Florida International University researchers have investigated the mutation-induced structural changes in the delta variant of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that could contribute to its immune evasion abilities.
The scientists have identified that certain alterations in the receptor-binding interface facilitate the delta variant escaping antibody-mediated neutralization. The study is currently available on the bioRxiv* preprint server.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Background
Among various genetic variants of SARS-CoV-2 identified so far, the B.1.617.2 variant, also called the delta variant, is considered to have the highest infectivity. Soon after its first identification in India, the delta variant sparked a second wave of devastating complications which has sapped the country's economy and healthcare system.
Given its rapid and exponential spread within and outside India, the delta variant has been designated as the Variant of Concern (VOC) by the World Health Organization.
Although currently available vaccines have shown neutralizing efficacy against the delta variant, evidence suggests that neutralizing antibodies induced by natural SARS-CoV-2 infection or vaccination are relatively less effective against the variant.
In the current study, the scientists have investigated how mutation-induced structural changes in the receptor-binding interface of the delta variant affect its binding to angiotensin-converting enzyme 2 (ACE2) and neutralizing antibodies.
Specifically, they have analyzed the antibody – RBD and ACE2 – RBD complexes available in the protein data bank and compared the differences in antibody/ACE2 – RBD interactions caused by mutations in the delta variant.
Important observations
The scientists conducted molecular dynamics simulations of the delta RBD and observed that both L452R and T478K mutations are present in the receptor-binding motif spanning the residues 438 to 508. Thus, this interface is a potential target of many neutralizing antibodies.
By analyzing the structural alterations in this interface, they observed the amino acid residues in the β-loop-β motif (residues 472 to 490) have the highest flexibility for major variants of SARS-CoV-2, including B.1.1.7, B.1.351, and B.1.617.2.
Further analysis identified a stronger hydrogen bonding between the β strands and a significantly increased salt-bridge interaction between the R454 and D467 side chains in the delta variant. The increased interaction between the β strands, which appeared to be due to the L452R mutation, is responsible for enhanced stability in the receptor-binding interface.
The T478K mutation in the delta variant lies in the β-loop-β motif, which contains a disulfide bond between the residues C480-C488. Interestingly, the scientists observed that by adopting a more stable and different conformation than other variants, the delta variant exhibited reduced flexibility in the β-loop-β motif.
Taken together, the scientists concluded that the changes in the β-loop-β motif and the β strands are collectively responsible for overall structural changes in the receptor-binding interface, which in turn is associated with a reduced interaction of the delta RBD with neutralizing antibodies.
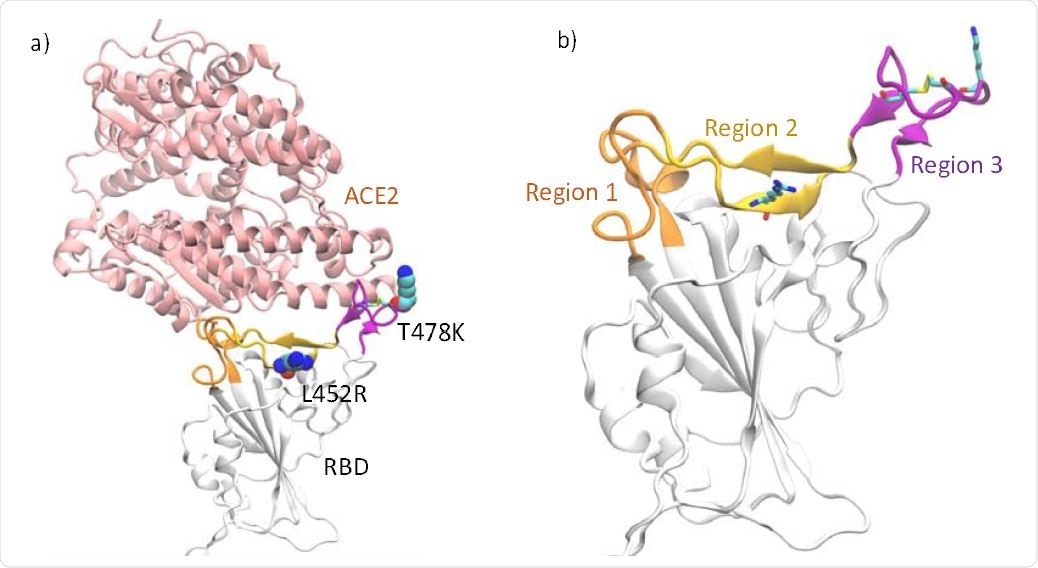
a) RBD complexed with ACE2. The locations of the mutations in the RBD of Delta variant are highlighted in VDW representation b) The loop segments consisting of residues 438- 447 and 499-508 (Region 1) are highlighted in orange, the β-sheet region consisting of residues 448-455 and 491-498 (Region 2) are highlighted in yellow and the receptor-binding loop consisting of residues 472−490 (Region 3) in purple. The disulfide bond in the loop as well as the mutations in the Delta variant are shown as sticks.
Interaction between delta RBD and neutralizing antibodies
By analyzing around 118 antibody–RBD complexes, the scientists observed that the majority of neutralizing antibodies interact with the β-loop-β residues Y473, A475, N487, and E484.
In their analysis,, they observed that for the delta variant, there was an increase in the distance between different antibody-binding sites in the delta receptor binding interface, which could be attributed to weakened antibody – delta RBD interaction and resistance of the delta variant to antibody-mediated neutralization.
Study significance
The study highlights how L452R/T478K RBD mutations are associated with structural changes in the ACE2- and antibody-binding interfaces in the delta variant and how these structural changes impact viral infectivity and immune evasion ability.
The study findings reveal that the receptor-binding β-loop-β motif in the delta variant adopts an altered but stable conformation that causes separation between some of the antibody-binding epitopes. Collectively, these changes can reduce the binding affinity of neutralizing antibodies for the delta variant.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources