The COVID-19 pandemic began in the last month of 2019 and spread rapidly throughout the world, causing over one million deaths so far, besides many tens of millions of confirmed infections. It is particularly dangerous because of its rapid and extensive infectivity combined with high mortality. Now, a new study published on the preprint server medRxiv* in September 2020 shows that sebum lipids change in COVID-19. This could help develop a cheap and straightforward method of non-invasive diagnosis.
The virus, SARS-CoV-2, enters the human body via the angiotensin-converting enzyme 2 (ACE2) receptor on the target host cells. Most symptoms relate to the respiratory system. A significant minority of cases will develop severe and hyperinflammatory features involving the lungs, leading to acute distress syndrome (ARDS), often associated with a cytokine storm.
Thus, the clinical manifestations of COVID-19 reflect both the direct harm caused by the virus infection and the immune response of the host. Mass testing is necessary to contain the virus and reduce the burden on the health services, usually involving the collection of upper respiratory samples and testing for viral RNA by polymerase chain reaction (PCR).
However, this approach has a sizable false-negative rate due to many factors and fails to offer prognostic value. Thus, research is on to assess the impact of the virus on the host rather than the virus itself, as an additional testing modality.
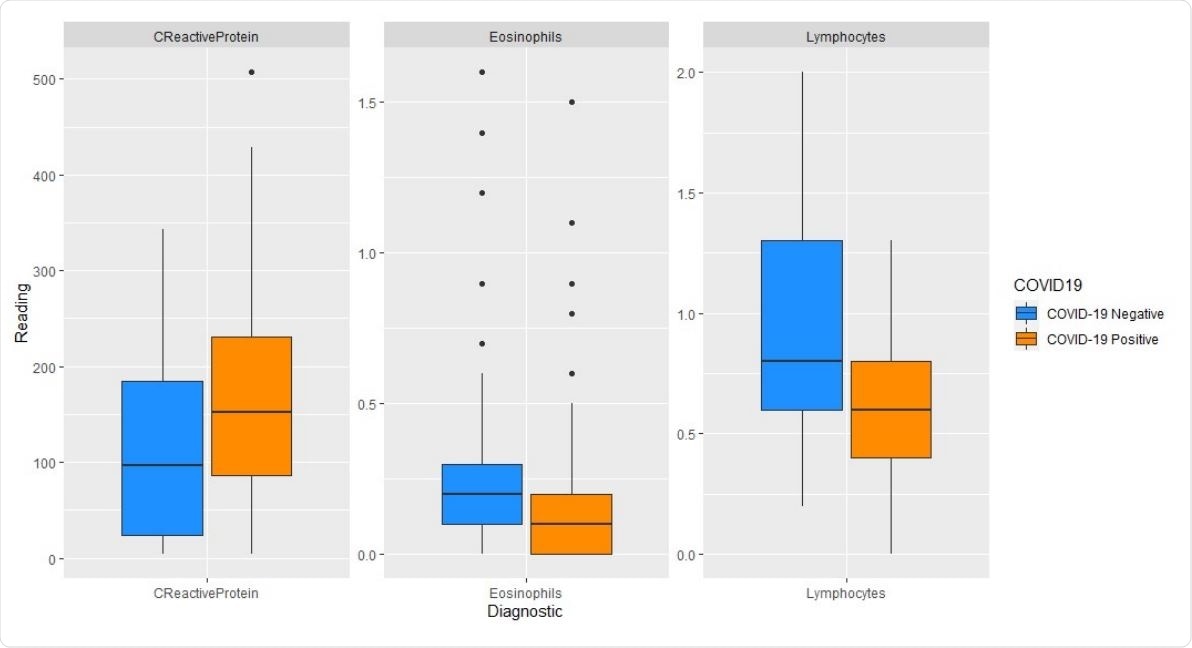
Boxplots of diagnostic readings from plasma: COVID-19 negative versus positive

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
COVID-19 and the Sebum Lipidome
The current study focuses on the effect of COVID-19 on the sebum lipidome. Earlier studies have shown that patients with COVID-19 do have disrupted lipidomes, as shown by an examination of the blood plasma and nasopharyngeal swab lipids.
The researchers in the current study suggest that since dogs can be trained to sniff out COVID-19 patients, the skin of these individuals is likely to show a dysregulated lipidome. Thus, they say, “Lipidomics, therefore, offers a promising route to a better understanding of - and potentially diagnosis and prognosis for - COVID-19.”
The study centers on examining sebum, the greasy fluid secreted from the sebaceous glands in the skin, using liquid chromatography mass spectrometry. A sebum sample is painless and straightforward to obtain. The study of sebum lipids has been shown to yield characteristic features in illnesses like Parkinson’s Disease. Thus, the researchers sought to establish the sebum lipid patterns distinctive for COVID-19. This could be developed, if so, as a non-invasive sampling method for diagnostic testing in the future.
The COVID-19 International Mass Spectrometry (MS) Coalition was set up in May 2020 by a number of UK institutions to derive information on the virus at molecular level from infected individuals. This would help understand how the virus affects metabolic pathways so as to improve the diagnosis and treatment of COVID-19. The current study is part of the work of this coalition.
Study Group
The study included 67 participants, 30 with and 37 without clinical symptoms of COVID-19. All in the first group had a positive RT PCR test as well. The male-female ratio was slightly higher in the first group, perhaps because the infection is more severe in males.
The first group had a lower prevalence of chronic underlying illness, but diagnostic markers like CRP were higher. Lymphocyte counts were lower, however, while bilateral changes on the X-ray of the chest were more likely to occur in this group. Oxygen requirement was also higher, and they were more at risk of needing CPAP. They had higher odds of progressive disease and lower odds of survival.
Analysis by lipid class
The researchers found that COVID-19 patients had lower levels of triglycerides, diglycerides, and monoglycerides in the serum samples. This suggests a dysregulation of skin lipids. The differences between the levels of these lipids between the first and second groups were similar to those for CRP levels, and thus equally valuable as indicators of the presence of COVID-19.
Earlier work agrees on the existence of disrupted lipid levels in plasma, but the evidence is discordant as to whether lipid levels go up or down. Mild COVID-19, for instance, may be associated with elevated plasma triglyceride (TAG) levels, but this may decline with more severe disease. However, most of the lipid in the skin is synthesized locally rather than being supplied by the blood.
Population-level clustering analyses
Population-level clustering analyses (PCA) failed to show clustering of serum lipid concentrations by type, while OPLS-DA showed some insignificant and non-predictive separation of COVID-19 and non-COVID-19 individuals. When confounding factors like age, CRP, and lymphocyte count were adjusted for, they found that there was little improvement in the predictive or diagnostic value.
By itself, low lymphocyte counts were better strongly correlated with a diagnosis of COVID-19. Age was less important, indicating it does not affect the skin lipids significantly.
Classification by Comorbidity
The model performed better in four subsets when separate comorbidities and medication classified the sample.
These included those patients with a chronic illness that was treated by medication, namely, high cholesterol, type 2 diabetes mellitus (T2DM) and IHD, and those on statins. These subsets had better predictive power as well as the improved separation of COVID-19 positives from negatives. The reasons might include the increased homogeneity of these subsets concerning confounding factors.
Reasons for Increased Predictive Power
When patients are classified by treatment for comorbidity, it leads to a more uniform distribution in each subset. For instance, patients treated for high blood pressure or for T2DM are likely to be of the same range of ages, and even more so for those with high cholesterol. Sex-based subsets also provide adequate separation of cases from negatives.
The researchers postulate that if a more extensive set of patients and controls is used to allow better stratification, the predictive power will rise still more.
Another reason may be that medication use alters the effect of the confounding factors, reducing the patients to a more uniform baseline that is useful to estimate the disturbance resulting from the impact of COVID-19 on the lipidome. For instance, statins are classically used to treat high cholesterol and diabetes and ischemic heart disease as a preventive for better long-term outcomes. Analysis of the statin group yields good predictive power and separation.
Noting that this is a pilot study, the researchers say the risk of overfitting the model can be brought down by using more data for training the model and then validating the model on several sets of data.
Implications and Future Directions
The non-COVID-19 patients in this study were sampled in May, June, or July, and therefore had a lower incidence of other respiratory illnesses caused by seasonal respiratory viruses. This may have led to a potential absence of confounding factors since the latter could also cause lipid metabolism to register a change that could potentially prevent the identification of characteristic COVID-19 features.
In the UK, such viruses do circulate in fall and winter, and this must be considered in future research. Moreover, samples of sebum from COVID-19 patients taken over time will help to identify the time frame along which the sebum lipids become normal again following COVID-19 and the predictive power of these changes, which will determine its application in clinical or mass testing.
The study concludes, “COVID-19 infection leads to dyslipidemia in the stratum corneum.” Sebum lipidomics can help identify COVID-positive and negative patients with greater certainty if they are grouped by comorbidity. The ease with which sebum samples are obtainable, transported, and stored makes this a promising approach for sebum sampling for the diagnosis and prognosis of COVID-19.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources