Scientists have developed a modified CRISPR tool that can stimulate the de-differentiation of somatic cells into induced pluripotent stem cells, eliminating the need for the artificial insertion of genes, which has previously been shown to cause cancer.
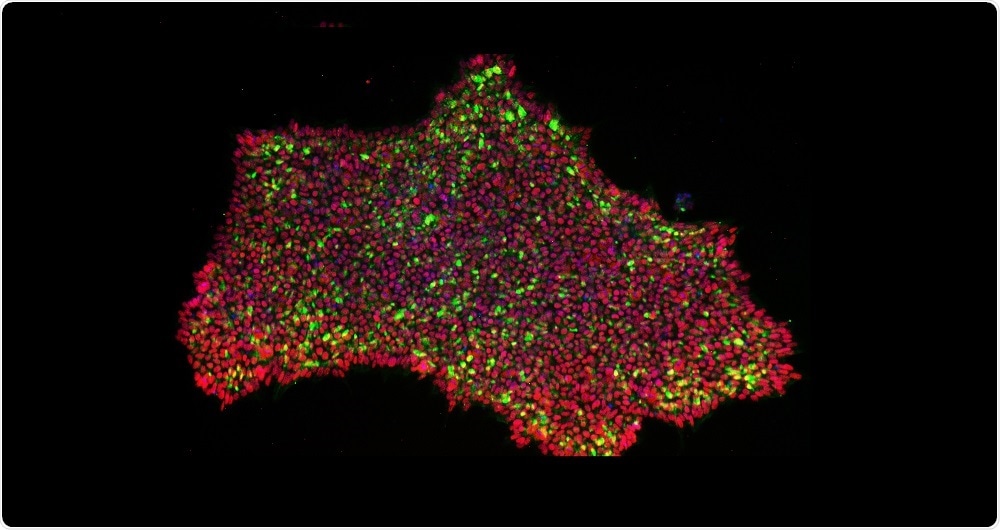
CRISPRa reprogrammed induced pluripotent stem cell colonies stained for pluripotency marker expression. (Image Credit: Otonkoski Lab / University of Helsinki)
In 2012, Shinya Yamanaka, a Japanese scientist, made the Nobel-Prize winning discovery that it is possible to take adult skin cells through a process of reprogramming in order to convert them into induced pluripotent stem cells (iPSCs), which can develop into any cell type of the human body.
Some genes are essential for such conversion, called the Yamanaka factors. Previously, these factors had to be inserted into adult skin cells using artificial methods, creating the potential for genetic mutations and the development of cancer.
However, a recent study has reported that it is possible to activate the endogenous genes of the adult skin cells (in order to produce iPSCs) through the use of a new gene editing technology called CRISPRa.
The tool makes use of an engineered version of the Cas9 gene that usually functions to cut DNA. In CRISPRa, this genetic ‘scissor’ is blunted, making it unable to excise the section of DNA. This allows it to activate the target gene without producing gene mutations.
Professor Otonkoski, one of the lead researchers in this study, explained that the use of the CRISPR/Cas9 system to activate genes opens up great possibilities in the area of cell reprogramming by targeting multiple genes simultaneously.
Activating genes already within the cell rather than inducing overexpression of transgenes mimics human physiology more closely, and is therefore a safer way to induce cellular differentiation. This results in cells that appear and function more normally, compared to iPSCs produced using previous methods.
Another crucial discovery was that the tool could successfully activate a gene known to control the very early development of the post-fertilization embryo.
The new study also indicates the possibility of improving multiple reprogramming functions by focusing upon only those genes or related elements that are characteristic of the cell type that is targeted by the experiment.
It is also likely to help researchers better understand how genes are activated during early embryonic life. The lead researcher, PhD student Jere Weltner affirms, “The technology may find practical use in bio banking and many other tissue technology applications."
The study was recently published in the journal Nature Communications.