A team of scientists from the United Kingdom has revealed that markers of endothelial cell injury are associated with severe and fatal outcome of coronavirus disease 2019 (COVID-19). The study is currently available on the medRxiv* preprint server.
A growing pool of evidence suggests that the severe form of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is associated with elevated blood levels of inflammatory markers, including interleukin-6 (IL-6) and C-reactive protein.
This is often accompanied by the induction of neutrophil level (neutrophilia) and reduction of lymphocyte level (lymphopenia) in the blood. However, these characteristic features are not specific to COVID-19, as significantly elevated inflammatory marker levels have also been observed in patients with acute respiratory distress syndrome and sepsis.
Similarly, a severe viral or bacterial infection in the human respiratory tract has been shown to cause neutrophilia.
Regarding other prognostic markers, post-mortem studies have shown that fatal COVID-19 is associated with pulmonary vessel vasculitis and microthrombosis. These pathological features are associated with distinct endothelial responses in COVID-19 patients.
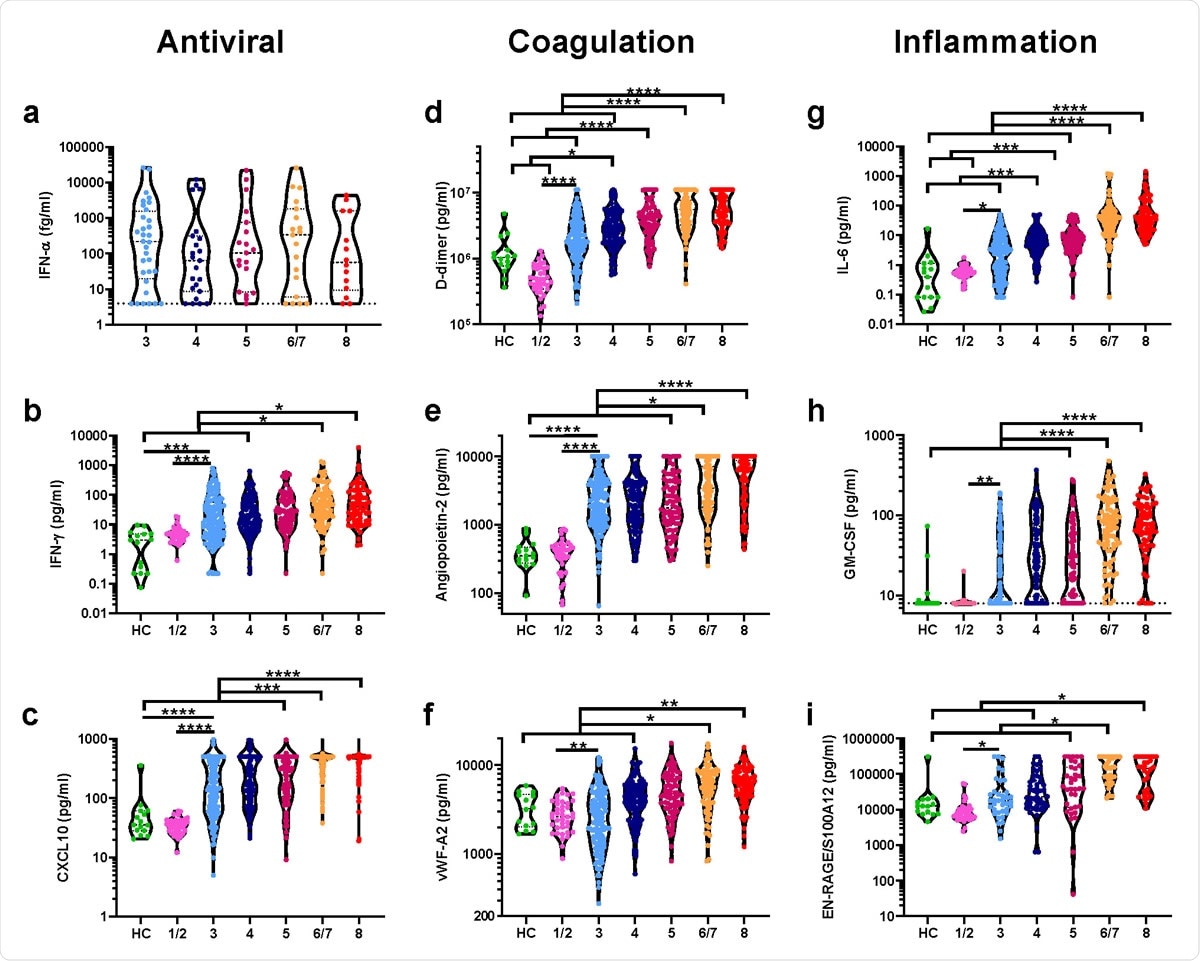
Antiviral, coagulation, and inflammation associated mediators distinguish severity groups early in disease. Plasma samples from the time of study enrolment were analyzed for levels of the antiviral cytokines a) IFN-α, b) IFN-γ, and c) the interferon-induced chemokine CXCL10 in healthy control (HC, n=15), patients with COVID-19 not requiring hospitalization (‘1/2’, n=39), and ospitalized patients with COVID-19 that would: not require oxygen support (‘3’, n=32-128), require an oxygen face mask (‘4’, n=23-103), require non-invasive ventilation or high-flow nasal cannulae ‘5’, n=19-78), require invasive mechanical ventilation (‘6/7’, n=19-87) or progress to fatal disease (‘8’, n=14-69). Mediators associated with coagulation and endothelial injury were also quantified in these plasma samples; d) D-dimer, e) Angiopoietin-2, and f) von-Willebrand factor A2 (vWF-A2). Similarly, mediators associated with inflammation were quantified: g) IL-6; h) GM-CSF; and i) EN-RAGE/S100A12. Data were analyzed for statistical significance using Kruskal-Wallis tests with Dunn’s tests for multiple comparisons between all groups. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Current study design
The scientist from Imperial College London, University of Edinburgh, University of Liverpool, and the Roslin Institute analyzed 1047 plasma samples collected from 619 hospitalized COVID-19 patients to evaluate the correlation between circulating inflammatory markers and disease severity.
The scientists hypothesized that the variation in plasma levels of inflammatory markers would reflect the immunopathology and disease outcome in COVID-19 patients.
Analysis of routine hematological and biochemical parameters revealed no statistically significant difference between COVID-19 patients with different levels of severity at the time of hospital admission.
Important observations
By analyzing 33 inflammatory markers in plasma samples, the scientists were able to differentiate patients with distinct profiles of inflammatory markers. Moreover, they observed that the levels of certain markers, such as D-dimer, EN-RAGE, CXCL10, and GM-CSF, were specifically associated with adverse COVID-19 outcomes.
To further establish their hypothesis, the scientists analyzed plasma samples collected from 15 healthy donors and 39 COVID-19 patients who were not hospitalized.
The results showed that the level of interferon-γ (INF-γ) was elevated in hospitalized COVID-19 patients compared to that in healthy donors and non-hospitalized COVID-19 patients. However, they did not observe any reduction in IFN-α levels among severe COVID-19 patients.
Markers of endothelial injury
The scientists analyzed endothelial injury markers as pulmonary vasculitis is known to be associated with fatal COVID-19 outcomes.
They observed that the levels of angiopoietin-2 and von-Willebrand factor A2 (endothelial injury markers) were elevated in all hospitalized COVID-19 patients, and the elevation was highest in severely affected COVID-19 patients.
Similarly, they observed elevated levels of coagulation markers in hospitalized and severely affected COVID-19 patients. From these findings, they concluded that endothelial injury and coagulation are expected outcomes in hospitalized COVID-19 patients.
Inflammatory markers
Elevated levels of several inflammatory markers, including IL-6, TNF-α, IL-2, GDF-15, G-CSF, and EN-RAGE/S100A12, were observed in hospitalized and severely affected COVID-19 patients.
By performing a hierarchical correlation matrix analysis, the scientists identified a group of inflammatory markers (GM-CSF, CXCL10, vWF-A2, and IL-6), significantly associated with fatal COVID-19 outcomes.
By analyzing the levels of inflammatory markers over the time-course of hospitalization, the scientists observed that while some markers maintained stable expression patterns throughout the course of hospitalization, some markers gradually decreased over time (IFN-γ and CXCL10).
Moreover, some markers, such as angiopoietin-2, D-dimer, GM-CSF, and EN RAGE, remained elevated or increased over time.
From these findings, the scientists concluded that persistent coagulation and inflammation are strong predictors of fatal COVID-19 outcomes.
Compared to non-severe patients, severely affected COVID-19 patients displayed an intensified inflammatory response within first 4 days of symptom onset. Coagulation and endothelial injury markers also showed the similar trend.
However, in case of lung injury markers, such as EN-RAGE, no variation was observed between severely affected and non-severe COVID-19 patients within first 4 days of symptoms.
Study significance
Overall, the study findings indicate that fatal COVID-19 is associated with an early-phase elevation in markers of coagulation, endothelial injury, and inflammation and a late-phase elevation in lung injury markers.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources