The current COVID-19 pandemic is caused by an RNA virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), an enveloped virus with spikes composed of glycoproteins studding its bilayered lipid envelope. With its propensity for causing severe or critical pneumonic disease in a significant minority of patients, the virus has become a significant target of diagnostic and therapeutic research. A recent study published on the preprint server arXiv* in October 2020 shows the potential for using functionalized magnetic nanoparticles to detect the presence of the virus in a sample.
Testing is critical to containment efforts since not only symptomatic but asymptomatic patients, and contacts must be tested on a large scale in order to break the chain of transmission. As of now, the gold standard consists of polymerase chain reaction (PCR), which is complicated as well as time-consuming. Moreover, the procedures require sophisticated equipment, with a high cost, and requires operation by technically skilled staff. If carried out improperly, the odds of a false positive or negative result are high.
With an exponentially rising case count, it has become an overwhelming strain to test for COVID-19 in clinical situations. Moreover, in developing and underdeveloped countries, limited healthcare resources are likely to aggravate this difficulty. This lends urgency to the task of developing new methods or instruments to achieve a rapid diagnosis of this infection.
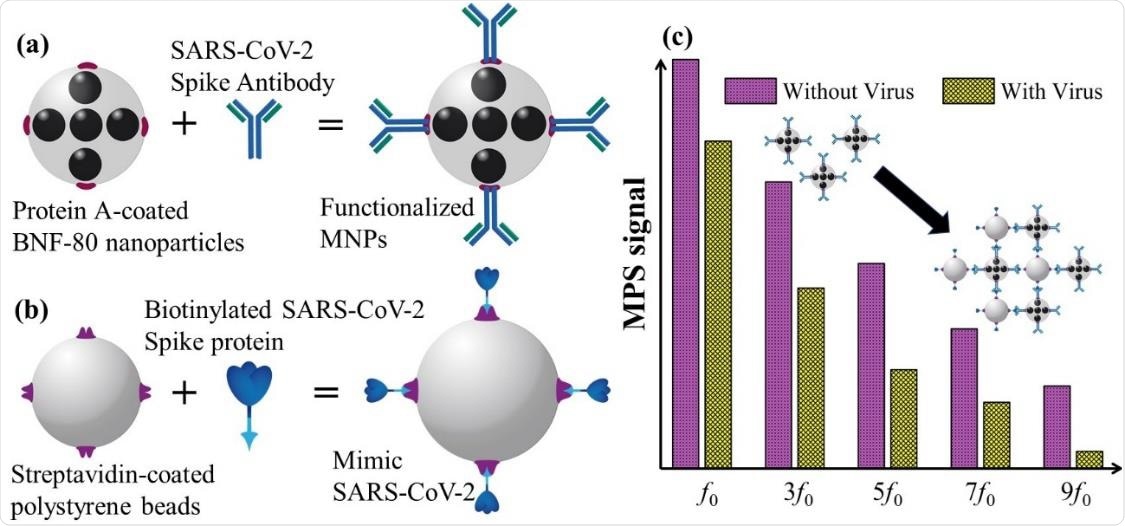
Schematic of functionalized MNPs, (b) schematic of mimic SARS-CoV-2, (c) Schematic of MPS signal with and without mimic virus.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Magnetic Nanoparticles in Particle Detection
One of the upcoming techniques being explored is homogeneous biosensing based on magnetic nanoparticles (MNPs) in order to detect biomolecules of a specific kind. This could include nucleic acids or proteins. The principle of the test is the fluctuation in magnetization and the magnetic characteristics of the MNPs when exposed to magnetic fields varying in intensity over time.
The extent to which the biomolecules bind to functionalized MNPs is dictated by Brownian relaxation, which causes a change in their hydrodynamic size or induces cross-linking. This results in significant alterations in the Brownian relaxation time of the MNPs and the magnetization dynamics when exposed to time-varying magnetic fields.
The researchers quote the example of the observable decrease in harmonics in magnetic particle spectroscopy (MPS) due to the reduction in Brownian relaxation, when MNPs bound with biomolecules are exposed to an alternating current magnetic field of sufficient intensity. The higher harmonics are found to reduce more rapidly than the fundamental harmonic, causing a decrease in the harmonic ratio without relation to the MNP concentration.
This is, therefore a sound basis for the quantitative detection of specific biomolecules, using the measure of MNP magnetization and its susceptibility/spectra in ac magnetic fields. The use of MPS offers an inexpensive, flexible, and sensitive method of determining MNP magnetization and dynamics to detect biomolecules.
Experimental Evidence of Efficient Detection
The current study suggests the use of MPS to measure the binding of the spike protein to MNPs functionalized with antibody, and thus to detect the virus. This proof-of-concept experiment uses these MNPs as sensors to detect fake viral particles, comprising 100 nm polystyrene beads conjugated with the spike protein. They engineered an MPS apparatus for signal measurement, especially the ratio of the 3rd to 1st harmonics.
As well, they used a rotating magnetic field (RMF) system to assess the Brownian relaxation time using ac susceptibility (ACS) spectra. A range of particle concentrations was used to evaluate the sensitivity and the limit of detection (LOD) of this technique, which is found to be 0.084 nM (5.9 fmole).
This setup allows very sensitive detection of the mimic virus, with four repeated measurements being taken over a period of 36 seconds overall. If this is further customized, the LOD can be still further reduced to measure an average of 36 measurements within this period.
Using the ASSURED criteria of the World Health Organization (WHO), this method is found to be a good fit for disease control, being Affordable, Sensitive, Specific, User-friendly (requiring minimal training), Rapid and robust (results require below 30 minutes), and Deliverable to end-users, while using innocuous and cheap materials. The only non-applicable criterion is Equipment-free, but this test can be built as a point-of-care (POC) device at a low cost.
Implications
While current virus detection tests are binary, failing to yield information on the status of infection, this can help to quantitatively detect living virus particles, which will help to tailor treatment and containment measures to the tested individuals. In fact, the detection of the virus is more sensitive than of the spike protein on the functionalized MNPs, due to a much more significant change in hydrodynamic size with the former.
The researchers sum up: “The proposed approach is of great promise to highly sensitive and rapid detection with a low cost, easy handling of the sample to be detected (mix-and-measure).’
This approach can also be adjusted to produce a spatial map of virus distribution that will be useful not only in the containment of the current pandemic but in further fundamental research on the infection and proliferation of the virus.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources