Hologic, Inc.today announced the U.S. Food and Drug Administration (FDA) has approved the Company's premarket approval (PMA) application for the Adiana permanent contraception system.
The Adiana system is designed to provide women a minimally-invasive, non-incision alternative to traditional, surgical means of permanent contraception. In January 2009, Hologic received CE marking approval for the Adiana system and commenced marketing and sales of this product in certain European countries.
The Adiana permanent contraception procedure is minimally invasive, requires no incisions and can be performed in the comfort of the doctor's office using local anesthesia. Patients are normally able to return to work or resume their daily activities within one day. In contrast, traditional methods of permanent contraception, such as tubal ligation, require more invasive surgical procedures, usually are conducted in a hospital under general anesthesia and typically require four to five days of recovery. As a result, these more invasive surgical procedures can pose serious risk of complications, including anesthesia-related problems and damage to organs or blood vessels. There were approximately 70,000 female transcervical contraception procedures performed in the U.S. last year.
"While tubal ligation is the most common form of permanent contraception used throughout the world, hysteroscopic procedures such as those employing the Adiana permanent contraception system enable women to choose lifetime protection from pregnancy without the risks of general anesthesia and the longer recovery periods associated with traditional sterilization procedures," said Ted Anderson, M.D., Ph.D., associate professor of obstetrics and gynecology at Vanderbilt University, and clinical investigator of the pivotal clinical trial for the Adiana system. "The Adiana procedure is a safe and simple alternative that not only provides patients with peace of mind in preventing pregnancy, but also leaves nothing in the uterus that might limit women's options for future gynecologic tests and procedures." 
The Adiana procedure is indicated for women who desire permanent birth control by occlusion of the fallopian tubes. Ideal candidates include women who do not want children in the future and would like the peace of mind and convenience of permanent birth control. The procedure may also be an ideal solution for women who desire permanent birth control but are poor candidates for surgery. 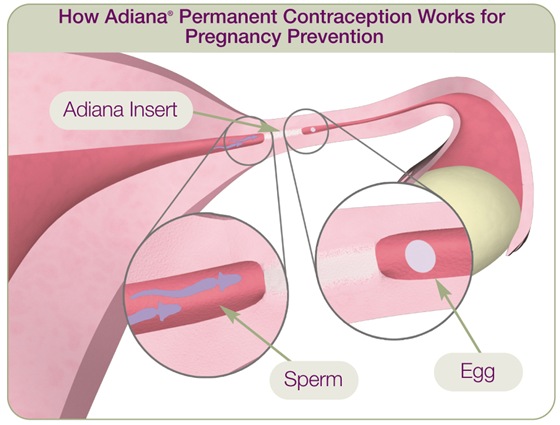
"Many women seek relief from the uncertainty and hassle of temporary birth control methods once they know their childbearing is complete. However, few know that permanent contraception is available without incisions, the use of general anesthesia, or need for lengthy hospital stays," said Tony Kingsley, senior vice president, GYN surgical products at Hologic. "With the Adiana permanent contraception system, we are very excited to be able to offer these women a new alternative for their long-term contraception needs."
During the Adiana procedure, a slender, flexible instrument is passed through the body's natural openings to deliver a low level of radiofrequency (RF) energy to a small section of each fallopian tube. A tiny, soft insert, about the size of a grain of rice, is then placed in each fallopian tube in the location where the energy was applied. During the three months following the procedure, the patient continues to use temporary birth control while new tissue grows in and around the Adiana inserts, eventually blocking the fallopian tubes. At three months, a special x-ray test (called a hysterosalpingogram or HSG) is performed to confirm the fallopian tubes are completely blocked and the patient may begin relying on Adiana for permanent contraception.
"This is another significant milestone for Hologic and our GYN surgical products business," said Tony Kingsley. "With FDA approval of the Adiana system, we are now able to provide physicians with another non-hormone, minimally-invasive therapy that addresses a significant issue for women in their post-childbearing years."
January 2009, "U.S. Markets for Women's Health Devices," iData Research, Vancouver, British Columbia, Canada. iData is an international market research and consulting group.