A team of scientists from Sweden and India has recently characterized the host cell metabolic alterations associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Their findings reveal that SARS-CoV-2 modulates the host cell's central carbon metabolism to facilitate replication and infection propagation. Moreover, they observe that the severity of coronavirus disease 2019 (COVID-19) primarily depends on the blood levels of glucose, mannose, and glutamate. The study is currently available on the bioRxiv* preprint server.
Since its emergence in late December 2019 in Wuhan, China, SARS-CoV-2, the causative pathogen of COVID-19 disease, has infected 114 million people and claimed 2.52 million lives globally.
Although 80% of COVID-19 patients remain asymptomatic or mildly symptomatic, the risk of developing severe disease is higher among individuals with metabolic comorbidities, such as diabetes and obesity. Moreover, there is evidence showing that COVID-19 severity is associated with a number of metabolic alterations, including increased amino acid and fatty acid synthesis and altered lipid and energy metabolism.
Regarding viral life-cycle, it is known that glucose and glutamine as extracellular carbon sources are typically required for the viral replication and that a virus is capable of modulating a number of host cell metabolic pathways, including central carbon metabolism, of facilitating its life-cycle.
Regarding SARS-CoV-2 infection, studies have shown that targeting glycolysis and PI3K/AKT signaling pathways by small molecule inhibitors can lead to a significant reduction in viral load in infected cells.
In the current study, scientists have explored the involvement of key host cell metabolic pathways in SARS-CoV-2 replication. They have also investigated whether alterations in host cell metabolic profiles are associated with COVID-19 severity.
Study design
The scientists analyzed 92 inflammatory mediators in plasma using a targeted proteomics approach. Moreover, they conducted plasma metabolic profiling using untargeted metabolomics, followed by lymphocyte and monocyte immune phenotyping towards metabolite transporters.
In a separate set of in vitro experimentations, they conducted quantitative untargeted proteomics using SARS-CoV-2-infected lung, liver, kidney, and colon cells to understand virus-mediated metabolic remodeling.
Important observations
The proteomics and metabolomics analyses were conducted using plasma samples collected from hospitalized COVID-19 patients having either mild or severe form of the disease. The targeted proteomics data revealed significantly elevated cytokines and chemokines in both mild and severe COVID-19 patients. Interestingly, the scientists observed a reduced level of interleukin 12 (IL-12) in severe COVID-19 patients compared to that in mildly affected patients.
A total of 444 significantly altered metabolites were identified in COVID-19 patients using untargeted plasma metabolomics. The majority of these metabolites were lipids and amino acids. With further analysis, the scientists noticed that amino acid metabolism pathways were primarily affected by SARS-CoV-2 infection. Interestingly, they observed that the levels of glycolysis and TCA cycle-associated metabolites varied significantly between COVID-19 patients with diverse disease severities.
While comparing mild and severe COVID-19 patients, the scientists observed that various amino acid-related pathways, insulin signaling pathways, and macrophage-mediated IL-12 production and signaling were maximally affected in severe patients. They identified plasma levels of glucose, mannose, and glutamate as the major determinants of disease severity with further analysis. Although they observed an elevated plasma level of mannose-binding lectin in COVID-19 patients, there was no association between the mannose and mannose-binding lectin levels.
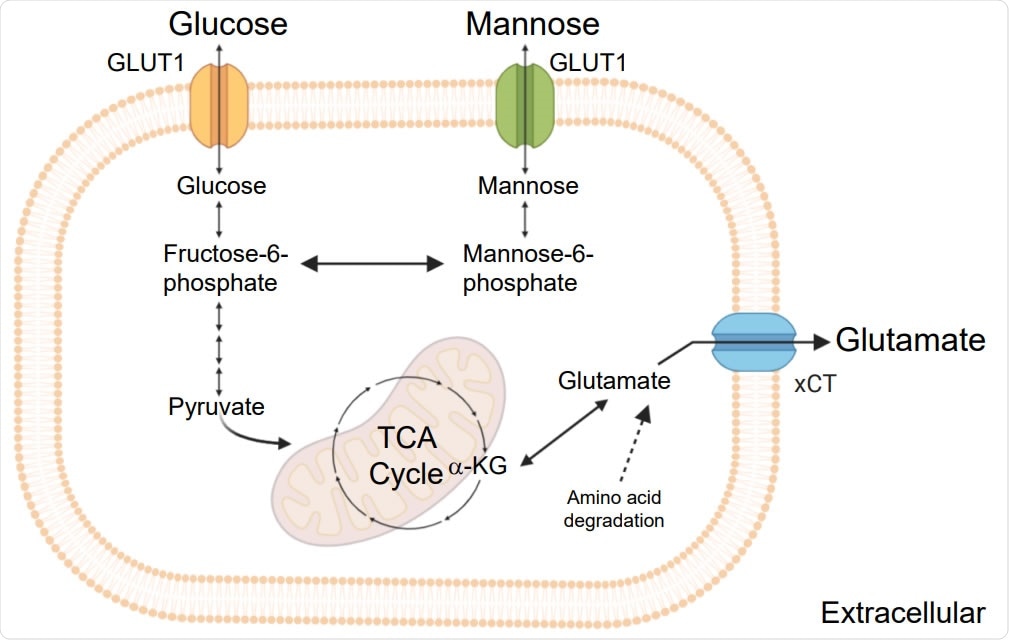
Schematic representation of the key steps of glycolysis, mannose and glutamate metabolism and TCA cycle.
Given that the metabolite transporters can regulate the functions of immune cells, such as lymphocytes and monocytes, by controlling nutrient supply, the scientists conducted immune phenotyping of glucose, mannose, and glutamate transporters. They observed significantly reduced lymphocyte levels, mildly elevated intermediate monocyte levels, and noticeably decreased non-classical monocyte levels in COVID-19 patients. These observations indicate a significant involvement of monocytes in altered immune responses during SARS-CoV-2 infection. Interestingly, they observed significantly elevated expressions of metabolite transporters in all subpopulations of monocytes studied.
The in vitro experimentations conducted to evaluate acute host cell metabolic alterations in response to SARS-CoV-2 infection revealed that proteins associated with glycolysis/gluconeogenesis and fructose and mannose metabolism were significantly elevated in lung cells whereas mitochondrial TCA cycle proteins were significantly reduced. This indicates that SARS-CoV-2 infection might cause mitochondrial dysfunction.
Apart from glucose and glutamate, they observed elevated pyruvate levels, lactate and α-ketoglutarate, indicating significant involvement of glycolysis and glutaminolysis pathways in SARS-CoV-2 infection. Given these findings, they blocked these pathways and observed a significant reduction in viral replication. By varying the amount of glucose and mannose in culture media containing SARS-CoV-2-infected cells, they observed that viral replication is affected by increased glucose levels.
Study significance
The study indicates that to facilitate replication, SARS-CoV-2 modulates host cell central carbon metabolism, converting carbohydrates into precursors for metabolism. Moreover, the study identifies metabolites of the carbohydrate and amino acid metabolism pathways as potential biomarkers for predicting COVID-19 severity.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources