Scientists at the University of California San Diego School of Medicine have shown that removal of an enzyme called the PH domain Leucine-rich repeat Protein Phosphatase 1 (PHLPP1) could help laboratory mice with sepsis in a model they had created for their experiment. The study titled, “PHLPP1 counter-regulates STAT1-mediated inflammatory signalling,” was published this week in the journal eLife.
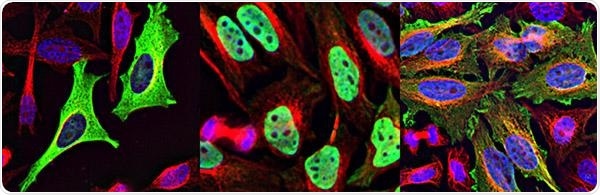
HeLa cells expressing PHLPP (left), the segment with the nuclear localization signal (middle), and that same segment mutated (right). Image Credit: UC San Diego School of Medicine
Sepsis is a condition where an infection takes over the whole body and the immune system fails to fight it off and as a result of the overdrive of the immune cells in an attempt to fight off the infection, the major organs may be damaged. There is usually a resultant organ failure and death in patients with sepsis. According to the Centers for Disease Control and Prevention, CDC, sepsis is diagnosed in 1.7 million adults and it kills 270,000 in the United States annually. One third of all the hospital deaths are attributed to sepsis says the CDC. Treatment at present involves treating and eliminating the source of infection with antibiotics and providing supportive care using oxygen and intravenous fluids.
The team of researchers found that enzyme PHLPP1 helps remove phosphates from other proteins. This controls cell behaviours and also alters the inflammation pathways. They found that in the mice models of sepsis if they blocked the said enzyme, there was a significant improvement in the condition.
The researchers hope that what they have demonstrated in the mice could one day be translated to humans and could mean a new therapy for sepsis in humans as well. Senior author of the study Alexandra Newton, PhD, professor in the Department of Pharmacology at UC San Diego School of Medicine said in a statement, “Most research on inflammation has typically focused on kinases, enzymes that add phosphate tags to other proteins. It's exciting to have a completely new target for sepsis -- the enzymes that remove them.”
This team has been studying PHLPP1 for the last few years and has found its many uses within the body. For example, this enzyme also plays a role as a tumour suppressor. In order to study the role of the enzyme in inflammation Newton and her team collaborated with Chris Glass, an inflammation expert from UC San Diego School of Medicine. Their collaboration yielded findings that showed several genes in the immune cells are responsive to the manipulations of PHLPP1. There is a transcription factor called STAT1 on the inflammatory cells, write the researchers. One of the most important roles played by PHLPP1 enzyme was removal of the phosphate tag from the STAT1 and thus reducing the risk of inflammation, wrote the researchers.
Newton and her colleagues developed mice which were genetically engineered to miss their PHLPP1 enzyme. In these mice the gene coding for the enzyme was removed. These mice were then taken to the lab of Victor Nizet, an expert on bacterial infections at UC San Diego School of Medicine. Nizet’s team next injected the mice with live E. coli bacteria and lipopolysaccharide (LPS). LPS is a component or part of the cell wall of bacteria. It acts as a trigger for the immune cells which respond to it my multiplying at a rapid rate. The bacteria and the LPS was administered simultaneously to both normal control mice and PHLPP1 missing mice in the experiment.
Results revealed that mice that did not have PHLPP1 survived the sepsis due to the bacterial infection while the control mice all died of the sepsis induced by the infection within five days of the infection.
Logically the next step was to identify molecules that could suppress the action of PHLPP1 in the mice. This would help the mice with the sepsis explained Newton. These inhibitors of PHLPP1 would be developed as the anti-sepsis drugs as well they wrote. These molecules when identified with certainty would be tested on mice models of sepsis before they are proven safe and effective for human clinical trials, they wrote.
Authors wrote in conclusion, “...data indicate that PHLPP1 inhibitors could be explored as adjunctive therapies to antibiotics and supportive care of patients with Gram-negative sepsis, a leading cause of mortality in intensive care units.”
Nizet said, “Sepsis is the leading cause of death in intensive care units throughout the world, but unfortunately there is not a single approved drug treatment for sepsis. Discoveries like ours of fundamental signaling pathways that control immune cell behavior during sepsis offer clues for controlling the dangerous inflammation of sepsis while preserving the critical bacterial killing properties of white blood cells.”
Journal reference:
PHLPP1 counter-regulates STAT1-mediated inflammatory signaling,Cohen Katsenelson, Ksenya and Stender, Joshua D and Kawashima, Agnieszka T and Lordén, Gema and Uchiyama, Satoshi and Nizet, Victor and Glass, Christopher K and Newton, Alexandra C, eLife 2019;8:e48609, https://doi.org/10.7554/eLife.48609, https://elifesciences.org/articles/48609