The study involved nearly 20 years of extensive epidemiological research. The researchers recruited children in Tanzania at birth, beginning in 2001, and followed them up for years to identify children who developed an acquired immune response. Michal Fried and Patrick Duffy of the National Institutes of Health led this phase of the study.
The researchers initially focused on identifying resistance levels of the children, on determining which children were resistant and which susceptible to the parasite. They needed this information to identify antibody molecules that were unique to the resistant children but not found in the others.
Once this had been determined, the team chose 12 resistant and 14 susceptible children from the Tanzanian cohort. Blood samples were taken from the children at age two since immunity seems to develop around this time following exposure to natural infection.
The scientists used a specialized technique to add proteins from the malarial parasite singly to the blood samples, step by step. This enabled them to detect antibodies to a particular protein in the blood samples from a resistant child, which was not present in the susceptible population. This led to the identification of PfGARP as present in resistant children and thus potentially contributing to malaria resistance.
PfGARP is a protein produced by malarial trophozoites, the form of the parasite that lives and multiplies inside the patient's red cells. The protein is then carried to the outer cell membrane of the parasite. This is where the antibody can bind to it.
The researchers went on to examine the protective efficacy of the antibody. They looked at the association of antibodies against this malarial protein with protection against malaria, in a sample of 246 children. They found that the absence of this antibody boosted the chances of severe malaria 2.5 times compared to those who had the antibody.
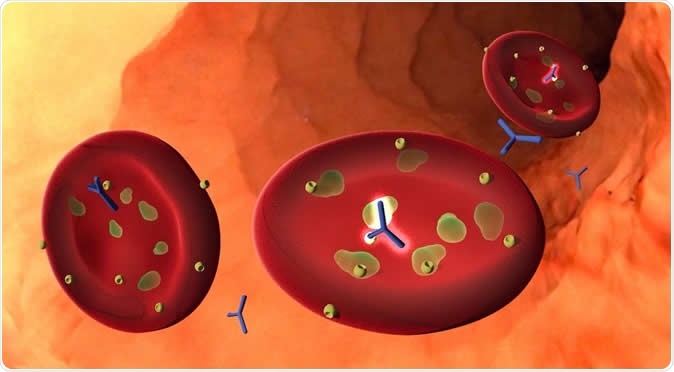
A new study identifies an antibody, often present in children with natural immunity to severe malaria, that attacks a particular malaria protein called pGARP. When the antibody binds the protein, it causes malaria cells to undergo a form of programmed cell death. The discovery could be the basis for a new malaria vaccine. Image Credit: Kurtis Lab / Big Ocean
How do anti-PfGARP antibodies work?
Researchers worked out that the antibody to the protein PfGARP appeared to activate a "kill switch" that caused parasite cells inside human red blood cells to self-destruct, thus decreasing parasite load in the body. Says researcher Jonathan Kurtis, "When we introduce the antibody to samples in Petri dishes, we end up with 98% or 99% dead parasites."
It seems rather unusual for a parasitic organism to evolve such a "kill switch" mechanism. Kurtis hypothesizes that the mechanism might be a response to the parasite sensing distress in its host. When the host dies, after all, so does the parasite. Therefore, if the host is very sick, it is in the parasite's interest to reduce its load, enabling the host organism to regain health.
Malaria Vaccine Antigen Discovery
Malaria Vaccine Antigen Discovery from BIG OCEAN on Vimeo.
The response to the anti-PfGARP antibody might be the host's way of 'hijacking' one such mechanism. The study has implications for vaccine research, with researchers going on to develop two different vaccine types. Both were tested in nonhuman primates and showed significant protection of these animals against human malaria.
The scientists say they are confident that vaccination of individuals with PfGARP to generate anti-PfGARP antibodies, or directly injecting PfGARP antibodies, may help guard against severe malaria. Their optimism about the new strategy's chances of success stems from its ability to attack the parasite at a different point in the infection cycle, buying more time for the vaccine to act.
"What's exciting is that this is a vaccination strategy that attacks malaria in a way that it has never been attacked before -- one in which the parasite becomes complicit in its own demise. We are hopeful that this vaccine, perhaps combined with other malarial antigens, will translate into a strategy that can help prevent severe malaria in people."
The malarial parasite's life cycle
Malaria research has mapped out the life cycle of the mosquito-borne malarial parasites called Plasmodium. When an infected mosquito bites a human, thread-like plasmodial cells called sporozoites travel through the blood to the liver, where they develop into merozoites that burst out of the liver to invade red blood cells. Once inside the red cell, the merozoites morph into trophozoites, which frantically feed on and destroy the cell before exploding out of it to infect more cells.
Previous vaccines have targeted the first stage - the sporozoites, with limited success. Kurtis attributes this to the tiny time window to stop the parasite - it takes only five minutes for the parasite to reach the liver, and even a single sporozoite can multiply rapidly to infect the individual. Stopping the sporozoites would require a considerable antibody dose.
This is where the new antibody has an advantage - it targets the trophozoite stage, which lasts up to a day, buying time for the antibody to target cells and kill the trophozoites before they infect other red blood cells.
"This gives us 24 hours as opposed to 5 minutes to intervene," Kurtis said. "During that time, the parasite expresses PfGARP -- a kill switch. We have designed a vaccine that activates it."
Could this be 'a vaccine that can save lives'?
Researchers say that a human vaccine is still years away since many different versions must be tested, first in animal models and then in humans. Even then, there is no guarantee that it will work precisely as expected. Nonetheless, the new approach that utilizes natural mechanisms of resistance has opened up possibilities about the unknown vulnerabilities of the parasite, and different ways to attack them.
Kurtis says, "This was an incredible team effort involving infectious disease experts, pathologists, epidemiologists, geneticists, and molecular biologists. It really took all of these people to make this possible, and we're hopeful that the end result will be a vaccine that can save lives."
Journal reference:
Raj, D.K., Das Mohapatra, A., Jnawali, A. et al. Anti-PfGARP activates programmed cell death of parasites and reduces severe malaria. Nature (2020). https://doi.org/10.1038/s41586-020-2220-1