Researchers in Portugal have provided evidence that might explain why infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes mild or even asymptomatic illness in some individuals, but severe coronavirus disease 2019 (COVID-19) in others.
Salome Pinho from the University of Porto and colleagues showed that circulating T cells exhibit a specific “glycan switch” following infection with SARS-CoV-2 and that this switch is more pronounced in asymptomatic versus symptomatic individuals.
The researchers say this change in the T cell glycosylation profile appears to be triggered by a serum inflammatory factor, the identification of which could lead to a potential new biomarker and therapeutic target.
The team also demonstrated that circulating monocytes in asymptomatic patients exhibit up-regulated expression of a protein called Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin (DC-SIGN).
Furthermore, a higher level of DC-SIGN expression in monocytes was correlated with a better patient prognosis.
“These new findings pave the way for identification of a novel glycan-based response in T cells that may confer protection against SARS-CoV-2 infection in asymptomatic patients, highlighting a novel prognostic biomarker and potential therapeutic target,” writes the team.
A pre-print version of the research paper is available on the medRxiv* server, while the article undergoes peer review.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
SARS-CoV-2 infection has highly variable effects on people
The disease course following SARS-CoV-2 infection is highly variable, ranging from mild or even asymptomatic to severe illness characterized by pneumonia, acute respiratory distress syndrome and multi-organ failure.
Although critical illness usually develops in older individuals, some relatively young and healthy individuals also develop severe COVID-19.
Pinho and colleagues say a better understanding of patient-specific immune responses is urgently needed to help identify prognostic biomarkers and therapeutic targets, and to improve the stratification of patients for vaccination.
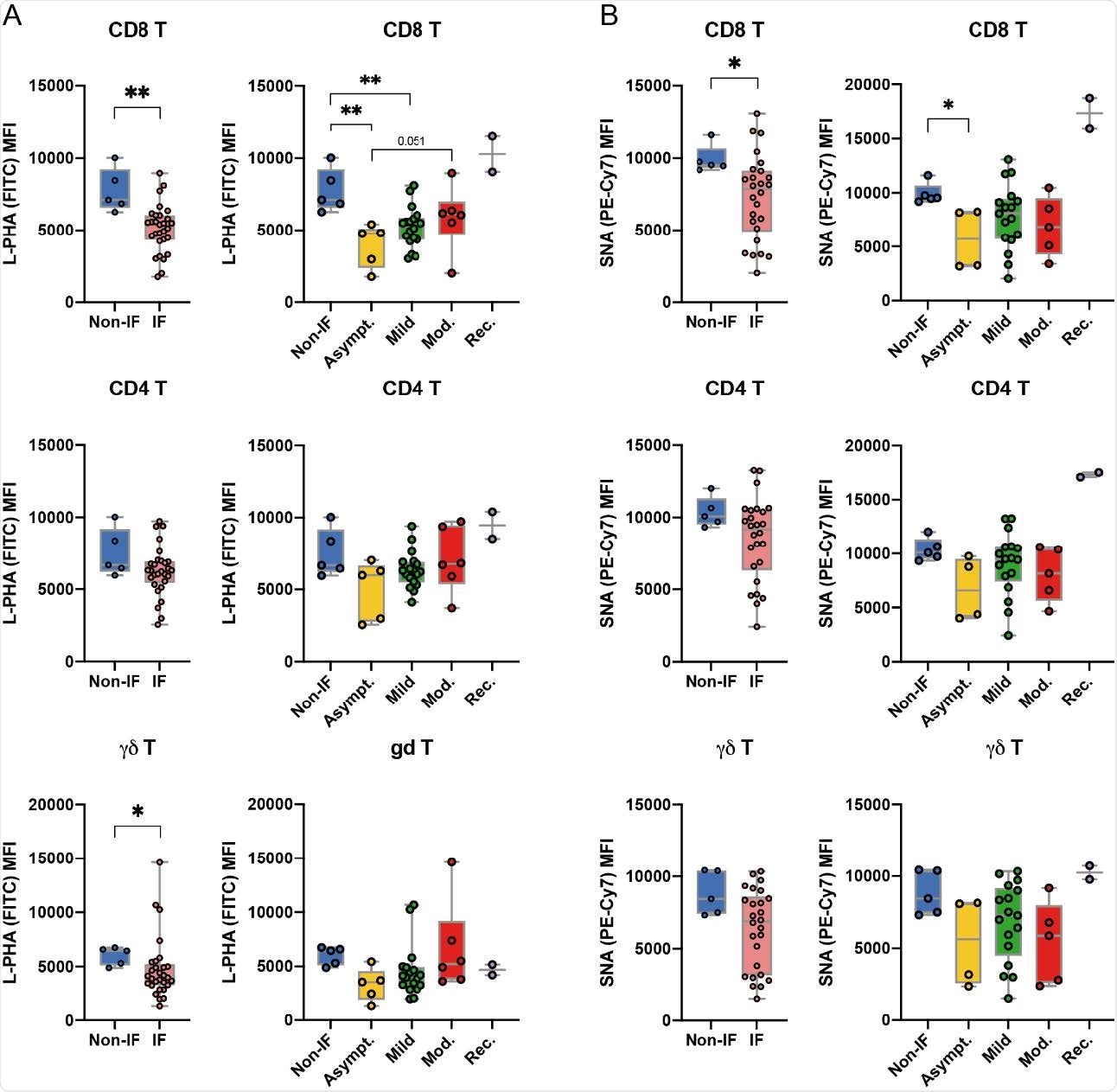
Peripheral T cell glycoprofile is altered upon SARS-CoV-2 infection at diagnosis. Levels (Mean intensity fluorescence, MFI) of (A) L-PHA lectin binding, detecting β1,6-GlcNAc branched N-glycans and (B) SNA lectin binding, detecting a-2,6 sialylation, in CD8+ , CD4+ and γδ T cell subsets of non-infected donors (Non-IF; n=5) and infected (IF; n=30) patients. Levels of (A) L-PHA and (B) SNA lectin binding for each disease severity group: asymptomatic (Asympt.;n=5), mild (n=18), moderate (Mod.; n=6) and recovered (Rec; n=2) Each dot represents one patient. Mann-Whitney t-test was performed to evaluate statistically significant differences between each group-pair. * p-value<0.05, **<0.005.
Where do glycans come in?
Glycosylation refers to the controlled, enzymatic addition of glycans (carbohydrates) to proteins or lipids present on cells. Glycans are significant regulators of immune cell function and determine the activation and differentiation of T cells.
Pinho and the team have previously shown that glycans modulate the T cell activation thresholds associated with immunopathogenesis in autoimmune diseases and cancer.
As is the case with most viruses, the SARS-CoV-2 viral envelope is highly glycosylated with structures such as oligomannose and branched N-glycans.
These structures are specifically recognized by glycan-binding proteins >such as DC-SIGN, which promote viral recognition and elimination.
However, “the glycosylation profiles of immune cells in SARS-CoV-2-infected individuals and how they impact their effector functions remain completely unknown,” says the team.
What did the current study involve?
Pinho and colleagues analyzed blood samples taken from 32 infected patients hospitalized between May 2020 and July 2020 and compared them with samples taken from healthy, uninfected controls.
The researchers demonstrated that SARS-CoV-2 infection induced a change in the glycosylation profile of circulating T cells.
More specifically, T cells from infected individuals exhibited a decrease in branched N-glycan structures (particularly on CD8+ and γδ T cells), compared with T cells from non-infected individuals.
This specific “glycan switch” was more pronounced in the T cells of asymptomatic patients than in those of symptomatic patients.
The results suggest that this glycan switch was triggered by the occurrence of a serum inflammatory factor shortly following infection.
“It is essential to identify the critical serological factor(s) that instructs this T cell glyco-reprogramming, as a potential new biomarker and therapeutic target,” said Pinho and colleagues.
The study also demonstrated that patients with a good prognosis exhibited up-regulated expression of DC-SIGN in circulating monocytes.
“This study reveals the identification of a specific glycosignature of T cells, as well as a prognostic biomarker in COVID-19,” writes the team.
What are the implications of the study?
“We herein propose that the immune response against SARS-CoV-2 infection appears to be influenced by the glycosylation profile of circulating T cells, which defines their effective functions,” writes the researchers.
Pinho and colleagues say the findings pave the way for the identification of a specific blood glycosignature that could be used to stratify patients at diagnosis according to their risk of worsening disease.
“This will certainly contribute to improving vaccination strategy and patient risk stratification, optimizing effective allocation and management of healthcare resources such as ventilators and intensive care facilities,” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Pinho S, et al. SARS-CoV-2 infection drives a glycan switch of peripheral T cells at diagnosis. medRxiv, 2021. doi: https://doi.org/10.1101/2021.02.17.21251918, https://www.medrxiv.org/content/10.1101/2021.02.17.21251918v1
- Peer reviewed and published scientific report.
Alves, Inês, Manuel Machado Vicente, Joana Gaifem, Ângela Fernandes, Ana Mendes Dias, Cláudia Sousa Rodrigues, José Carlos Oliveira, et al. 2021. “SARS-CoV-2 Infection Drives a Glycan Switch of Peripheral T Cells at Diagnosis.” The Journal of Immunology 207 (6): 1591–98. https://doi.org/10.4049/jimmunol.2100131. https://journals.aai.org/jimmunol/article/207/6/1591/234181/SARS-CoV-2-Infection-Drives-a-Glycan-Switch-of.