The ongoing coronavirus disease 2019 (COVID-19) pandemic is caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). The virus binds to the target host cell via the angiotensin-converting enzyme 2 (ACE2). This receptor is also part of the renin-angiotensin system (RAS) that regulates the cardiovascular system and renal function.
ACE2 is a RAS inhibitor. Some animal studies show that among the RAS inhibitors (RASIs), some may increase ACE2 levels, thus promoting viral entry into human cells. The combination of this action with their extensive use in a population already at increased risk for severe COVID-19 has been a major issue with many physicians.
However, repeated exploration of this aspect by epidemiological studies have failed to show any association of severe COVID-19 with the use of RASIs. A new study, appearing as a preprint on the medRxiv* server, demonstrates the protective effect of these drugs in older COVID-19 patients with hypertension.
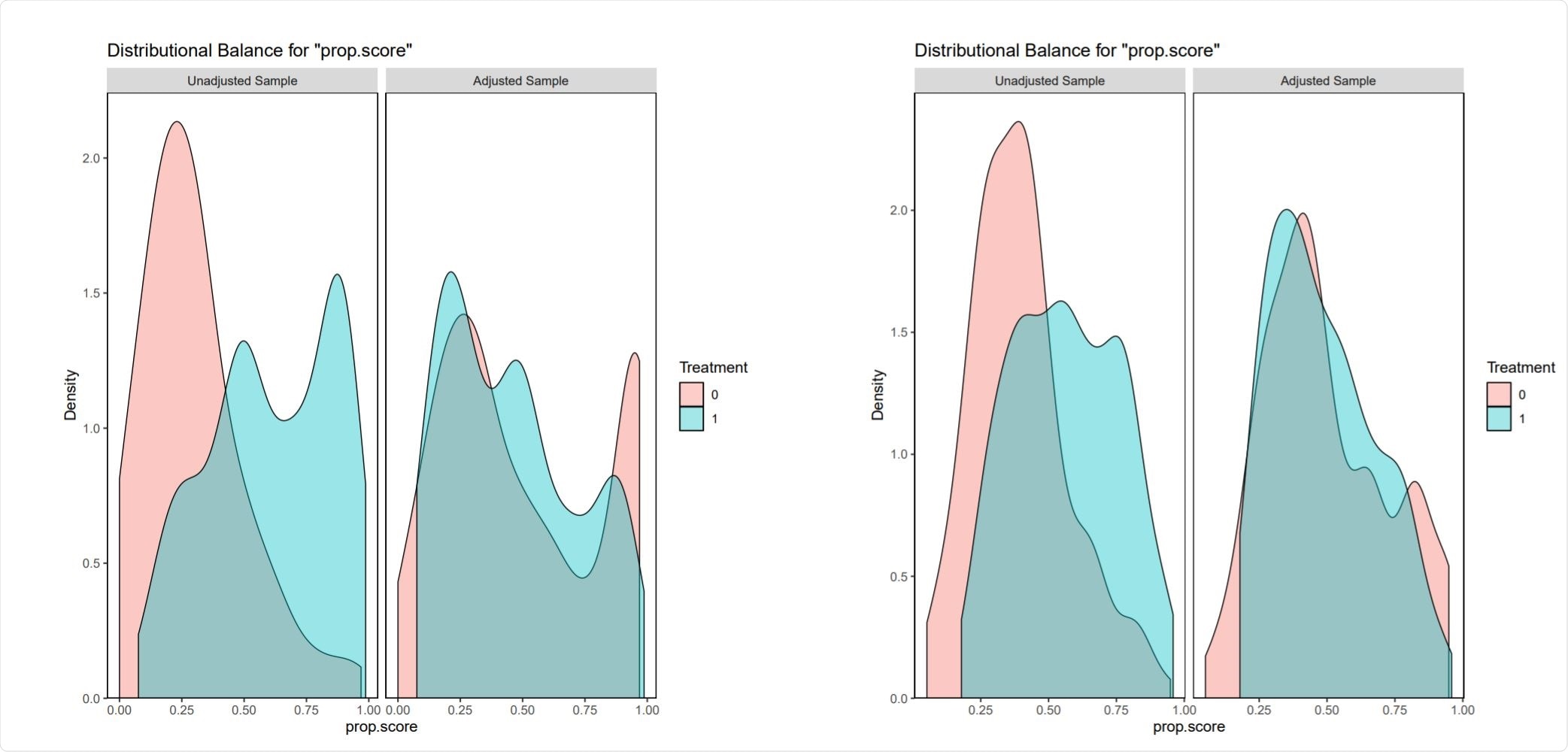
Visual assessment of the degree of balancing achieved by the matching in our analyses of the effects of ARBs. Image Credit:

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Renin-angiotensin system inhibitors
COVID-19 is known to affect certain groups disproportionately in terms of severe or critical disease and mortality. The drugs of concern here are called RAS inhibitors (RASIs) and are widely prescribed for the treatment of high blood pressure and certain heart conditions. These diseases are also known to be high-risk conditions for severe COVID-19.
Researchers have been questioning whether the use of RASIs affects the outcome of COVID-19. This study reports that, despite this association, the use of these drugs is linked to lower mortality among older patients with COVID-19.
Study details
The researchers examined the medical history of over 1 300 successive admissions with COVID-19 between February 23 and April 7, 2020. The median follow-up period was over a month. Their analysis showed that the use of RASIs reduced mortality among elderly patients with hypertension who developed COVID-19, even when other medical risk factors were adjusted for. This effect was strikingly absent in younger patients with hypertension and COVID-19.
RASIs fall into two categories of medication, the angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs).
The investigators classified patients using ACEIs separately from those using ARBs. They then stratified both categories by age so as to accurately estimate the effect size of RASIs in the older patients who were at more risk for severe COVID-19.
In approximately 1,300 patients, there were 353 deaths, accounting for just over a quarter of the patient cohort. There were 486 patients on RASIs at admission. These were compared for mortality with the other 866 patients. Of the total cohort, 968 were men and the median age was 68 years.
However, in patients taking RASIs, the median age was 73 years vs. 65 years in the non-RASI cohort. The former were more likely to have other coexisting diseases, such as hypertension at ~94% vs. ~27%; diabetes at 30% vs. ~13%, angina or a history of cardiac revascularization by stenting, bypass surgery, etc., in ~17% vs. ~8%. Other conditions such as chronic heart failure, history of a heart attack, atrial fibrillation, and vascular disease were also more common in the RASI group.
COVID-19 patients on RASIs were more likely to be on diuretics, beta-blockers, calcium channel blockers, statins, anti-diabetic agents, and anti-platelet drugs.
Earlier study reached ‘rash’ conclusion
In support of this analytical practice, they cite an earlier study focused on the effect of RASIs being used prior to the development of COVID-19.
Prior use of ACEI/ARBs was not significantly associated with COVID-19 diagnosis or mortality among patients with hypertension.”
In the current paper, the authors describe this conclusion as ‘rash,’ commenting, “Had they taken age-related effect modification into account, thereby avoiding effect dilution due to inclusion of young patients, they would have probably seen evidence of a statistically significant Covid-19-beneficial effect of ACEI/ARBs in the older age.”
As the analysis narrowed from the whole group of COVID-19 patients to only hypertensives, and then to older hypertensives, a clear protective effect became observable.
While 32% of the whole group was on RASIs, 24% were hypertensives not on RASIs. Among hypertensives, 32% were on RASIs, and 40% not on RASIs. Among older hypertensives, 40% were on RASIs, and 58% not on RASIs.
Prior RASI use associated with lower risk of death
The final outcome among this older hypertensive group revealed that RASI use prior to hospitalization was associated with lower mortality, which was reduced more markedly in the ARB users. The analysis did not include those on both drug types.
Those patients taking ACEI had a 43% drop in mortality compared to non-RASI users in the above-68 hypertension group. The long-term use of ARBs was linked to a more than 50% drop in the risk of death. Both these groups had similar clinical status at admission, age profile, laboratory test parameters, and therapeutics usage patterns.
The adjusted risk for admission to the intensive care unit (ICU) was similar between ARB users and non-RASI users, as well as the adjusted risk of using a variety of anti-COVID-19 treatments, such as tocilizumab, hydroxychloroquine, antibiotics, and corticosteroids.
Among the RASI users over the age of 68 years, 138 continued to use these drugs after admission. There were more ACEI users than ARB users, though not by much. The number of patients who continued their medication was higher among the former. Crude death rates were higher for those who stopped RASIs compared with those who continued them.
Possible mechanisms of benefit
The offered explanation is “the ability of RASIs to avert Covid-induced cardiovascular complications.” These drugs may oppose the effects of angiotensin II (Ang II), which is harmful to cardiovascular health.
In fact, an earlier study of 12 COVID-19 patients showed significantly higher Ang II levels, with a direct linear correlation between this, the viral load, and the occurrence of liver injury. The high Ang II levels may cause acute respiratory distress syndrome, ARDS, in such patients. This rise would be prevented by RASIs, protecting this older hypertensive subgroup.
Again, the higher expression of ACE2 in older people may contribute to the increased benefit seen with RASI use in older hypertensives. RASIs also prevent abnormal clotting and could therefore inhibit the thromboembolic phenomena seen in severe COVID-19.
Future directions
The current study, therefore, appears to demonstrate that the prior use of ACEIs or ARBs is associated with a lower risk of death in COVID-19, in elderly hypertensives, after adjusting for a large panel of medical variables.
Further research will elucidate the timing at which RASI use should be initiated, whether before the infection or if starting the COVID-19 patient with hypertension on these agents is adequate. Randomized controlled trials may reveal whether these drugs should be continued in hypertensive patients or not, following admission with COVID-19.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources