Following the infection with SARS-CoV-2, a causative agent of the current COVID-19 pandemic, there is an activation of both innate and adaptive immune systems in our body. Thus, in many ways, the disease severity depends on how we respond to the infection.
Neutralizing antibodies that are generated are highly specific for viral epitopes predominantly found in the SARS-CoV-2 spike glycoprotein, but T cells are also recruited in order to secrete antiviral cytokines and kill infected cells.
If the early immune response of the host is unable to control and clear the virus, the disease marches toward a secondary phase characterized by an uncontrolled host inflammatory response, which is responsible for known severe manifestations and may finally result in organ damage.
This is why vaccines were the real game-changer in this pandemic. Even though we are only a little more than 18 months into the pandemic, four vaccines are already widely used around the world: Pfizer/BioNtech's BNT162b2, Moderna's mRNA-1273, Janssen's Ad26.COV2S and AstraZeneca's ChAdOx1.
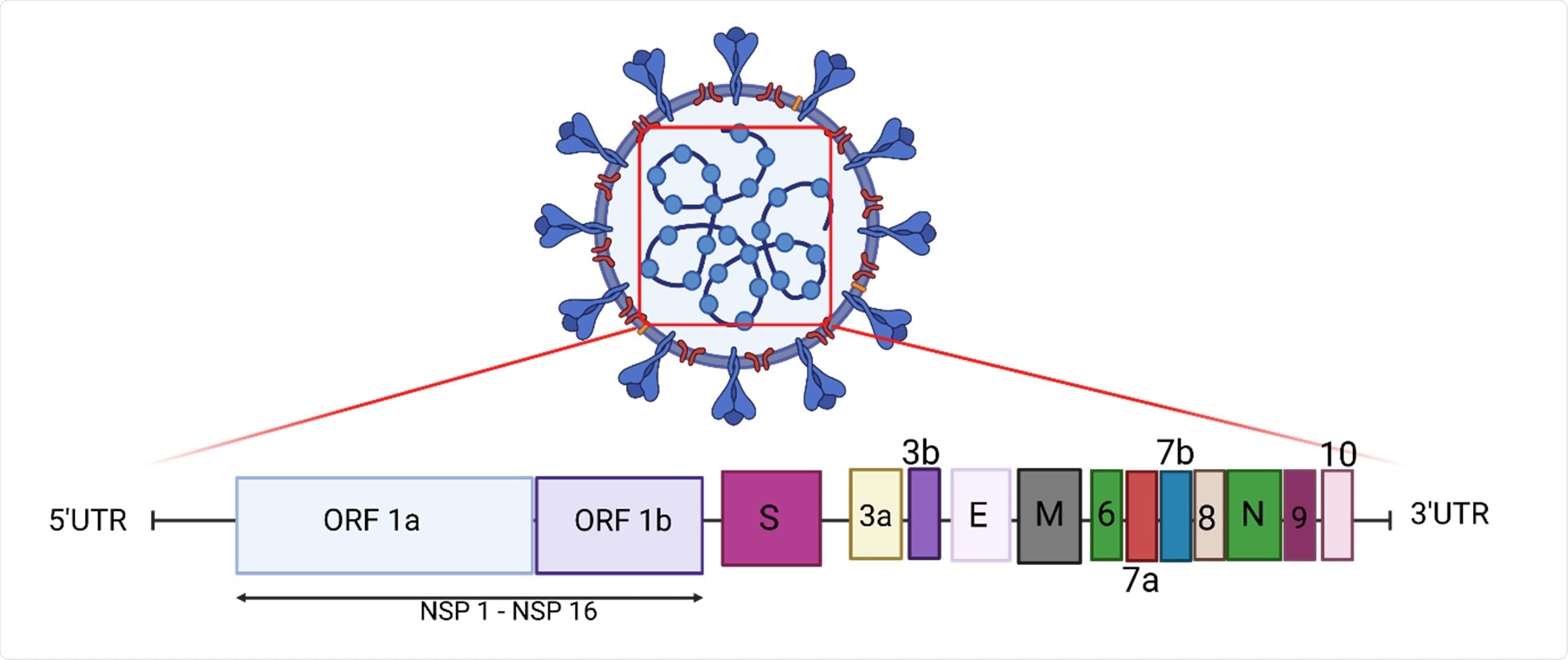
The genomic structure of SARS-CoV-2. The genome encodes two large open reading frames (ORFs), ORF1a and ORF1b, which encode 16 non-structural proteins (NSP1-NSP16). The structural genes encode the structural proteins, spike (S), envelope (E), membrane (M), and nucleocapsid (N), and the accessory genes (3a, 3b, 6, 7a, 7b, 8, 9 and 10) (Created with Biorender.com accessed on 9 September 2021).
A swift rise of viral variants
However, towards the end of 2020, there was already an emergence of several different SARS-CoV-2 variants of concern (as a result of mutational changes in the viral genome) that showed increased transmissibility and possible evasion of immune responses generated from prior SARS-CoV-2 infection.
Of note, variants of concern are those strains of SARS-CoV-2 that have higher transmissibility rates, more severe clinical presentations, lower response to treatment, or inadequate diagnostic yields with the use of established procedures and tests.
Likewise, these viral variants are also a threat to the efficacy of vaccines. As a result of the presence of mutations in spike glycoprotein (especially in neutralizing antibody epitopes), laboratory experiments have revealed a drop in the ability of post-vaccination serum samples to neutralize variants of concern.
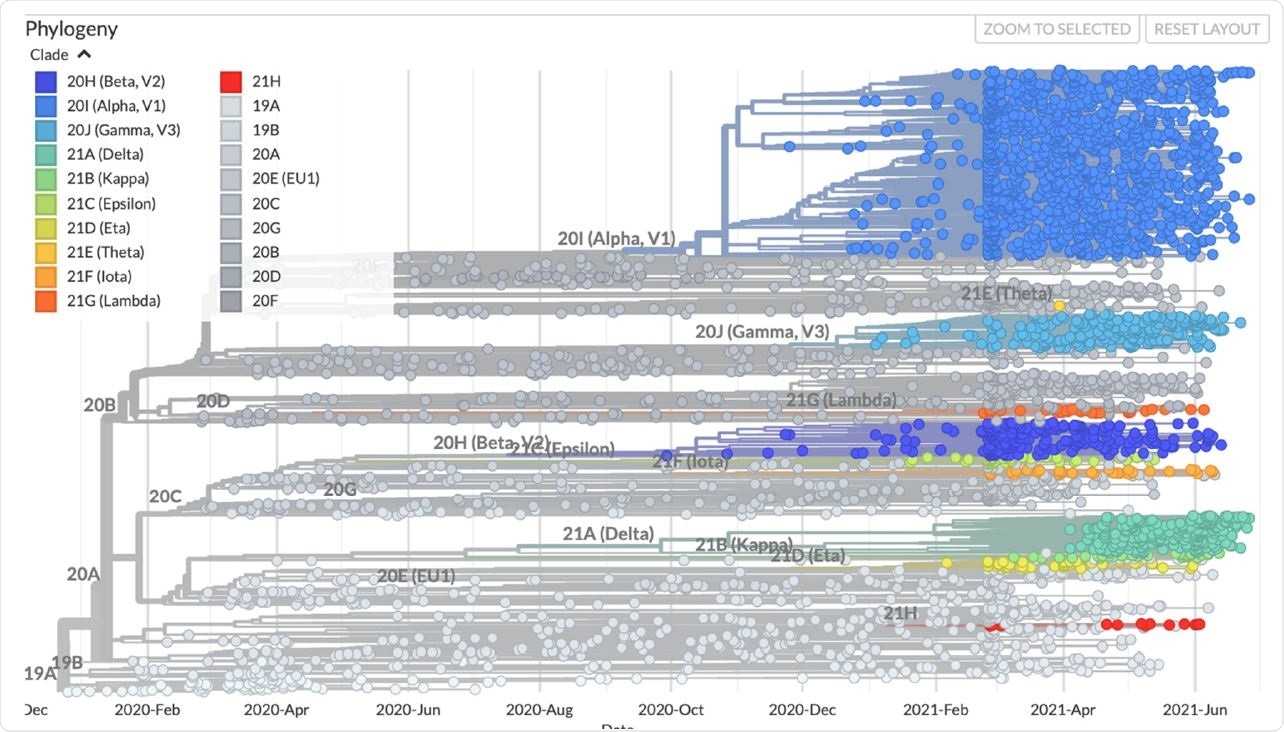
The emergence of new variants over time. The colors represent the ancestral clade. The variants of concern and variants of interest are shown on the graph. The image was taken from www.nextstrain.org under a CC-BY-4.0 license and is unchanged (accessed on 15 July 2021).
The possibility of reinfection
Even though variants of concern carry mutations that elude complete neutralization by the antibodies, and there are several reports of reinfections, it is still unclear whether the risk of reinfection is actually increased. Theoretically, the risk is there if spike glycoprotein changes, primarily due to its role in cell entry and because it is targeted by protective antibodies.
Some studies show that this may indeed be the case, but it depends on the variant in question. A study from the United Kingdom proved reinfection in 0.7% of tested individuals, but this did not correlate with the proportion of infections in those regions caused by the alpha variant – indicating a minor effect of this variant on the risk of reinfection.
On the other hand, a longitudinal serological study of unvaccinated repeat blood donors from Manaus in Brazil showed that, in the midst of the emergence of the gamma variant, a total of 16.9% of presumed infections had serological proof of prior infection with SARS-CoV-2.
Vaccines and variants of concern
While the effectiveness of vaccines at protecting against symptomatic infection with variants of concern may be decreased, everything points to the conclusion that they remain somewhat effective in preventing severe forms of COVID-19, hospitalization and/or death. It is also known that cellular immune responses remain largely preserved.
Nevertheless, at the moment, it is unclear whether variant-specific boosters will be necessary going forward. Such booster vaccines could either use the spike glycoprotein from the original SARS-CoV-2 Wuhan strain or the one derived from a variant of concern. Data from Israel demonstrate that a third shot during the widespread circulation of delta variant can definitely provide short-term protection against confirmed infection and severe disease.
However, it remains to be seen whether the spike protein will be able to mutate further to evade immune responses while at the same time preserving high levels of infectivity. Along with the kinetics of immune responses, this will ultimately determine whether updated vaccines or boosters will be required and at what frequency