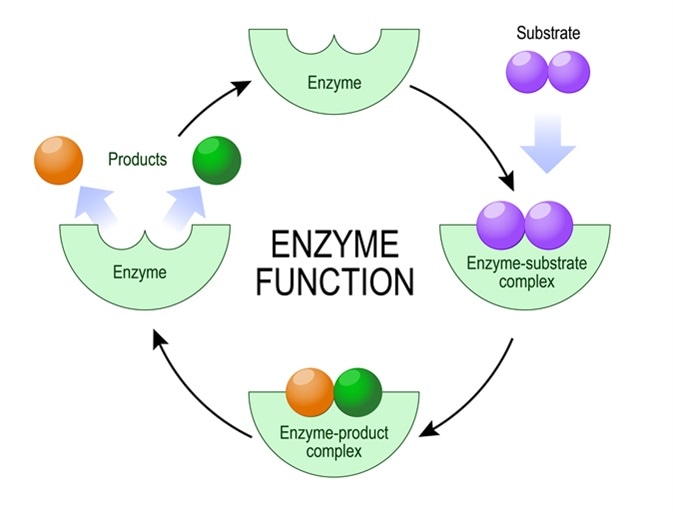
Enzyme function. Image Credit: Designua / Shutterstock
Additional Factors
Some enzymes require the addition of another non-protein molecule to function as an enzyme. These are known as cofactors, and without these enzymes remain within the inactive “apoenzyme” forms. Once the cofactor is added, the enzyme becomes the active “holoenzyme”.
Cofactors can either be ions, such as zinc and iron ions, or organic molecules, such as vitamins or vitamin-derived molecules. Many of these cofactors will attach near the substrate binding site to facilitate the binding of the substrate to the enzyme. Cofactors can be classed as “prosthetic groups” or “coenzymes” depending on how tightly they are bound to the enzyme; coenzymes bind more loosely to the enzyme, and are thus modified during the enzymatic reaction, while prosthetic groups are more tightly bound to the enzyme and are not modified.
Enzymes co-factors and inhibitors
Prosthetic Groups
These can be ions, such as Zn2+ ions used in dehydrogenase enzymes or Fe2+ ions used in alkaline phosphatases. Molecules such as tryptophan tryptophylquinone (TTQ) act as a prosthetic group in reactions catalyzed by methylamine dehydrogenase. Another molecule, flavin adenine dinucleotide (FAD), can be remade during the enzymatic reaction, and therefore can be considered to be a prosthetic group as its overall concentration does not change.
Transferring Molecules by the use of Coenzymes
Some coenzymes will transfer a molecule during the enzymatic reaction. For example, coenzyme A transfers acyl groups. Thiamin pyrophosphate is another coenzyme, which transfers aldehydes. Pyridoxal phosphate transfers amino groups, biotin transfers carbon dioxide (CO2) and carbamide coenzymes transfer alkyl groups. Other coenzymes, such as nicotine adenine dinucleotide (NAD), coenzyme Q and FAD transfer electrons or hydrogen atoms.
From Vitamins to Cofactors
Vitamin based cofactors are essential for processes such as vision, blood clotting and hormone production.
- Retinal, the aldehyde version of vitamin A, are cofactors for opsins which are present in the eye. Opsins are apoproteins which are responsible for vision; rhodopsin is needed for dim-light vision, and iodopsin is needed for bright-light and color vision.
- Thiamin pyrophosphate, mentioned above, is a coenzyme derived from thiamin, or vitamin B1. Thiamin pyrophosphate is a cofactor in enzymes which catalyze oxidative decarboxylation and transketolase reactions.
- Riboflavin, or vitamin B2, is the precursor not only for FAD but also flavin mononucleotide (FMN). As mentioned above, FAD transfers electrons, and this is also the case for FMN. These coenzymes also carry hydrogen atoms for oxidation reactions in the citric acid cycle and the electron transport chain.
- Vitamin B6 gives rise to the coenzymes pyridoxal phosphate and pyridoxamine phosphate. As many as 120 enzymes require one of these coenzymes as cofactors; these include decarboxylases, dehydratases, desulfydrases, racemases, synthases, and transaminases. Pyridoxal phosphate and pyridoxamine phosphate are also involved in the break-down of amino acids, the building blocks of proteins.
- Vitamin B12, or cobalamin, are modified to become the coenzymes methylcobalamin and deoxyadenosylcobalamin. These coenzymes are required in the enzymes which change homocysteine to methionine, an amino acid, and in the oxidation of amino acids and fatty acids. As well as this, these coenzymes assist the removal of a methyl group from methyl folate, which is needed to regenerate tetrahydrofolate, another coenzyme.
- Ascorbic acid, or vitamin C, is used as a cofactor in hydroxylases. One hydroxylase which needs vitamin C is the enzyme which catalyze the hydroxylation of proline and lysine, two amino acids. This creates cross-links in collagen, making its structure stable. Formation of bile acids from cholesterol also requires the presence of vitamin C as a cofactor.
- Gamma-carboxylases can require vitamin K as a cofactor. This transfers CO2, and leaves a carboxylic acid group which is able to bind calcium. Gamma-carboxylases catalyze the formation of osteocalcin, which is a protein responsible for bone remodeling, as well as the formation of prothrombin, which is needed for the blood to clot.
Further Reading
Last Updated: Jul 18, 2023