Introduction
Generally, in protein and peptide analyses, knowing a molecule’s oligomeric state is useful in determining whether the protein exists in its biologically active form. To this end, the first step usually involves separating and characterizing the molecular species using size exclusion chromatography (SEC). SEC isolates the molecules according to their hydrodynamic volume. While hydrodynamic volume is mostly proportional to molar mass, analytical SEC does not give the precise mass determination. However, this can be overcome by combining SEC with light scattering, which enables the oligomeric state of the eluting macromolecules to be fully characterized. This article describes the effect of sample concentration and buffer composition on the oligomers of insulin detected by SEC in combination with multi-angle light scattering (SEC-MALS).
Multi-angle Light Scattering
Multi-angle light scattering (MALS) is one of the main methods used to measure the molar mass of macromolecules in solution. This method does not make any assumptions about the structure of the molecule and eliminates the need for molar mass standards. Previously, studies have demonstrated the separation of oligomers using SEC and their detection has been achieved using light scattering methods. In addition, composition-gradient MALS (CG-MALS) measurements have been used to ascertain the stochiometry and affinity of insulin association.
Experimental Framework
SEC was carried out using the Agilent 1260 HPLC system equipped with a degasser, an auto sampler and an isocratic pump. The separation column used was the Superose 12 300/10 from GE Healthcare. A DAWN® HELEOS® 18 angle light scattering detector featuring a WyattQELS™ for online DLS detection, an Agilent variable wave-length detector at 280 nm and an Optilab® rEX refractive index detector were used for detection as shown in Figure 1. ASTRA® software from Wyatt Technology was used for the SEC-MALS analysis.

Figure 1. Instrumentation for the analysis.
The mobile phase included 2 mM phenol, 10 mM Tris, 140 mM NaCl and 200 ppm NaN3 at pH 7.7. Three insulin preparations containing 0.6 mM insulin were examined. Sample 1 was an insulin analogue that did not contain zinc; Sample 2 was human insulin with 0.1 mM zinc; and Sample 3 was human insulin with 0.3 mM zinc.
Results and Discussion
Insulin molecules tend to form dimers and other oligomers in the absence of zinc. These oligomers can be separated by SEC and characterized using light scattering. The results of Sample 1 with molar masses specified throughout the chromatogram is shown in Figure 2. The major fraction has a molecular weight of 5.8 kDa and corresponds to the insulin monomer. This species is well resolved from the minor fraction corresponding to a hexamer with a molar mass of ~35 kDa. In addition, the light scattering signal suggests that the molar mass for the secondary peak reflects a set of oligomers ranging from monomer to hexamer — a conclusion that could not be drawn using the UV signal alone. Since light scattering is very sensitive to high molecular-weight species, MALS was able to detect the hexamer easily despite only minimal UV signal being present.
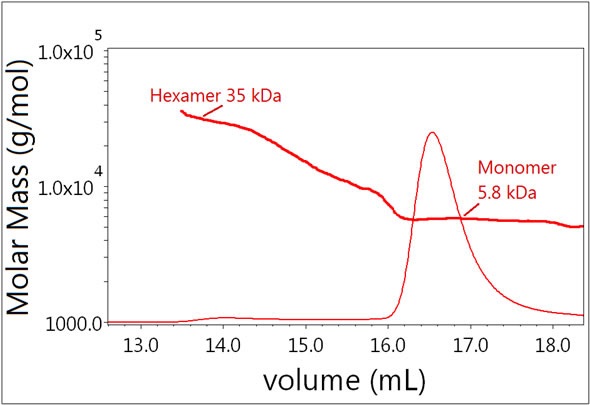
Figure 2. Plot of molar mass vs. time of Sample 1. The UV signal at 280 nm is plotted as an overlay. The molar masses were calculated using dRI and LS.
On the contrary, the majority of Sample 2 exists as a hexamer (Figure 3, blue) and a considerable fraction seems to be undergoing monomer-dimer equilibrium. This lower molecular weight peak elutes a little faster when compared to the monomer peak for Sample 1 and the quantified molar mass is heterogeneous across the peak, spanning the masses of the monomer and dimer (~6-12 kDa).
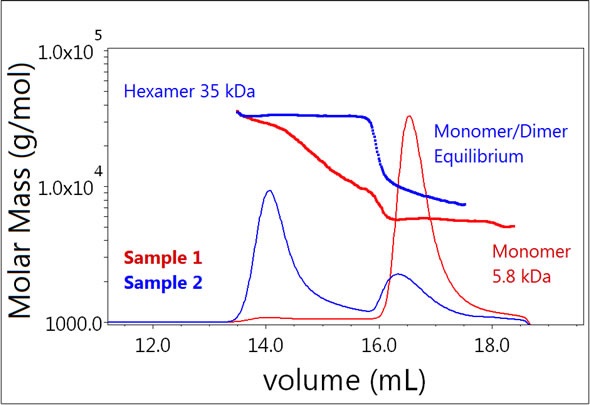
Figure 3. Plot of molar mass vs. time of Sample 1 and 2 (monomer, dimer, hexamer). The UV signal at 280 nm is plotted as an overlay. The molar masses were calculated using dRI and LS.
On the addition of zinc, the equilibrium is completely shifted towards the hexamer as observed in Sample 3 (Figure 4). Furthermore, the results demonstrated the presence of another molecular species detected as the dodecamer of the insulin molecule in Sample 3. Although this peak is present in trace amounts, it generated a clear signal that was instantly observed by the LS detector.
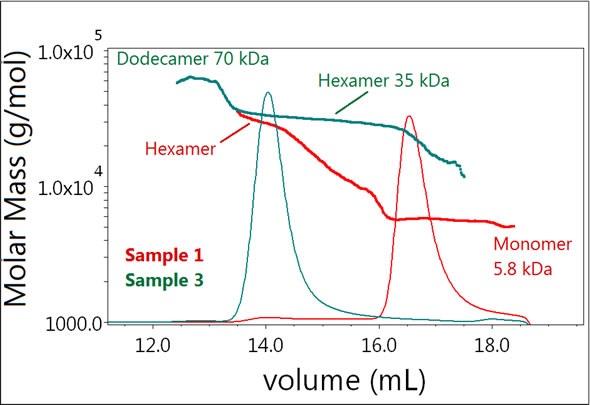
Figure 4. Plot of molar mass vs. time of Samples 1 and 3. The UV signal at 280 nm is plotted as an overlay. The molar masses were calculated using dRI and LS.
SEC-MALS enables the strength of a reversible self-association to be assessed. When the volume injected on the column is increased, the overall protein concentration also increases as it moves through the detectors, which shifts the equilibrium of the eluent toward the oligomeric complexes. In Samples 2 and 3, any changes in equilibrium association were examined by injecting 50 µL or 200 μL of the same sample solution onto the SEC column.
The equilibrium of Sample 2 (Figure 5) is clearly affected by the increased injection volume. As the insulin concentration increases, the monomer-dimer equilibrium is shifted towards dimer and higher order oligomers. For the 50 μL injection, the lowest measured molar mass was ~6 kDa, consistent with the monomer. For the 200 µL injection, however, the smallest measured molar mass was consistent with the dimer. The hexamer fraction is not affected by the increase in eluting concentration and does not show any tendency to form larger oligomers.
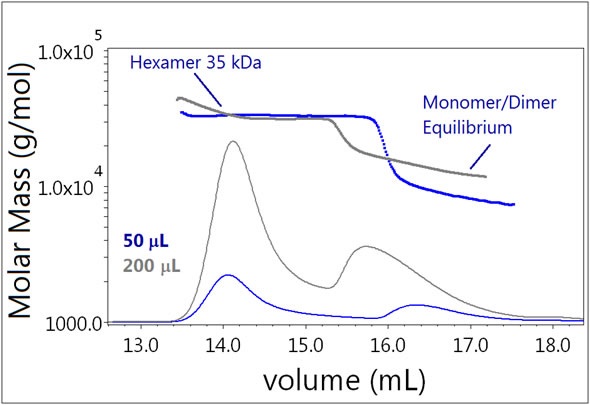
Figure 5. Plot of molar mass vs. time of Sample 3. The dRI signal is plotted as an overlay. The UV signal is saturated due to the high sample load. The molar masses were calculated using dRI and LS data.
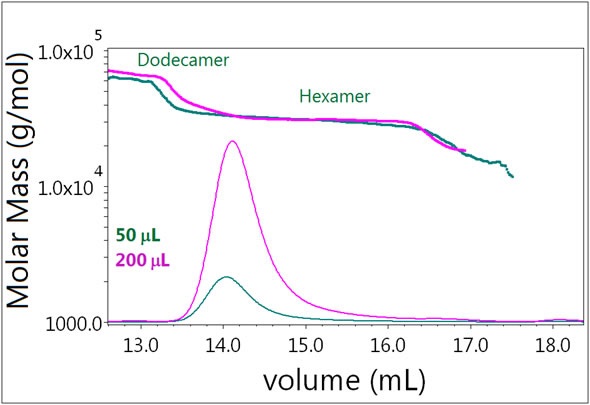
Figure 6. Plot of molar mass vs. time of Sample 3. The dRI signal is plotted as an overlay. The UV signal is saturated due to the high sample load. The molar masses were calculated using dRI and LS data.
In Sample 3, changing the injection volume has no effect, as shown in Figure 6. In the presence of 0.3 mM zinc, the system remains locked in its hexamer form and the same molar masses are ascertained at both concentrations. The mass fraction of dodecamer also does not increase. Evidently, the self-association behavior of the hexamer differs significantly from that of the monomer.
Conclusion
The above results clearly demonstrate the ability of MALS in examining the association phenomena for large proteins such as antibodies as well as for peptide molecules having lower molecular masses. As the therapeutic use of proteins and peptides becomes increasingly more important, so will light scattering as a technique for molecular characterization.
References
Adapted with permission from “Identification of Insulin Oligomeric States using SEC-MALS”, kindly submitted to Wyatt Technology Corp. by P. Wahlund et al., Novo Nordisk A/S, Måløv, Denmark. Graphs and illustrations reprinted with permission from Wyatt Technology.
- M. H. Jensen, P.-O. Wahlund, J. K. Jacobsen, B. Vestergaard, M. van de Weert, S. Havelund (2011). Self-association of long-acting insulin analogues studied by size exclusion chromatography coupled to multi-angle light scattering. Journal of Chromatography B, 879(28), 2945-2951.
- Jonassen, S. Havelund, T. Hoeg-Jensen, D. B. Steensgaard, P.-O. Wahlund, U. Ribel (2012). Design of the Novel Protraction Mechanism of Insulin Degludec, an Ultra-long-Acting Basal Insulin. Pharmaceutical Research, 29(8), 2104-2114.
- Arun K. Attri, Cristina Fernández, Allen P. Minton (2010). pH-dependent self-association of zinc-free insulin characterized by concentration-gradient static light scattering. Biophysical Chemistry, 148, 28-33.
- Arun K. Attri, Cristina Fernández, Allen P. Minton (2010). Self-association of Zn-insulin at neutral pH: Investigation by concentration gradient-static and dynamic light scattering. Biophysical Journal, 148, 23-27.
About Wyatt
 With a long history of excellence in scientific instrumentation, Wyatt Technology is the recognized leader in innovative light scattering instruments, accessories, software and services for determining the properties of macromolecules and nanoparticles in solution. Wyatt provides cutting-edge solutions for in-line multi-angle static light scattering (SEC-MALS), field-flow fractionation (FFF-MALS), composition gradients (CG-MALS), high-throughput and traditional dynamic light scattering (DLS), electrophoretic mobility via phase-analysis light scattering (MP-PALS), differential refractometry and differential viscosity. With a staff composed of 20% Ph.D. scientists and many more dedicated and experienced support personnel, Wyatt's aim is to delight the customer with the best products, training, customer support and service available in the industry.
With a long history of excellence in scientific instrumentation, Wyatt Technology is the recognized leader in innovative light scattering instruments, accessories, software and services for determining the properties of macromolecules and nanoparticles in solution. Wyatt provides cutting-edge solutions for in-line multi-angle static light scattering (SEC-MALS), field-flow fractionation (FFF-MALS), composition gradients (CG-MALS), high-throughput and traditional dynamic light scattering (DLS), electrophoretic mobility via phase-analysis light scattering (MP-PALS), differential refractometry and differential viscosity. With a staff composed of 20% Ph.D. scientists and many more dedicated and experienced support personnel, Wyatt's aim is to delight the customer with the best products, training, customer support and service available in the industry.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.