Brain injury patients undergo several interventions to attain normal (TBr) values. The cranial neuronal sensitivity to temperature variations implies that the brain must have isothermal temperatures. However, findings from previous studies conducted on animals and humans have indicated substantial spatiotemporal variations in human TBr values.
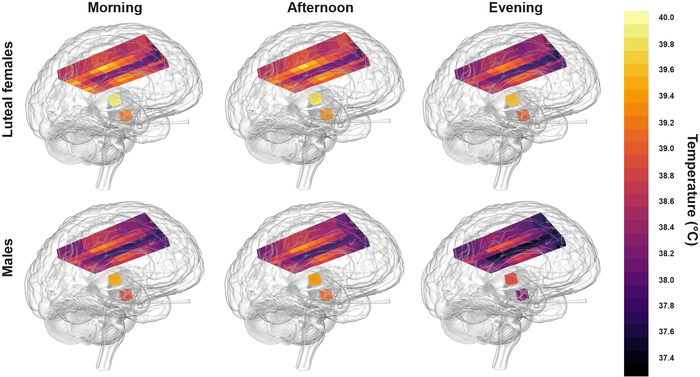
Healthy human brain temperature extremes showing relatively hot temperatures in post-ovulation females in the morning compared with coldest temperatures in males at night. Lighter yellow represents hotter temperatures. CREDIT N Rzechorzek/MRC LMB/Brain
About the study
In the present study, researchers assessed the spatiotemporal variations in TBr rhythms and assessed whether the daily mapping of TBr rhythmicity could predict survival outcomes of TBI patients.
Data were obtained from the intensive care unit (ICU) of Western General Hospital, Edinburgh, United Kingdom (UK), and from the collaborative European neurotrauma effectiveness research in the TBI (CENTER-TBI) database. Data were analyzed only for those TBI patients for whom direct TBr measurements were available and who were not on targeted temperature management (TTM) therapies.
In addition, 40 healthy adults (20 men and 20 women) aged 20 to 40 years were prospectively enrolled for cranial thermometry analysis by magnetic resonance spectroscopy (MRS). MR imaging (MRI) scans were obtained from the patients during the morning (between 9:00 am and 10:00 am), afternoon (between 4:00 pm and 5:00 pm), and night (between 11:00 pm and midnight) hours.
On the day of MRI scanning, subjects were permitted to consume food between 6:00 am and 8:00 am, between 12:00 pm and 2:00 pm, and between 6:00 pm and 8:00 pm. Consumption of caffeinated items was permitted between 6:00 and 8:00 am and between 12:00 pm and 2:00 pm. Alcohol use was not allowed at any time. In addition, the subjects were asked to refrain from excessive physical activities on the MRI scanning day.
TBr values were obtained using a thermistor which was inserted into the brain parenchyma and white matter of the frontal lobe at a depth of 18 mm beneath the dura mater. The body temperature (TBo) values were primarily obtained by measuring tympanic, bladder, external axillary, and nasopharyngeal temperatures. Secondary TBo sites were the skin, esophagus, and rectum. Data obtained during the initial two hours of temperature monitoring were excluded to ensure no influence of the electrode stabilization time on the study results.
The study's prime objective was to determine healthy TBr variations at different times of the day. The secondary study objectives comprised an assessment of time-based and sex-based TBr variations, a comparison of TBr and TBo variability, and exploring time-dependent TBr changes in different areas of the brain. The team also investigated if TBr mapping could predict TBI patient survival.
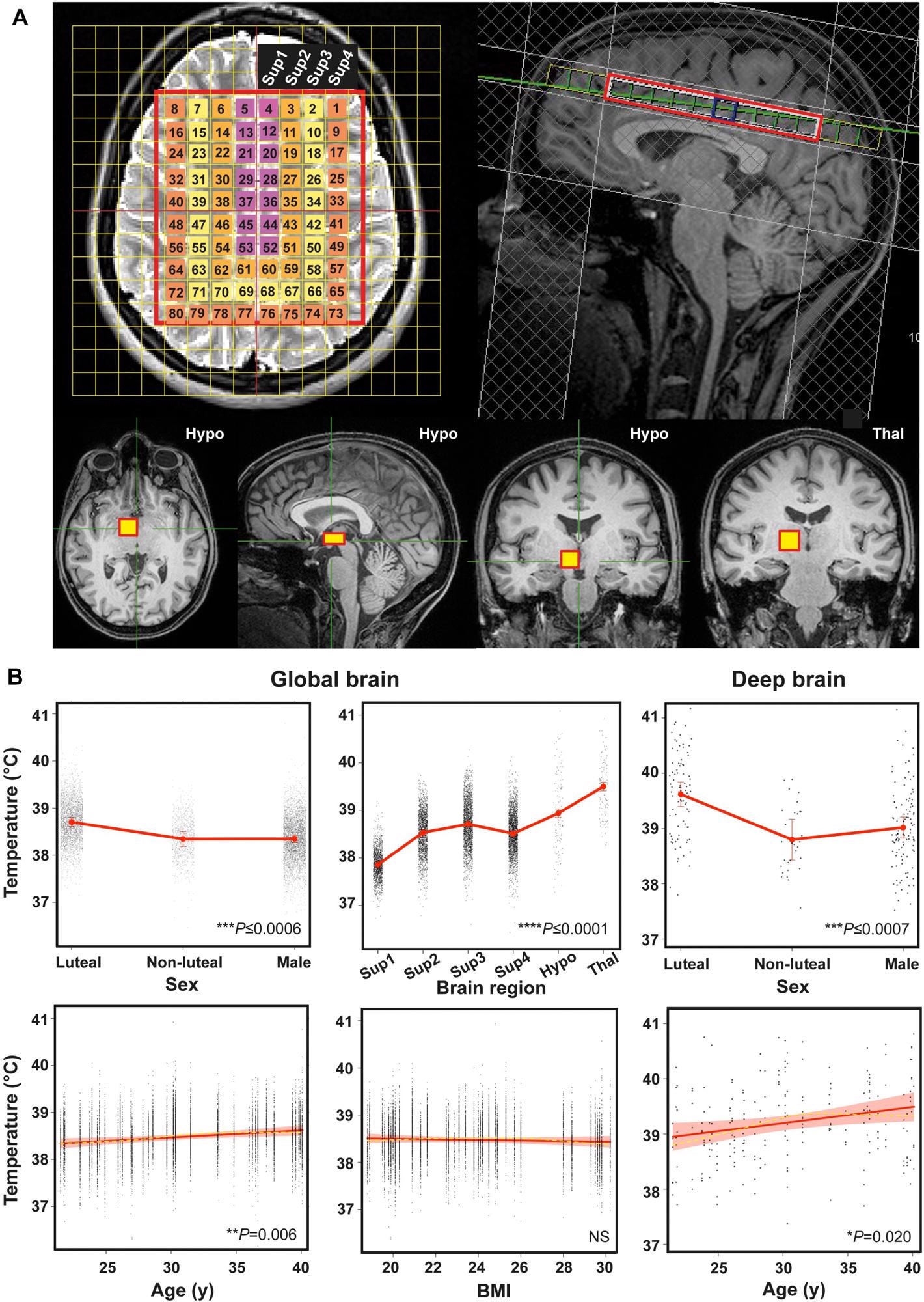 Human brain temperature is spatially heterogenous. (A) Representative annotated MR images to show MRS extraction protocol immediately after whole-brain structural acquisition. T2-weighted axial (top left) and T1-weighted mid-sagittal (top right) image showing multivoxel MRS overlay for more superficial brain regions including cerebral grey and white matter; note positioning superior to corpus callosum. From this multivoxel acquisition, MRS data was extracted from each of the numbered voxels individually; for the final statistical model, the whole cerebral region was split into four superficial groups of voxels (Sup 1–4, depicted as separate colours in the overlay, from medial to lateral). T1-weighted axial, sagittal and coronal images (bottom three images from left side, respectively) showing orthogonal positioning of single voxel in right hypothalamus (yellow box). T1-weighted coronal image (bottom right) showing positioning of single MRS voxel in right thalamus (yellow box). (B) Linear mixed modelling results for global TBr by sex, age, brain region and BMI, and for deep TBr (including thalamus and hypothalamus) by sex and age. Solid red lines represent model fits, shaded areas and double-ended error bars represent 95% CIs, dark grey circles display residuals (single temperature data-points) and smoothed dashed yellow lines represent partial residuals. For sex, P-value reflects comparisons of each group with luteal females. For brain region, P-value represents comparisons of each region relative to superficial region 1 (parasagittal group of voxels). Sup 1–4 = superficial brain regions 1–4 from medial to lateral; Hypo = hypothalamus; Thal = thalamus.
Human brain temperature is spatially heterogenous. (A) Representative annotated MR images to show MRS extraction protocol immediately after whole-brain structural acquisition. T2-weighted axial (top left) and T1-weighted mid-sagittal (top right) image showing multivoxel MRS overlay for more superficial brain regions including cerebral grey and white matter; note positioning superior to corpus callosum. From this multivoxel acquisition, MRS data was extracted from each of the numbered voxels individually; for the final statistical model, the whole cerebral region was split into four superficial groups of voxels (Sup 1–4, depicted as separate colours in the overlay, from medial to lateral). T1-weighted axial, sagittal and coronal images (bottom three images from left side, respectively) showing orthogonal positioning of single voxel in right hypothalamus (yellow box). T1-weighted coronal image (bottom right) showing positioning of single MRS voxel in right thalamus (yellow box). (B) Linear mixed modelling results for global TBr by sex, age, brain region and BMI, and for deep TBr (including thalamus and hypothalamus) by sex and age. Solid red lines represent model fits, shaded areas and double-ended error bars represent 95% CIs, dark grey circles display residuals (single temperature data-points) and smoothed dashed yellow lines represent partial residuals. For sex, P-value reflects comparisons of each group with luteal females. For brain region, P-value represents comparisons of each region relative to superficial region 1 (parasagittal group of voxels). Sup 1–4 = superficial brain regions 1–4 from medial to lateral; Hypo = hypothalamus; Thal = thalamus.
Results
Out of 134 records meeting the eligibility criteria were screened, 114 records had a 24-hour TBr record (criteria A), of which 110 individuals had adequate (≥36 hours) TBr data for analysis of daily temperature rhythmicity (criteria A and B). Out of these 110 patients, 10 patients had adequate data for only TBo. Data on ICU outcomes were available for 113 out of 114 patients (criteria A and C) and 109 patients had ≥1 TBI severity scores such as Glasgow Coma Scale, pupillary light reflex, or Glasgow Coma Scale motor response (criteria A and D). In total, 105 patients met all the study criteria.
The mean TBr values of the study participants ranged between 32.6°C and 42°C, with the reported average TBr value (38.5 C ± 0.8°C) higher than the normal TBo (37.5°C ± 0.5°C). Out of the study of 100 participants who met the criteria for analyzing TBr rhythms, 25 participants exhibited daily TBr rhythmicity, and the TBr range was lower for older individuals.
Among healthy individuals, TBr values ranged between 36.1°C and 40.9°C, and the average TBr value (38.5°C ± 0.4°C) was higher than the oral (mouth) temperature reported (36°C ± 0.5°C) with significantly higher values among luteal women compared to follicular women and men (mean difference of 0.4°C) The TBr values increased with advancing age of individuals (0.6°C for a 20-year increase in age), with the most notable differences observed in the deep region of the brain and showed a spatial variation of 2.4°C ± 0.5°C.
The lowest TBr values were found in the cortical grey matter regions lying close to the brain surface and adjacent to a major venous drainage channel. The highest TBr values were reported in the thalamus (1.64°C higher temperatures than cortical grey matter and 0.56°C higher than the hypothalamus). The hypothalamus displayed the most significant temporal variation (1.2°C ± 0.65°C).
Further, TBr values also showed diurnal variations, with the lowest TBr values (and highest CBF) during the night and the most notable differences were observed in the deep areas of the brain (0.9°C). Based on the data from healthy individuals, HEATWAVE was generated which provides a four-dimensional (4D) mapping of human TBr values.
When the clinical value of the HEATWAVE generated was ascertained, it was found that if the daily TBr rhythms could not be mapped, the likelihood of ICU deaths would be 21-fold higher, and that absolute maximum and minimum TBr values did not significantly benefit in estimating survival outcomes. A warmer brain or higher TBr values were associated with improved patient survival and a 19-year increase in age elevated the chances of death by 11-fold.
Overall, the study findings highlighted the variations in TBr rhymes with sex, age, menstruation, time of the day, and region of the brain in humans and showed daily TBr rhythmicity (not absolute TBr values) could be a strong and independent predictor of survival of TBI patients.
Dr John O’Neill, Group Leader at the MRC Laboratory for Molecular Biology, said: “To me, the most surprising finding from our study is that the healthy human brain can reach temperatures that would be diagnosed as fever anywhere else in the body. Such high temperatures have been measured in people with brain injuries in the past, but had been assumed to result from the injury.” “We found that brain temperature drops at night before you go to sleep and rises during the day. There is good reason to believe this daily variation is associated with long-term brain health—something we hope to investigate next.”
Journal reference:
- Nina M Rzechorzek, Michael J Thrippleton, Francesca M Chappell, Grant Mair, Ari Ercole, Manuel Cabeleira, The CENTER-TBI High Resolution ICU (HR ICU) Sub-Study Participants and Investigators, Jonathan Rhodes, Ian Marshall, John S O’Neill, A daily temperature rhythm in the human brain predicts survival after brain injury, Brain, 2022; DOI: https://doi.org/10.1093/brain/awab466, https://academic.oup.com/brain/advance-article/doi/10.1093/brain/awab466/6604351?login=false