Embryos require adequate amounts of energy so that they can successfully progress through each of their cell divisions.
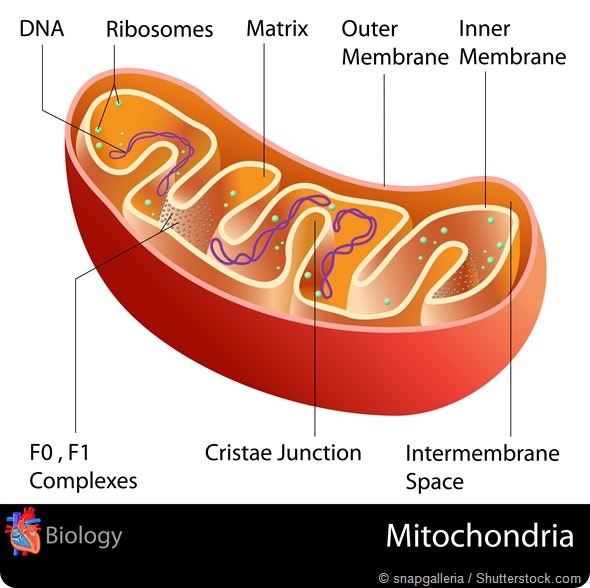
Mitochondria have a key function during early development, as they are the main energy providers of embryos. They are also involved in the regulation of other key cellular processes, such as apoptosis, amino acid and protein synthesis.
Up until now, why has little been known about the biological and clinical implications of mitochondrial variation between embryos prior to implantation?
Until recently the technical tools necessary for such studies were not available. The development of methods such as next generation sequencing (NGS) have made the detailed analysis (quantity and sequence) of the mitochondrial genome (mtDNA) of human embryos possible.
Please can you outline your recent research into the quantity of mitochondrial DNA (mtDNA) in embryos?
Correct mitochondrial function and mtDNA gene expression are critical during the first few days of life. We were therefore interested in carrying out a thorough investigation of mtDNA in human preimplantation embryos.
Specifically, we examined the presence of possible relationships between mtDNA content, female patient age, embryo chromosome status, viability and implantation potential.
Additionally, we attempted to shed light on the stage during which the embryo first initiates its own mtDNA replication.
We also carried out a detailed analysis of the mitochondrial genome searching for mutations, deletions and polymorphisms.
This study was carried out on samples removed from 430 embryos and a total of 240 couples participated. These couples were having IVF treatment in combination with preimplantation genetic screening (PGS)). They were referred to Reprogenetics by 12 different IVF clinics in Europe and the USA.
What were your main findings?
Our main findings were the following:
- We observed statistically significant correlations between advancing female age and mtDNA quantity. Embryos at the final stage of development before implantation occurs (blastocyst stage) and generated by women of advanced reproductive age (38 years or older) consisted of more mtDNA compared to blastocysts generated by younger women (37 years or younger).
- We observed statistically significant correlations between embryo ploidy and mtDNA quantity. Chromosomally abnormal embryos consisted of more mtDNA compared to chromosomally normal embryos. The relationship between mtDNA quantity and the incidence of chromosomal abnormalities was independent of female age. In other words, in any given female age chromosomally abnormal blastocysts consisted on average of higher levels of mtDNA compared to chromosomally normal ones.
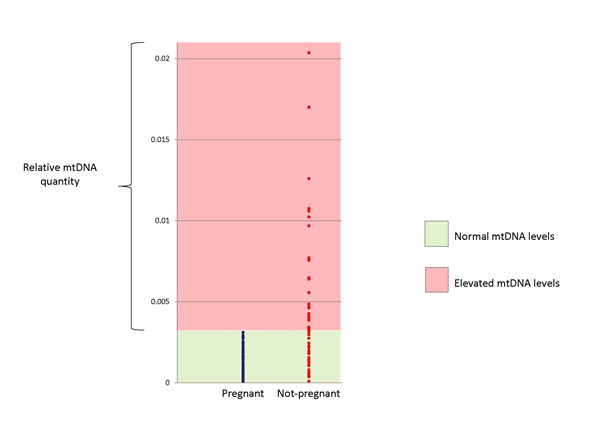
- We observed statistically significant correlations between embryo viability and implantation potential and mtDNA quantity. Chromosomally normal embryos capable of establishing an ongoing pregnancy after IVF treatment consisted of lower mtDNA amounts compared to embryos, which did not implant after transfer to the female patient’s uterus.
- We identified a threshold mtDNA amount above which a chromosomally normal embryo was incapable of leading to an ongoing pregnancy after transfer. This threshold amount was independent of embryo morphology or the IVF clinic where the couples were being treated.
Were you surprised to find that mtDNA levels were higher in embryos from older women? How do you think this finding can be explained?
I was not very surprised, as the success rate of IVF dramatically decreases with increasing female age. The increase in mtDNA with advancing age raises the question of whether mitochondria and their genome might play a direct role in the decline of female fertility with age.
It may be that high mtDNA levels are indicative of compromised mitochondria unable to generate the expected amount of energy to support embryo development. However, assessment of the mitochondrial genome via NGS did not identify any obvious mutations.
It is possible that the underlying defects are unrelated to alterations in the DNA sequence. Alternatively, elevated quantities of mtDNA might be associated with increased metabolic requirements of the embryo, rather than organelles of suboptimal function. Perhaps embryos produced by older oocytes are under some form of stress and therefore have larger energy requirements. Functional experiments are necessary in order to address these questions.
Please can you outline the mtDNA quantity threshold that you found? How was this confirmed?
The threshold number is generated by an algorithm which would not be meaningful to the reader. The key point to note is that a threshold exists above a chromosomally normal embryo is not capable of leading to an ongoing pregnancy after transfer.
We first identified this threshold via a retrospective data analysis. In order to confirm its predictive value, we carried out a blinded prospective study.
During this study, embryos were selected for transfer based on the result of comprehensive chromosome screening (CCS) (they were normal) and after morphological assessment.
We subsequently analyzed their mtDNA and observed that all the embryos with mtDNA quantities above the previously established threshold did not lead to ongoing pregnancies after transfer.
What is the ‘quiet embryo’ hypothesis and are your results consistent with it?
The ‘quiet embryo hypothesis’ was initially proposed by Professor Henry Leese, and suggests that viable embryos have relatively lower or ‘quiet’ metabolism, whereas those under stress, and of reduced developmental potential, tend to be more metabolically active.
Our results may be consistent with this hypothesis, as the abnormally high levels of mtDNA seen in non-viable embryos may be symptomatic of some form of stress that results in elevated energy requirements.
What value could mtDNA have for in vitro fertilization (IVF) treatment?
This study identified for the first time a clear association between mtDNA quantity and the ability of a human embryo to implant in the uterus. The discovery of a new biomarker of embryo viability, independent of standard assessments such as morphology, is a rare event and of great clinical potential.
In the current study an mtDNA threshold was established, above which implantation failure was 100%. The data obtained suggests that embryo deficiencies associated with elevated mtDNA explain up to one-third of implantation failures affecting blastocysts diagnosed as chromosomally normal.
The defined mtDNA threshold does not appear to be altered by variation in the processes used by different IVF clinics, indicating that evaluation of mtDNA in embryos could form the basis of a simple, inexpensive and widely applicable clinical test.
Clinical use of this new biomarker in combination with PGS could increase the IVF success rate by an additional 10% to that obtained by PGS alone.
How do Reprogenetics plan to build upon these findings moving forwards?
Reprogenetics is currently carrying out a large (100 IVF cycles) blinded non-selection study in collaboration with the IVF clinic at the NYU Langone Medical Center to further assess the rate of outcome improvement if mtDNA quantification is combined with PGS.
Reprogenetics has also started offering this type of mtDNA analysis in IVF/PGS cycles where multiple chromosomally normal embryos are available, so as to provide priority of transfer.
Where can readers find more information?
Readers can find more information in our recently published research article (Fragouli E, et al. (2015) Altered Levels of Mitochondrial DNA Are Associated with Female Age, Aneuploidy, and Provide an Independent Measure of Embryonic Implantation Potential. PLoS Genet 11(6): e1005241. doi:10.1371/journal.pgen.1005241) and the Reprogenetics website (www.reprogenetics.com)
About Dr Elpida Fragouli
Elpida Fragouli obtained a bachelor degree in Molecular Biology from the University of Surrey and a Masters qualification in Molecular Biology, Biochemistry and Genetic Manipulation from the University of Sussex. She went on to obtain a second Masters degree in Preimplantation Genetics and Fetal Medicine from University College London, followed by a PhD in human genetics.
After completing her studies she performed clinical diagnostics at the UCL Centre for PGD and carried out research into the cytogenetics of human oocytes. She later moved to the USA, taking a research position at Yale University Medical School. In 2007 she returned to the UK, and now works as the Lab Director at Reprogenetics UK as well as holding a research position at the University of Oxford.
Elpida played a key role in the development, validation, and clinical application of comparative genomic hybridization (CGH), the first comprehensive chromosome analysis method to be widely applied to the study of human embryos. The proliferation of chromosome screening techniques seen during the last few years owes much to the Elpida's early work in this area.
Of particular note are her studies that highlighted the blastocyst as the optimal stage for aneuploidy screening to be undertaken. Her work has led to the most detailed characterization of chromosome abnormalities in human oocytes and embryos published to date.
Elpida's current research interests focus on understanding the mechanisms leading to aneuploidy, and developing methods for the non-invasive and invasive assessment of gametes and embryos. She has published more than 30 peer-reviewed papers and book chapters and has been the recipient of multiple awards for her work, including the New England Fertility Society-PCRS Exchange Prize for best submitted abstract (2007) awarded for her work on CGH, the ESHRE Basic Science Prize (2011) for her study of the transcriptome of cumulus cells, and the ASRM SART (2015) Prize for the assessment of the mitochondrial genome of human embryos. Elpida is a familiar figure at international scientific and medical conferences, her cutting-edge work frequently leading to invitations to speak.
Elpida feels strongly about the need to improve the accuracy and efficacy of preimplantation genetic diagnosis (PGD). She also has a strong commitment to teaching and the sharing of scientific knowledge. Towards this end she spent several years on the Editorial Board of various scientific journals such as Human Reproduction, Molecular Human Reproduction, Reproductive Biomedicine Online, and the Journal of Assisted Reproduction and Genetics, and teaches and supervises postgraduate students at the University of Oxford.