Research has been ongoing in the field of genetic mutations and changes and brain and other neural disorders including Autism Spectrum Disorders (ASD) and a new study in mice models of autism has shown a possible link between mutations or deletions of the SHANK3 gene and a rare disorder called Phelan-McDermid syndrome and ASD.
The study from researchers at the MIT in collaboration with those in China, have found that mice with the SHANK3 mutations show traits of autism including avoidance of social interactions. The study titled, “Anterior cingulate cortex dysfunction underlies social deficits in Shank3 mutant mice” was published in the latest issue of the journal Nature Neuroscience.
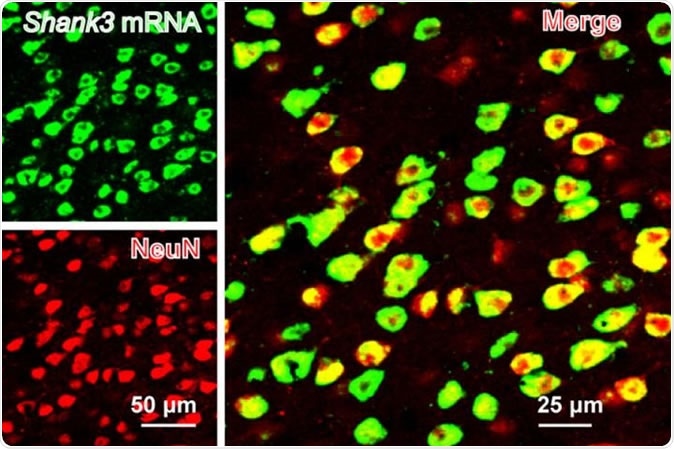
SHANK3 (green) is expressed along with a neural marker (NeuN) in the mouse anterior cingulate cortex. Image Credit: Guoping Feng
The team of researchers have successfully produced mutations at the SHANK3 gene in the mice and studied their autism like traits. They noted that this mutation also led to structural and functional defects and alterations in the anterior cingulate cortex (ACC) region of the brain in the mice. Guoping Feng, the James W. and Patricia T. Poitras Professor at MIT, investigator at MIT’s McGovern Institute, senior scientist in the Broad Institute’s Stanley Center for Psychiatric Research and lead author of the study explained, “Neurobiological mechanisms of social deficits are very complex and involve many brain regions, even in a mouse model. These findings add another piece of the puzzle to mapping the neural circuits responsible for this social deficit in ASD models.” Feng and his team collaborated with Wenting Wang and Shengxi Wu at the Fourth Military Medical University, Xi'an, China for this study.
The team created SHANK3 mutant mice in their lab and attempted to study the brain regions associated with autism. They write that more than one region of the brain has been previously associated with autism and especially altered social interactions. This included the prefrontal cortex (PFC) and its projections within the brain including the nucleus accumbens and habenula. These studies however have found no direct link between avoidance of social interactions in SHANK3 mutant mice and changes in the PFC. For this study, thus, the team focussed on the ACC region of the brain of the mutant mice. In a normal human being and animals the ACC region is known to play a role in social interactions as well as in cognitive functions, decision making, cost-benefit analysis and motivation.
Among the mice that were mutant with SHANK3 deletions, the researchers noted that there was a structural as well as functional alteration at the synapses between the neurons especially at the ACC region. The team noted that this disruption at the excitatory neurons of the ACC was responsible for disruption of the communication of the neurons especially when social interactions and related behaviours took place. There was a marked reduction in social interactions they noted, among those with SHANK3 deletions. They wrote that they noted, “...structural and functional impairments occur in glutamatergic synapses in the pyramidal neurons of the anterior cingulate cortex (ACC) in mice with a mutation in Shank3, a high-confidence candidate ASD gene.”
As a next step, the team activated the ACC neurons and checked if this could restore the social interactive behaviours in the mice. To this end they used optogenetics techniques as well as specific targeted drugs (including “α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor-positive modulator”) to see if they could rekindle the excitatory neurons in the ACC region and correct the social interactive behaviour and avoidance. The results seemed promising, they write.
The authors wrote in conclusion that their, “findings provide direct evidence for the notion that the ACC has a role in the regulation of social behavior in mice and indicate that ACC dysfunction may be involved in social impairments in ASD.”
Wenting Wang explained, “Next, we are planning to explore brain regions downstream of the ACC that modulate social behavior in normal mice and models of autism. This will help us to better understand the neural mechanisms of social behavior, as well as social deficits in neurodevelopmental disorders.”
This study was funded by Natural Science Foundation of China and the National Institute of Mental Health (NIMH), the Poitras Center for Psychiatric Disorders Research and Hock E. Tan and K. Lisa Yang Center for Autism Research at the McGovern Institute at MIT.
Related study
A recent study from Marco Pagani and colleagues from the Centre for Neurosciences and Cognitive Systems, Italy showed that deletion of the SHANK3 gene was associated with disrupted prefrontal connectivity. The study titled, “Deletion of Autism Risk Gene Shank3 Disrupts Prefrontal Connectivity,” was published on 3rd July 2019 in the Journal of Neuroscience.
The authors concluded that “SHANK3 deficiency affects higher-order socio-communicative functions,” but the actual mechanism is unclear. The team used high-resolution functional and structural MRI in adult male mice with SHANK3 deletion to show that there was a reduced connectivity in the prefrontal regions of the brain. They noted that this deletion caused, “reduced short-range cortical projections density, and reduced gray matter volume.” They concluded, “SHANK3 deficiency may predispose to intellectual disability and socio-communicative impairments via dysregulation of higher-order cortical connectivity.”
Source:
Journal reference:
Anterior cingulate cortex dysfunction underlies social deficits in Shank3 mutant mice, Baolin Guo, Jing Chen, Qian Chen, Keke Ren, Dayun Feng, Honghui Mao, Han Yao, Jing Yang, Haiying Liu, Yingying Liu, Fan Jia, Chuchu Qi, Taylor Lynn-Jones, Hailan Hu, Zhanyan Fu, Guoping Feng, Wenting Wang & Shengxi Wu, Nature Neuroscience volume 22, pages 1223–1234 (2019), https://www.nature.com/articles/s41593-019-0445-9