Even as the global death toll of the COVID-19 pandemic crosses 1.5 million, with no signs of fast subsidence, researchers are hunting for effective antivirals, both therapeutic and preventive, among older drugs as well as developing newer compounds. Among these, many leads are showing promise of safety and efficacy against the virus.
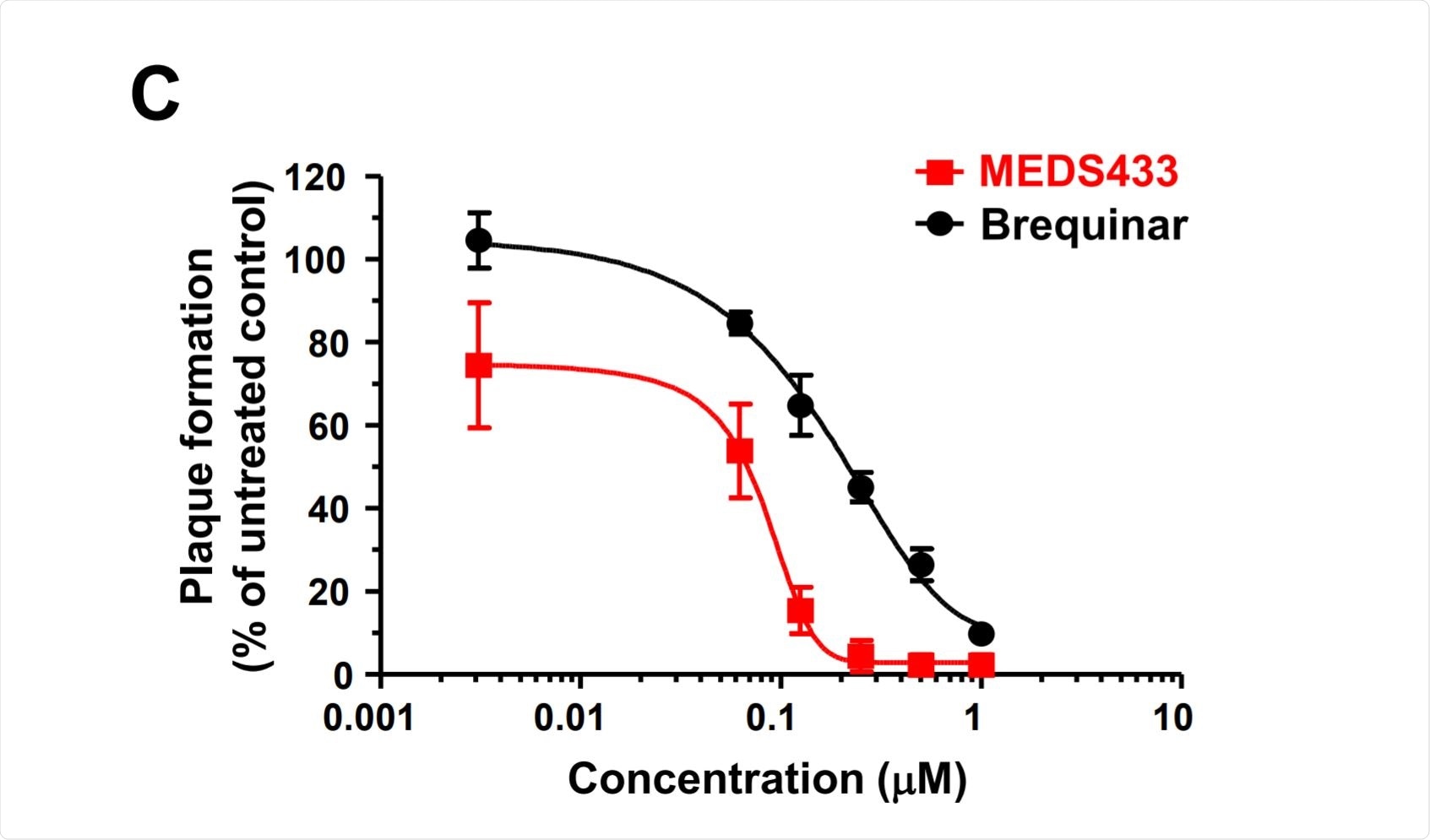
Antiviral activity of MEDS433 on SARS-CoV-2 replication. Image Credit: https://www.biorxiv.org/content/10.1101/2020.12.06.412759v1.full.pdf

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Small Molecules Targeting Host Factors
Small molecules targeting various host factors that are essential for viral replication are being explored as an attractive alternative to more direct antivirals that inhibit viral enzymes. This is because the possibility of escape mutations exerting positive selection pressure on the virus is low in the former scenario, allowing these drugs to retain their efficacy for a longer period.
Among the small molecule candidates, one possibility for an effective antiviral includes inhibitors of RNA synthesis. Like DNA, RNA is composed of a nucleotide backbone, comprising two types of nitrogenous bases, purines, and pyrimidines. These are synthesized in the body de novo. One of the key enzymes involved in pyrimidine biosynthesis is human dihydroorotate dehydrogenase (hDHODH).
This enzyme is therefore essential for viral replication within the infected cells. It is responsible for the conversion of dihydroorotate to orotate, a rate-limiting step in de novo pyrimidine synthesis since it results in the production of uridine and cytidine for incorporation into the growing nucleic acid molecule. This being a crucial step, hDODH is grabbing much interest among scientists who are investigating host-targeting antivirals (HTA) against this virus.
As of now, two powerful hDODH inhibitors are about to enter phase II clinical trials for COVID-19. These are brequinar and PTC299. These have significant toxicity, however, which led to an arrest of their development for other clinical uses. This indicates the need to identify safer inhibitors of hDODH.
MEDS433 Suppresses N Accumulation
The current investigators looked at a derivative of brequinar, called MEDS433. It possesses a 2-hydroxypyrazolo[1,5-a]pyridine group. This new-generation hDODH inhibitor has comparable activity to brequinar, but its pharmacological parameters are superior to the latter.
To understand the potential of this compound as a lead for HTA development, the researchers looked at its effect on the replication of MEDS433 on Vero E6 cells infected with the wild-type virus, isolated from a clinical specimen. The infection was carried out at a multiplicity of infection (MOI) of 0.1 – that is, the number of viral particles added to the cell was 1,00,000.
Subsequently, MEDS433 was added at 0.5 micromoles. At 24 hours from infection, about 85% of infected cells were found to contain N protein in the absence of MEDS433. This compound, however, completely prevented the build-up of this viral protein. This shows that the de novo synthesis of pyrimidines is an effective target for the prevention of N protein expression in SARS-CoV-2 infected cells.
MEDS433 Inhibits Viral Replication
The researchers also performed virus yield reduction assays in the same cell line, first infecting them with the virus and then adding MEDS433 at increasing titers. At 48 hours from infection, they observed that viral replication was inhibited in a dose-dependent manner.
The half-maximal effective concentration EC50 was 0.063 micromoles, while the EC90 was 0.136 micromoles. This shows its greater efficacy, relative to brequinar with an EC50 and EC90 of 0.20 and 1 micromole, respectively.
MEDS433 Has Wide Therapeutic Margin
They also measured the half-maximal cytotoxicity concentration CC50, which was over 500 micromoles, indicating a large therapeutic window. The selection index (SI) of over 7,900 is very favorable and supports its specific antiviral activity, rather than a loss of viral replication simply because the compound destroyed the infected cell.
MEDS433 Acts in Biosynthetic Phase
Next, they conducted time-of-addition experiments, where the Vero E6 cells were exposed to the compound at 0.5 micromoles at time points from 2 hours before until 1 hour after SARS-CoV-2 adsorption on the cells, at an MOI of 0.1 – the pretreatment phase; at 1 hour before to the time of adsorption (co-treatment); and from 0 to 48 hours from infection (the post-treatment phase). The infectious viral particles in the cell supernatants were then measured by plaque assay, at 48 hours from infection.
They found that MEDS433 acted to reduce viral particle production only in the post-entry treatment step, which supports its role in preventing the build-up of N protein. In other words, this compound inhibits a post-entry biosynthetic step in viral replication.
To make sure that MEDS433 inhibited de novo pyrimidine synthesis, they used the same cell line, and examined the effect of adding uridine to the culture medium in increasing concentrations, to overcome the supposed inhibition of uridine biosynthesis in the presence of this compound.
The plaque reduction assay showed that MEDS433 lost its antiviral activity significantly when the concentration of uridine was a hundred times over the MEDS433 concentration. At still higher uridine levels, MEDS433 lost all antiviral activity.
MEDS433 is Inhibited by Orotic Acid
At this point, the investigators attempted to prove that MEDS433 inhibition of SARS-CoV-2 replication was solely due to its ability to inhibit hDODH, by supplementing dihydroorotate acid or its product, orotic acid, in the culture medium. They found the same results; the addition of orotic acid first reversed and then arrested the antiviral effect of MEDS433, the complete effect being seen at a concentration 1,000 times that of MEDS433.
Dihydroorotic acid was unable to achieve this even when raised to a concentration more than 3,000 times that of MEDS433. This confirms that the latter acts on a step after dihydroorotate acid is formed. Specifically, this drug inhibits hDODH in SARS-CoV-2 infected cells, and thus prevents or reduces viral replication.
Conclusion
This suggests the feasibility of pursuing MEDS433 as a potential antiviral agent in the current pandemic. With its increased effectiveness relative to brequinar, and a better safety profile compared to PTC299, this drug should be studied further to confirm its efficacy in the treatment of this infection in animal models, during the preclinical phase of research.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources