Researchers in the UK have demonstrated the potential of modulating the expression of angiotensin-converting enzyme 2 (ACE2) as a novel host-directed therapeutic approach to protecting against coronavirus disease 2019 (COVID-19).
The human ACE2 receptor is the main host structure that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) binds to as the initial stage of the infection process.
“Because host-directed treatments do not target the virus, they are less susceptible to resistance and more likely to be effective against multiple viral variants,” writes Fotios Sampaziotis from The Wellcome Medical Research Council Cambridge Stem Cell Institute and colleagues.
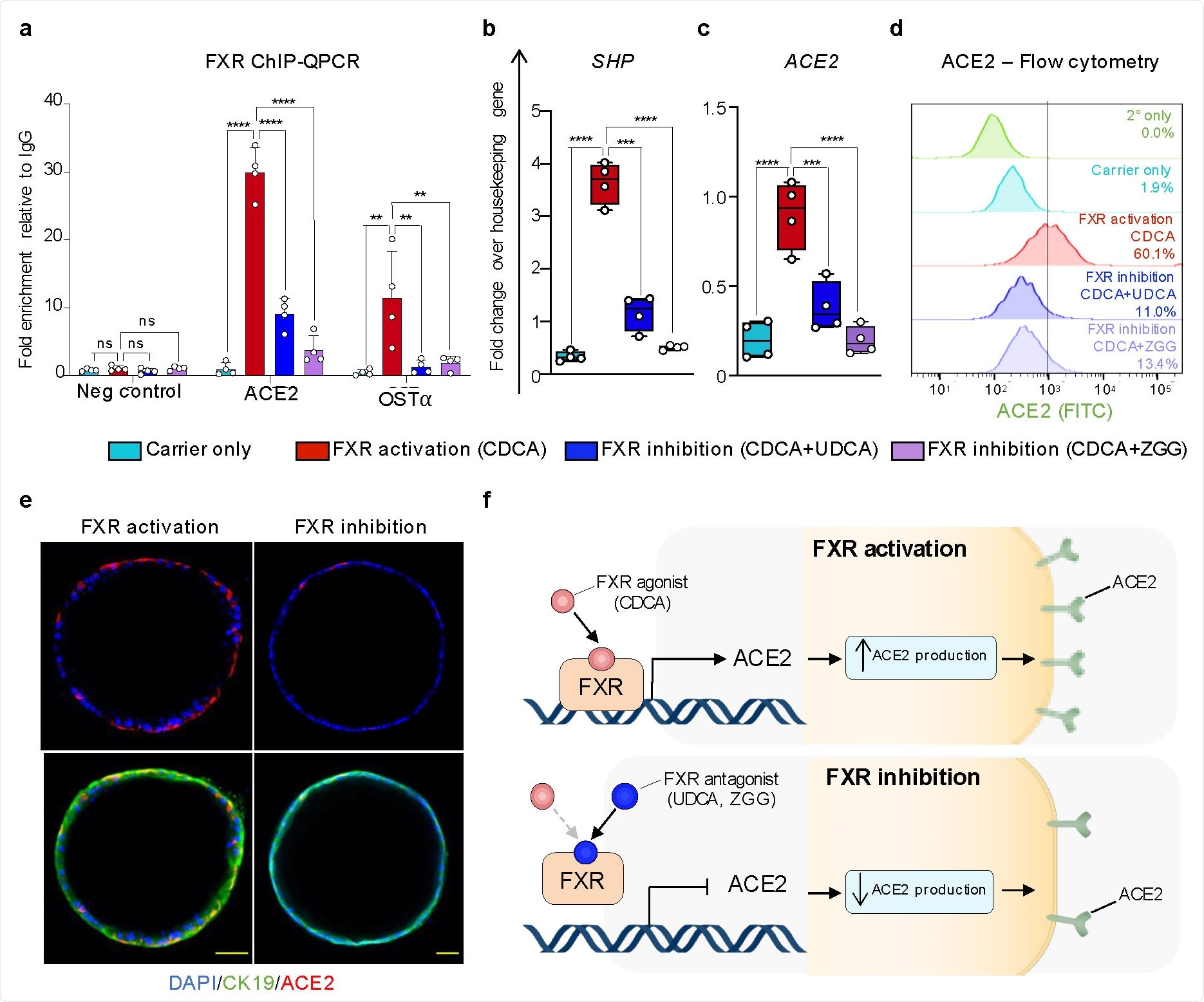
The researchers identified the bile acid farnesoid X receptor (FXR) as a direct regulator of ACE2 transcription in multiple tissues affected in COVID-19, including gastrointestinal and respiratory tissues.
They also showed that FXR antagonists downregulated ACE2 levels and reduced susceptibility to SARS-CoV-2 infection in lung, cholangiocyte and gut organoids. These compounds included an over-the-counter drug called z-guggulsterone (ZGG) and an off-patent drug called ursodeoxycholic acid (UDCA).
“We identify a novel function of FXR in controlling ACE2 expression and provide evidence that this approach could be beneficial for reducing SARS-CoV-2 infection,” says the team.
Sampaziotis and colleagues are calling for the findings to be validated in prospective double-blinded clinical trials.
A pre-print version of the research paper is available on the bioRxiv* server, while the article undergoes peer review.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Treatment options for established disease are limited
The mass rollout of vaccination to protect against SARS-CoV-2 infection represents the most promising approach to reducing transmission of the virus and combatting the COVID-19 pandemic.
However, the vaccines developed so far are not effective against established disease. They are limited by the emergence of vaccine-resistant viral variants and a lack of supplies in certain countries.
Treatment options for established disease are also limited, with currently used drugs such as dexamethasone and remdesivir only improving clinical outcomes in specific patient groups.
Host receptors for SARS-CoV-2 represent ideal therapeutic targets since they are essential for cellular entry and infection. Among these, ACE2 – the main receptor for SARS-CoV-2 – is particularly appealing.
“It directly binds the spike protein of different coronaviruses, with a high affinity for SARS-CoV-2, rendering it indispensable for viral entry,” says Sampaziotis and the team.
Accordingly, COVID-19 mainly affects ACE2-expressing tissues such as the lungs, the cardiovascular system, the digestive tract and the biliary tree.
“Modifying ACE2 expression could impede viral entry and protect against infection from SARS-CoV-2 and potentially other coronaviruses using the same receptor,” write the researchers.
Furthermore, since ACE2 is a host cell protein, its expression is not affected by mutations in the virus and therapies that modulate ACE2 expression may also be effective against SARS-CoV-2 variants.
What did the researchers do?
Using human cholangiocyte organoids as a proof-of-principle system, the researchers identified the bile acid receptor FXR as a direct regulator of ACE2 expression.
Expression of markers involved in SARS-CoV-2 entry in cholangiocytes. (a) Schematic illustration of different cholangiocyte populations corresponding to different areas of the biliary tree and cholangiocyte organoids (COs) derived from different areas of the biliary tree grown in absence or presence of the bile acids. (b) UMAP plot illustrating different cholangiocyte populations from (a) analysed by scRNAseq. (c) UMAP plots showing that viral entry related genes are predominantly expressed in extrahepatic cholangiocytes and COs treated with CDCA. (d) Immunofluorescence illustrating that ACE2 is predominantly expressed in extrahepatic cholangiocytes and CDCA11 treated gallbladder cholangiocyte organoids (GCOs). Scale bars 50 μm. (e) Violin plot
The team demonstrated that this mechanism applies in multiple COVID-19-affected tissues, including gastrointestinal and respiratory systems.
Next, Sampaziotis and colleagues showed that FXR antagonists downregulated the expression of ACE2 in multiple cell types. These antagonists included the over-the-counter phytosteroid ZGG and the clinically-approved, off-patent drug UDCA, which is used as a first-line treatment in primary biliary cholangitis.
The team found that administration of the FXR antagonists reduced susceptibility to SARS-CoV-2 infection in lung, cholangiocyte and gut organoids.
When the researchers repeated the experiments in human organs that were perfused ex-situ, they found that “systemic” UDCA administration in circulating blood reduced ACE2 expression and SARS-CoV-2 infection.
Furthermore, the team conducted a retrospective study of an international registry cohort of patients with COVID-19 and chronic liver disease that identified a correlation between UDCA treatment and positive clinical COVID-19 outcomes.
“A new potential clinical application for FXR inhibitors”
The team says the results illustrate the potential of ACE2 modulation as a novel host-directed therapy against COVID-19 and other coronaviruses.
“Our finding that FXR inhibition through UDCA or ZGG reduces ACE2 expression, and limits SARS-CoV-2 infection, identifies a new potential clinical application for FXR inhibitors,” writes Sampaziotis and colleagues.
The researchers point out that UDCA is easy to administer orally, well-tolerated, affordable and accessible to health systems worldwide, since it is off-patent.
“Nevertheless, our study is not a clinical trial and therefore, we cannot exclude the potential for confounding and selection biases,” they add.
“Consequently, it will be imperative to validate these results in prospective double-blinded clinical trials and fully assess the impact of this drug on ACE2 levels and SARS-CoV-2 infection,” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Sampaziotis F, et al. FXR inhibition reduces ACE2 expression, SARS-CoV-2 infection and may improve COVID-19 outcome. bioRxiv, 2021. doi: https://doi.org/10.1101/2021.06.06.446781, https://www.biorxiv.org/content/10.1101/2021.06.06.446781v1
- Peer reviewed and published scientific report.
Brevini, Teresa, Mailis Maes, Gwilym J. Webb, Binu V. John, Claudia D. Fuchs, Gustav Buescher, Lu Wang, et al. 2022. “FXR Inhibition May Protect from SARS-CoV-2 Infection by Reducing ACE2.” Nature, December. https://doi.org/10.1038/s41586-022-05594-0. https://www.nature.com/articles/s41586-022-05594-0.