Mar 25 2021
NHS Organisations can apply for funding from the Pathway Transformation Fund (PTF) to obtain a NObreath® used for asthma diagnosis and management.
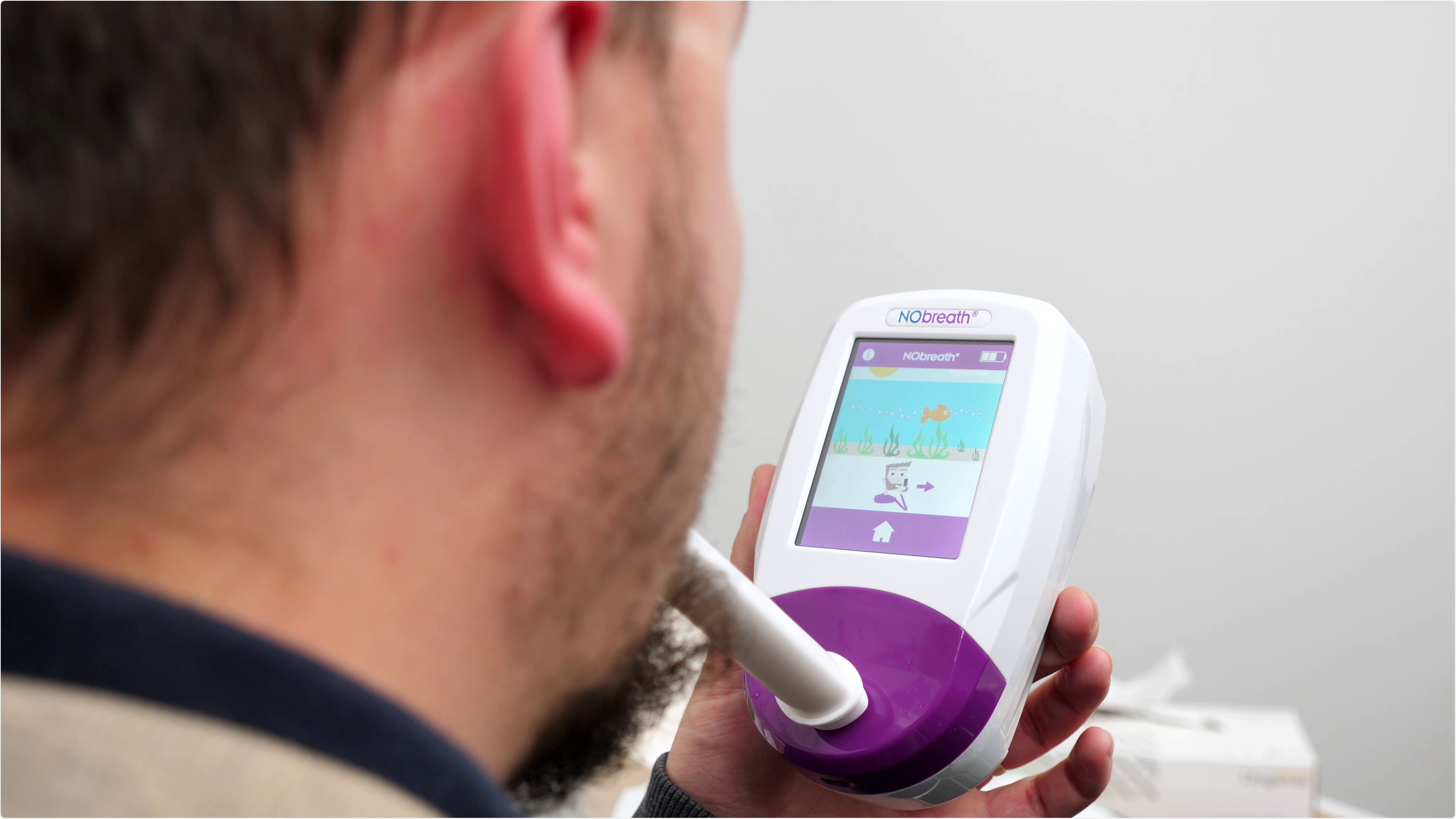
Image Credit: Bedfont Scientific
The NObreath® FeNO monitor from Bedfont® Scientific Ltd. has been included on the Rapid Uptake Products (RUPs) scheme by the NHS Accelerated Access Collaborative (AAC). One of the potential benefits of being part of the RUP programme is that NHS organisations can apply for a one-off funding from the Pathway Transformation Fund (PTF) to fund adoption through a competitive process.
The RUP programme is a partnership between the AAC, its 15 AHSN delivery partners, and a range of several patient groups, government bodies, and NHS bodies, who work together to help the adoption of new innovations in healthcare. The AAC identifies products with NICE approval, such as the NObreath® FeNO monitor, and supports them by helping these innovative products integrate into everyday practice.
The NObreath® FeNO monitor, from Bedfont®, is a quick, simple and non-invasive breath analysis monitor to aid in asthma diagnosis and management, providing a better procedure for both the GP and the patient. FeNO has often been described as the missing piece of the jigsaw; using it alongside existing asthma criteria aids in identifying patients who do/do not require on-going treatment1, whilst also differentiating between allergic (eosinophilic) and non-allergic asthma2, and if used daily, FeNO measurements can help to predict exacerbations and attacks3.
Jason Smith, Managing Director at Bedfont, explains, “Using FeNO measurements to evaluate airway inflammation in asthma represents a significant advance in respiratory medicine. We’re very excited to have the NObreath® on the RUP scheme and look forward to making a difference in asthma care. Bedfont will be hosting a series of free webinars to help integrate NObreath® into NHS services and provide help with PTF applications.”
To sign up for the first webinar, Optimising Asthma Care in Patients using FeNO, follow this link: https://us02web.zoom.us/meeting/register/tZMlce-opzwjGdYc78uXtWPkHIsfbrIAhe8c
The deadline for submitting PTF applications to support the spread and adoption of FeNO testing is 30th April 2021. It is recommended that applications are submitted with the support of your local Academic Health Science Network (AHSN) RUP lead. For more information on how to apply, please contact [email protected].
References
- Andrew D. Smith, Jan O. Cowan, Sue Filsell, Chris MacLachlan, Gabrielle Monti-Sheehan, Pamela Jackson and D. Robin Taylor. Diagnosing Asthma: Comparisons between Exhaled Nitric Oxide Measurements and Conventional Tests. Am J Respir Crit Care Med Vol 169. pp 473-478, 2004.
- Coumou HBel E. Improving the diagnosis of eosinophilic asthma [Internet]. Taylor and Francis online. 2017 [cited 22 March 2021]. Available from: http://www.tandfonline.com/doi/full/10.1080/17476348.2017.1236688
- Harkins M. Exhaled Nitric Oxide Predicts Asthma Exacerbation [Internet]. Taylor & Francis. 2017 [cited 22 March 2021]. Available from: http://www.tandfonline.com/doi/abs/10.1081/JAS-120033990