Melatonin is a hormone produced in the pineal gland from the essential amino acid tryptophan during darkness.
The production of melatonin is controlled by a part of the brain called the suprachiasmatic nucleus (SCN) and its timing fine-tuned by our exposure to light during the morning.
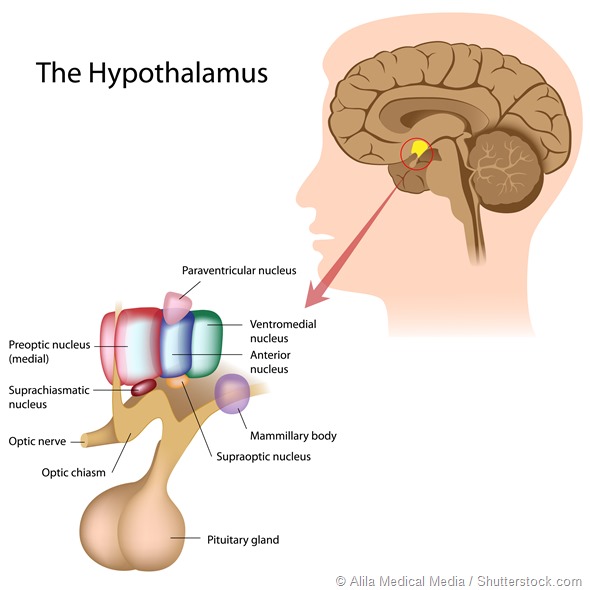
The SCN is often called our biological clock because it is responsible for the timing of many functions in our body including the timing of our sleep onset and awakening.
Interestingly melatonin plays a role in the timing of its own production through specific receptors in the SCN.
As such then, when people take a melatonin tablet in the early evening, it may facilitate an earlier production of melatonin and an earlier onset of sleep. When taken in this manner, however, it is rapidly removed from the bloodstream and has little or no effect on sleep itself, unlike most sleeping pills.
How much research has been carried out on melatonin to date and what areas have studies focused on?
A search of the website PubMed for melatonin reveals more than 20,000 scientific papers and reviews have been published on melatonin or mentioned it since it was discovered in 1958.
The breadth of research on melatonin is quite staggering, ranging from its role in reproduction in various species, including farm animals and humans, through to effects on cancer cells, blood pressure, glucose metabolism and sleep timing.
How has our understanding of melatonin changed over the past 40 years that you have been researching the hormone?
Our understanding of the role that melatonin plays in our physiology increased dramatically following the cloning of the first melatonin receptor gene in 1994 by Steve Reppert and his colleagues.
Although there are still many questions to be answered about how occupation of the receptor by the hormone brings about changes in cells, it is clear that melatonin has an important role in signalling the time of day to various organs in the body. In other words it is a chemical signal of the time of day.
How animals including humans use this information appears to be species specific, with some animals using the change in duration of secretion of melatonin over the year used to time puberty and fertility.
The role of melatonin in human fertility is controversial but as the eminent melatonin researcher Russel Reiter has said, to assume it would not have some sexual effects in humans would almost seem naive.
Along with actions on its receptors, melatonin has in recent years been found to be an anti-oxidant and a wide range of consequences of melatonin secretion and benefits of melatonin administration have been promoted.
Is it true that circadian disruption can exacerbate certain diseases? How much evidence is there for this?
There is a now considerable body of evidence that disruption of circadian rhythms is detrimental to the well-being of animals and humans.
Either total or tissue specific deletion of genes responsible for cellular rhythmicity, the “clock genes”, results in multiple changes in the physiology of the animal. These range from loss of melatonin rhythmicity, impaired glucose control, obesity, bone function, sleep-wake activity and increased incidence of some cancers.
Similarly when rhythms are disrupted by alterations in the environment, such as rapid phase shifting of the light/dark cycle to mimic chronic jet-lag or shift work brings about similar changes in glucose metabolism and weight control.
Finally there is a growing body of epidemiological evidence that shift work and the associated interference with sleep/wake cycles, light exposure and meal times increases the risk of developing chronic diseases, including cardiovascular disease, diabetes and possibly some cancers.

What are the main hypotheses proposed for why night-time activity and daytime sleep increases the risk of developing or exacerbating chronic diseases?
A current popular hypothesis is that the timing of meals, in particular the intake of high calorie food during the evening by shift workers plays an important role in altering our physiology and increasing the risk of chronic disease. This is because the enzymes involved in utilising the nutrients are rhythmically translated and finely tuned to our daily lifestyle.
When we eat at night, high levels of glucose, for example following a high energy snack, are not utilised or controlled efficiently. A shift worker who has persistently elevated night time glucose over many years and blood glucose is likely to have an increased risk of developing diabetes. This can be especially problematic if the person has other risk factors, such as being overweight, a smoker and a genetic risk.
Please can you outline your group’s finding with regards to circadian rhythm disruption during pregnancy?
My Institute, the Robinson Research Institute, is a leader in research into the impact of early life events on adult health. This is best illustrated by the impact of poor nutrition during pregnancy or impaired placental function on chronic disease risk in adult life.
Following our findings on the effects of genetic disruption of rhythms on glucose metabolism, we asked the question whether disruption of rhythms during pregnancy through simulated shift work might have an impact on the health of the offspring later in their life.
When we conducted the experiment on laboratory rats we did indeed find that when the offspring had reached maturity they had clear markers of an increased risk of diabetes. We are trying to obtain funding to extend this research in another animal model and to initiate retrospective studies in pregnant shift working women.

Do you think melatonin could one day be used to help control the circadian rhythm?
Melatonin is already being used by people without prescription, particularly in the USA, who are having difficulties sleeping because it is promoted as a “dietary supplement”. Its use has been described as the greatest uncontrolled clinical trial in history!
As a consequence there is a wealth of mis-information out on the Internet. For example some think it is a herbal substance, or an extract produced from cherries and not a drug!
When it is provided as a 3 milligram tablet for swallowing at night, melatonin is of course being used as a drug not as a supplement.
We produce approximately only 40 micrograms of melatonin across the whole night and the variability in circulating blood levels between individuals is very large.
The correlation between the amount of melatonin we have circulating and the presence of sleep problems is poor. What is more important is the time of day that the melatonin production commences; when the production of melatonin starts very late in the evening, sleep onset may also be late.
But not everyone who has difficulty getting to sleep has a delay in melatonin onset, indeed even in clinically diagnosed delayed sleep as many as 40% of the patients will have a normal timing of their melatonin rhythm.
In people with both delayed melatonin onset and delayed sleep, melatonin administration may help bring forward sleep by up to an hour. Unfortunately self medicators do not know what their own melatonin rhythm is doing. Another issue is that melatonin administration has been shown in numerous studies to have little impact on night-time awakening or sleep duration.
What further research do you think is needed to increase our understanding of melatonin and the circadian rhythm?
The word that is so often heard and written about melatonin is that is safe! In countries outside the USA where it is available on prescription there is a prolonged release formulation that has achieved registration for use for treatment of primary insomnia for people aged 55 years and over.
All other prescription of this and other formulations is “off-label” and there have been no properly conducted long term safety studies conducted in children, adolescents or young adults. Experimental studies in non-human mammals that have been conducted across the world to gain an understanding of the functions of melatonin have highlighted major roles in puberty, seasonality of fertility, metabolism, blood pressure control and breast function.
While desirable, it is most unlikely that long term human trials will ever be conducted in children or adolescents and so parents and others giving melatonin to their children and conducting their own experiments.
Should adverse outcomes emerge after prolong administration of melatonin it will not tenable to say that we were surprised! The warnings are out there that melatonin acts on many systems apart from potentially advancing the timing of sleep by a few minutes.
Where can readers find more information?
Ferracioli-Oda E, Qawasmi A and Bloch MH (2013) Meta-analysis: Melatonin for the treatment of primary sleep disorders. PLoS ONE 8:e63773.
My paper on safety of melatonin in Paediatrics is not open access unfortunately.
About Professor Kennaway
Professor David Kennaway is a Senior Research Fellow at the Robinson Research Institute, University of Adelaide leading the Circadian Physiology Laboratory and is the director of the Adelaide Research Assay Facility. His research interests have always centred around the impact of the environment, especially day length and light exposure, on physiological systems including reproductive processes, brain function and metabolism. In the course of his career he developed one of the first reliable blood melatonin radioimmunoassays which allowed him to determine the effects of season on patterns of melatonin production in sheep, to study the impact of altering these patterns on reproduction and then to develop a novel melatonin formulation for the agricultural market to control seasonal reproduction. He was the first to demonstrate that premature birth and light in the intensive care nursery alters the development of circadian rhythms in babies, which has led to changes in lighting practices in nurseries, facilitating timely development of sleep rhythms in infants. He showed that cells in the female reproductive tract are rhythmic and could play a major role in embryo development, that there is early life programming of central rhythmicity (melatonin production) by birth size in humans, thus providing the first evidence that circadian rhythmicity may be programmed in utero, and that disrupted circadian rhythmicity during pregnancy as can occur in shiftworkers, programs the offspring for subsequent metabolic disorders in adult life.
His current research interests centre on the physiological consequences of disrupted circadian rhythmicity in shift workers who are at high risk of developing chronic metabolic and cardiovascular diseases. He uses genetic animal models of rhythm disruption (Clock mutant and Bmal1 null mice) as well as environmental manipulation of animals and shift work simulation in human volunteers to develop an understanding of the mechanisms underlying these pathologies. In addition, he works closely with sleep researchers in Adelaide, Melbourne and Sydney on the neuro-behavioural impact of sleep loss and shift work in field and laboratory settings. He has authored more than 190 original research publications, reviews and patents. He is a member of the Editorial Boards of the Journal of Biological Rhythms, Chronobiology International, Molecular and Cellular Endocrinology and the Journal of Reproductive Health and Medicine and was the foundation Chair of the Australasian Chronobiology Society.