Dec 21 2015
A group of Russian scientists, including Dr Elena Kostryukova, the Head of the Laboratory of Postgenomic Research in Biology of the Scientific Research Institute of Physical-Chemical Medicine and a researcher at MIPT (Moscow Institute of Physics and Technology), and Maria Vakhitova, an MIPT postgraduate student, has discovered that the presence of certain bacteria in the gut may be linked to the development of type 2 diabetes.
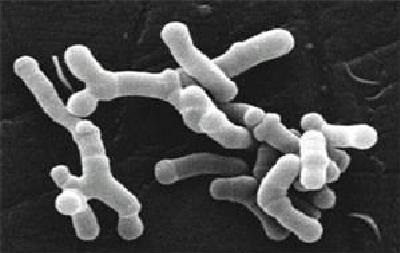
Bifidobacteria as seen under an electron microscope. Approx. 5 micrometres in length. Image: Julie6301 / Wikimedia
Type 2 diabetes, also known as non-insulin-dependent diabetes, is a serious metabolic disorder. The disease causes the body’s cells to lose their ability to respond to insulin – the hormone that controls glucose uptake. As the cells are unable to interact with insulin, they stop absorbing glucose and are therefore starved of energy, even with adequate nutrition, a high level of glucose in the blood, and a sufficient amount of insulin (in type 1 diabetes the pancreas does not produce any insulin, which causes glucose deficiency in the body’s cells).
According to the World Health Organization, there are approximately 285 million people with type 2 diabetes worldwide. The prevalence of the disease and the severity of its complications (people with diabetes are 20 times more at risk of developing gangrene of the lower limbs, for example) mean that doctors and scientists are not only looking for causes of the disorder, but also new methods of combating the disease. Recently, researchers have been developing serious arguments to support the theory that diabetes may be linked, among other things, to the composition of the microbial community living inside our intestines, the intestinal microbiota. Specialists from four Russian research centres studied changes in the microbiota of the large intestine, and their paper was recently published in the journal Endocrinology Connections.
The scientists analysed the gut microbiota composition in 92 patients, including 20 patients with type 2 diabetes and 48 healthy people without any chronic diseases; a further 24 people showed signs of a metabolic disorder which doctors had diagnosed as prediabetes – a condition that can lead to the development of type 2 diabetes over time. The glucose level, which is the most important metabolic indicator, was also monitored in the study participants by taking blood samples.
Gut microbiota is the name given to the entire population of microorganisms living in the intestines (even including certain single-celled fungi and archaea). They are also commonly called microflora – this is a historical term that emerged at a time when bacteria were considered a part of the plant kingdom.
Bacteria were the “focal point” of the study. In order to determine exactly which bacteria they were dealing with, the researchers analysed DNA extracted from stool samples. Having isolated the DNA, the scientists sequenced (counted and deciphered the sequence of base pairs in the DNA) the gene that encodes ribosomal RNA, rRNA – an important component of ribosomes, which are microscopic structures responsible for protein synthesis. The gene that encodes this structure is found in all classes of bacteria and there is a database that can be used to identify bacteria based on the nucleotide sequence in the gene. This part of bacterial DNA could be described as a bacterial passport.
After comparing the composition of the microbiome with the diagnosis (diabetes/prediabetes/normal glucose tolerance) and the diet of the study participants, the scientists came to a number of conclusions.
Most importantly, the researchers were able to link the level of glucose intolerance with the presence of three specific types of microbiota: Blautia, Serratia, and Akkermansia bacteria. They are all found in healthy people, but in cases of prediabetes and diabetes they are present in a far greater quantity.
The researchers were able to link increased levels of bifidobacteria (they are most commonly found in the intestines of breast-fed infants and they are considered to be one of the most important components of microbiota) to a high intake of dietary fibre, or roughage. This corresponds with the results of a previous study of gut microbiota in urban and rural populations in Russia which the same group of researchers were also involved in. During this study it was found that in people living in rural areas of the Tyva Region, levels of bifidobacteria were significantly higher than in residents of other regions: this was presumably due to the fact that the diet of the rural population was based more on natural products and the proportion of “industrial” food with a low fibre content, in contrast, was relatively small.
These findings bring scientists and medical professionals one step closer to understanding the complex reciprocal cause-and-effect relationship between the change in the proportion of certain types of bacteria, metabolic disorders, and diet. As the authors of the paper point out in discussing their findings, one of the possible ways that microbes affect diabetes could be by provoking an immune response – this theory was previously put forward by researchers from Finland.