In this interview, News-Medical talks to James O'Brien, Vice President at Leica Microsystems, about their latest development, the Mica microhub, and how this could revolutionize and advance the field of fluorescence microscopy.
Could you start by giving our readers a brief overview of the current microscopy landscape?
Microscopy and lab techniques, in general, have progressed a lot in the last 20 years. Customer requirements have prompted vendors to use to drive those techniques towards more reproducible and robust outcomes and usability.
Vendors need to make these techniques easier to use to drive and increase their adoption. When I was in university, you could specialize in your laboratory activities because there were not as many. You could become an expert in a particular technique as part of your research.
In today’s reality - especially when looking to publish - you cannot just leverage a single technique to drive your discovery. For example, if you’re looking at proteins, you’ll be asked to correlate your findings with transcriptomic information or information from another technique.
You are also getting asked to do this across more samples. You cannot publish on a rare event discovered in two instances - you need statistically valid reproducible evidence which requires a high number of events.
The reality of the lab has changed over the last 20 years, meaning that the reality for researchers has changed, the requirements to publish and get grants have changed, and all of this has driven the techniques forward.
This new reality that has pushed the majority of the lab environment has not forced microscopy to change in the same way. Microscopy has remained a technique where you have to be fairly experienced to get reproducible results and be efficient in collecting as much data as possible from as many samples as possible.
What is the Mica platform, and how did the project come about?
We introduce Mica, a fully integrated imaging system. The world's first Microhub!
Video credit: Leica Microsystems GmbH
By developing the Mica platform, we want to address this fundamental challenge of microscopy requiring significant expertise to achieve results.
With Mica, we have built a system based on embedded intellect and automation. We have provided a solution that eliminates the requirement for expertise turning the use of microscopy into a routinely done task: insert the sample and use a few clicks to acquire a high-quality image and useful spatial data.
This is very different from how things have historically been done in the world of microscopy. The systems are highly technical, require the user to set many parameters and optimize them through iteration to achieve high-quality results.
In the development of Mica, we prioritized the needs of principal investigators who focus on specific biological questions and require the extracted result in addition to the visual data. They are looking for something straightforward and easy to use. Mica provides that. In a few clicks, the system automates that expertise of optimizing parameters like the detector settings, pinhole size, gains, etc., using algorithms and software rather than the user setting these manually.
Has there been any resistance to this very different approach and way of working?
We have seen some resistance, partly because Mica could be seen as a threat to their expertise. For instance, in a core imaging facility, where it is essentially a service business providing imaging expertise, there was initially some pushback from some of the KOLs we spoke to.
Ultimately, when we did some introductions to the instrument, they saw the value for them as well. They spend a lot of time on training, retraining and troubleshooting in their laboratory, all with limited resources. They saw an opportunity to use a Mica to handle more routine work, allowing their experts to focus on the more advanced technologies which require their expertise.
From an adoption standpoint, a couple of wider factors have worked in Mica’s favor. One is that the acceptance of digital tools has been increasing. Artificial intelligence was frowned upon for use in image analysis some years ago, for example, because it is often seen as a black box.
We see now that this barrier has reduced, and our Avia product, which is using machine learning for image analysis, is seeing excellent uptake. We have incorporated some of the Aivia technology in Mica.
The other factor is that one of the core technological advances in Mica, hyperspectral unmixing, is known in the community. Hyperspectral unmixing - an algorithm-based approach to separating light comes out of the lab of Dr. Scott Fraser.
He has published a great deal on this, people understand it, and it is widely accepted. We partnered with Dr. Fraser to leverage his technology, optimize it and industrialize it into a product available to the broader community.
Beyond its ease of use and accessibility, are there other reasons Mica is such a game-changer?
There are also some technical aspects; for example, the system offers both widefield and confocal capabilities. Other systems on the market feature both of these technologies, but in Mica, we have fully integrated them.
When you look at a sample in widefield, you can see more of the sample. With Mica, you can be in wide-field mode, find something of interest, and with one click, the view is switched to a confocal mode to look at this in much more depth or a 3D view.
 Image credit: Leica Microsystems GmbH
Image credit: Leica Microsystems GmbH
What are the advantages of Mica over current alternative platforms?
Technically the system is up to four times faster than a conventional system. In terms of what this can mean for a customer, scanning a slide at high resolution on a conventional system could take up to 8 hours because you have to scan sequentially.
For example, in conventional microscopy, you would look at one color, take an image, move to the next color, and then perform the same iterative step.
With Mica’s FluoSync technology using hyperspectral unmixing, we can capture almost the entire spectrum of light and all information in one shot, and then using algorithms, we separate that into the four components. This makes the system up to four times faster than any conventional microscope.
By collecting all this information at once, we do not experience any lag in the temporal correlation. For example, when imaging a biological process that moves very quickly, it is easy to miss important events, creating uncertainty or causing the data to be no longer directly correlated as things shift in that time period.
Data captured via Mica is 100% correlated spatially and temporarily, giving us more confidence in the data and removing variability.
As we built the system from the ground up, and it is fully self-contained, we have built it as an incubator. That means we can run live-cell experiments for very long durations while maintaining optimal conditions to keep the cells healthy.
What is fluorescence microscopy, and how is it used?
Fluorescence microscopy involves, of course, magnifying what you are trying to observe, but you are also specifically labeling a target. This target is often a protein, and we would use an antibody conjugated to a fluorophore that attaches that to the analyte of interest.
We then excite that fluorophore, causing it to emit a light which we capture using a sensor.
Often you need to get more than one protein of interest out of a sample, so we apply different fluorophores to get different points on the spectrum. This is why we often see iterative processes in microscopy because many users look at one protein using one fluorophore and then move to the next.
One thing that is very different with Mica is that we do not talk about capturing four colors; we talk about capturing four labels. Generally, in a microscope, you would filter the light you are looking at to know which fluorophore to assign it to.
But the reality is when you excite the fluorophores, you are not just exciting one; you are often exciting a window of wavelengths.
For example, in the case of red light emitted from a fluorophore, part of this may actually come from the yellow because it shows up in the spectrum window. Researchers try to spread things out as far as possible to minimize the influence of other fluorophores. In conventional microscopy, this is just something that everybody accepts they have to deal with
This is not an issue in Mica because we do not look at a window in the spectrum - we take all the light emitted from a fluorophore and calculate this into a labeled protein. This means that we have the real response for every label we look at, and we can accurately quantify that response. That is very different from conventional microscopy approaches.
How will these changes impact the consumer?
Ultimately, the goal of researchers is to drive the discovery flywheel of making discoveries, publishing and attracting more funding. With Mica, we want to help them bring spatial context and spatial data into their research to help push this research forward via faster insights and faster access to the data.
We have also brought in analysis capabilities, meaning users can easily capture an image and start looking at a histogram. We are trying to shorten that cycle to progress their research faster, ultimately to their endpoint of publication.
For grant applications, you need uniqueness. Using a unique technology that can lead you to new discoveries can make a difference in a grant application With Mica, and at Leica, we strive to be a driving advantage for researchers to win funds and advance science.
What is it that takes the importance of this development beyond the field of microscopy?
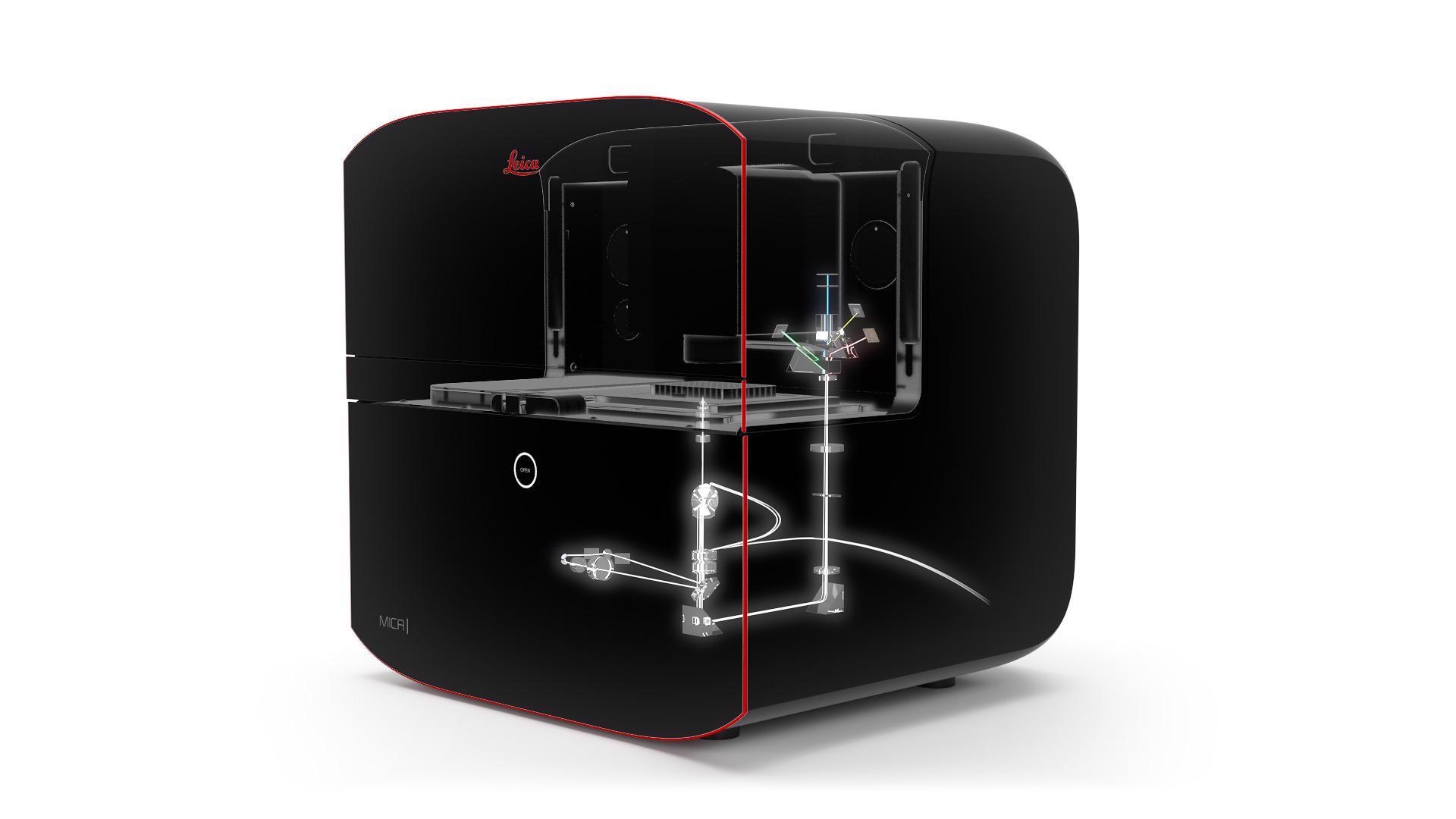
Image credit: Leica Microsystems GmbH
We avoided referring to Mica directly as a microscope, instead opting for the category of Microhub because we believe this is a fundamental change in how microscopy is utilized and perceived in the laboratory.
To call it a microscope does not do justice to what we have been able to bring into the system. This is a new approach to collecting spatial information, and in the end, that is what our customers are looking for.
We expect this piece of equipment to be universally adopted into laboratories, like a centrifuge or flow cytometer that is common across all lab environments. Our goal is to make this the standard laboratory device for collecting spatial data.
Due to its breadth of capabilities, we can shift a lot of experiments from different devices into one system. From a live cell, confocal and wide-field technology, and analysis capabilities, we are bringing together a lot of points related to microscopy into one specific device.
It is also a system that guides customers through the process to achieve results, instead of just being a tool that requires them also to find a way to their destination. Mica has reduced the number of steps in the routine process - 85% reduction on the front and 60% overall in the whole process.
It is so much more than a microscope – that is why we call it a Microhub.
About James O'Brien
James O'Brien is the Vice President of Life Sciences at Leica Microsystems. James O’Brien joined Leica Microsystems in 2019 as Vice President Applied Microscopy and took over the Life Science Research Business Unit in June 2020.
James joined Leica from Tecan where over 16 years he progressed his career from a field-based applications role, service leadership, sales operations to leadership of a variety of Tecan’s end customer businesses. Most recently James served as Vice President of Tecan’s Labwerx business. While working in global roles, James has lived in the US and Europe.
James’ educational background is in biology and his career began at Johns Hopkins University in genomic research. He also holds an MBA from Kenan-Flagler Business School with Honors.

About Leica Microsystems

Leica Microsystems, Inc., located in Buffalo Grove, IL, is the US selling unit headquarters for the Microsystems Division. With approximately 300 office and field-based employees, Leica also has a wide network of regional dealers and regional offices poised to serve customers across the United States and Canada.