The COVID-19 pandemic shows no signs of fading and has prompted energetic efforts to develop effective antiviral therapeutics and vaccines against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). A recent study published on the preprint server bioRxiv* in October 2020 reports more effective viral neutralization by bivalent anti-spike antibodies. This could promote the development of better therapeutics and vaccines.
This virus has about 80% of the same sequences as the earlier SARS-CoV. Both attach to the same host receptor, angiotensin-converting enzyme 2 (ACE2), via the S1 subunit of the viral spike protein. This is followed by the proteolytic cleavage of the spike protein at the S1/S2 interface, with the S2 undergoing marked conformation change shedding the S1 subunit to give rise to the post-fusion form of the spike. This facilitates viral entry into the cell.
The S1 subunit has the receptor-binding domain (RBD, which is targeted by many natural anti-spike antibodies as well as many vaccines under development. This is the part of the S1 subunit that directly attaches to the receptor.
In the prefusion state, the spike structure shows more significant changes in conformation in the S1 region, and this is especially the case with the RBD. This can have either the ‘up’ or ‘down’ conformations, and only the former can attach to the ACE2 receptor.
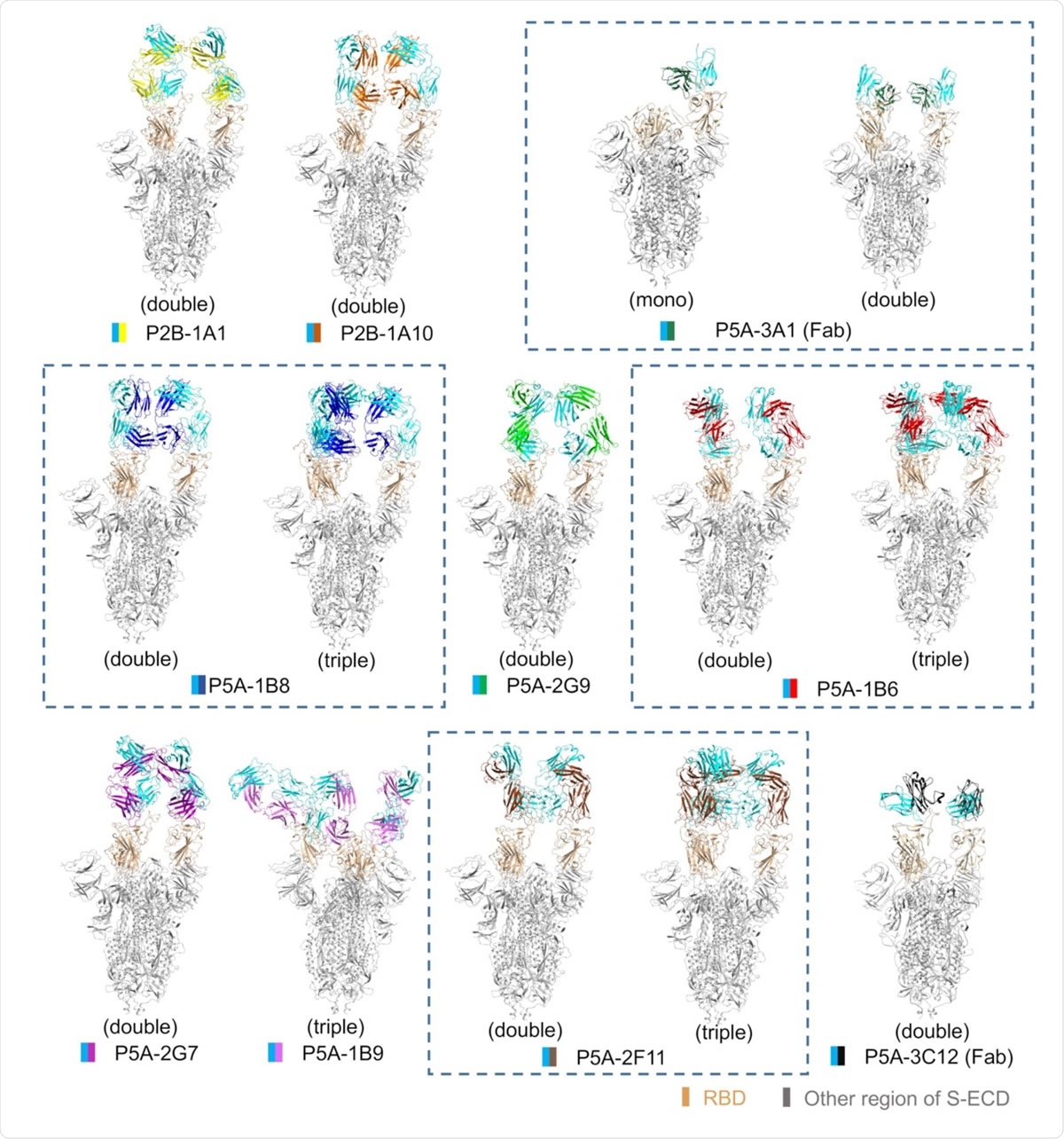
All solved structures of nAbs in complex with S protein. The domain-colored models of all complex are shown here. The structures containing different numbers of the same nAb are boxed with blue dash line. The structures are labeled according to the number of RBD bound with nAb as mono (1 RBD), double (2 RBDs) or triple (3 RBDs) binding, respectively.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Cryo-EM of Antibody-Spike Complex
The current study aims at understanding how neutralizing antibodies (nAbs), which are typically bivalent, bind to the trimeric spike protein. The researchers used ten cryo-EM structures of the spike-nAb (IgG) complex, with either full-length nAb or Fab fragment, or both.
Potent Human Convalescent nAbs
The investigators examined ten nAbs from plasma obtained from recovered COVID-19 patients. All were strongly neutralizing, competing with the ACE2 receptor to bind RBD. They also found that while some nAbs readily induced S1 subunit shedding, others did not.
Structurally, they found three patterns to the nAb-spike complexes. The first included four of the ten nAbs, which bound two ‘up’ RBDs. The second pattern, shown by three nAbs, had either two or three RBDs bound in the ‘up’ conformation to the antibody. Pattern 3 was shown by the most potent nAb examined here, which bound one RBD in the ‘up’ and two in the ‘down’ conformation, all three RBDs sharing the same binding interface.
Bivalent binding of nAbs
Earlier, studies have suggested that bivalent nAbs are more potent compared to Fabs in viruses like Dengue and rhinoviruses. The researchers, therefore, imaged the Fab-Spike complex separately. They found that this showed only two RBDs bound to the nAb, both in the ‘up’ conformation. With the IgG-spike complex, two or three RBDs bind to the nAbs in “up” conformation.
Despite different binding patterns, all full-length antibodies showed bivalent binding of the viral spike protein.
Greater Potency and S1 Shedding for IgG vs. Fab Forms
They also found a difference in the orientation of the RBDs bound to the Fab and the full-length IgG. The half-maximal inhibitory concentration (IC50) values were 700 times lower with the full-length antibody than the Fab form for two of three tested antibodies, while with the third, it was over 3,000 times less.
S1 subunit shedding was found to be at over 80% of the S protein at an incubation time of 120 minutes, with one of the IgG nAbs, while the Fab form of the same antibody caused hardly any shedding. Other nAbs showed mild shedding only.
The full-length IgG nAbs were thus able to bind more strongly and had more potent neutralization capacity, as well as inducing more significant S1 shedding, compared to the Fab fragment binding alone to the spike RBD.
Earlier, S1 shedding has been shown to be a possible correlate of neutralizing power since cross-reactive nAbs to SARS-CoV and SARS-CoV-2 are typically directed against RBD, prevent ACE2 binding, and promote S1 shedding. However, since the native spike protein exists as a homotrimer, it is unknown whether all the S1 subunits have to be shed to allow the spike protein to change from the prefusion to the post-fusion form.
Comparisons of antibody binding epitopes
The structure of the Fab and the scFv was constructed for 8/10 and the remaining 2/10 antibodies, respectively. This showed three groups of antibodies concerning the epitopes and the angles at which they bound to the RBD.
In the first group of 7 nAbs, there was a high overlap between the receptor-binding motif (RBM) on the RBD and the epitopes. These antibodies recognized 8-15 of the 17 epitope residues that mediate binding to the ACE2 receptor. The angles at which the antibodies make contact with the RBD are also comparable between nAbs in this group.
The current study confirms that of the 17 anti- SARS-CoV-2 nAbs in the Protein Data Bank (PDB) database, group 1 nAbs are the most common.
Group 2 contains only one nAb, the most potent nAb tested in this study against both the wildtype and the pseudo-SARS-CoV-2. This antibody shows pattern 3 binding to the RBD. Its direction of the RBD approach is different from that of group 1 nAbs.
Meanwhile, the group 3 nAbs bind to the remote loop of the RBM and have little overlap with the RBM alone, with an unique binding mechanism.
Of the three subgroups in group 1, the first two induce the shedding of over 77% of S1, but this ability is much weaker, at ~44% to ~57%, with subgroup 3 and groups 2 and 3. The researchers feel, “The S1 shedding ability of the nAbs may be facilitated by the large overlap with RBM.”
Subgroup 3 of group 1 nAbs shows rotation of the longitudinal axis of the Fab compared with the antibodies in subgroup 1 and 2. It is possible that only if these antibodies can achieve a specific angle of binding with the RBD can they cause shedding of the S1 subunit.
Again, the presence of the Fc region in the antibody-virus complex can increase the immune response by allowing T cell binding via their Fc receptors.
Implications
This showed that antibody binding in the full-length form uses a different mode and causes more RBDs to flip to the ‘up’ conformation than with the Fab form. The former involves bivalent and the latter monovalent binding. Bivalent binding is more effective in inhibiting viral entry and is associated with more significant S1 subunit shedding.
Earlier, the same researchers showed the dimeric nature of the ACE2 receptor. If so, each of the ACE2 molecules can bind one of the RBDs in the S trimer, thus inducing the bound RBD to flip ‘up’. Steric hindrance probably prevents the binding of the two ACE2 molecules in the dimer to different RBDs of the S trimer at the same time, and thus to flip them ‘up.’ This would require the binding of more than one ACE2 receptor.
Whereas earlier studies showed many complex spike-nAb structures, most of them used the Fab form of the antibody, particularly if X-ray crystallization was used for the structural visualization. The current study indicates that the full-length IgG is more physiological and should be preferred for structural analysis to shed light on the actual structure and function of nAbs targeting SARS-CoV-2.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources