Introduction
Hemoglobin is a major protein component of mammalian red blood cells that plays a vital role in transporting oxygen from the lungs around the body. Following the release of oxygen, carbon dioxide attaches to the hemoglobin and is then removed from the body.
Instrumentation
Composed of four polypeptides, the tetramer human hemoglobin equilibrates with a dimer form, depending on the conditions. Using size exclusion chromatography in combination with multi-angle light scattering (SEC-MALS), a miniDAWN™ detector and an Optilab® differential refractometer readily characterize haemoglobin by obtaining data on absolute molar masses and molar mass distributions.
Analysis and Results
The molar mass of hemoglobin is plotted against the elution time in Figure 1. The molar mass across the peak is not constant, which may be due to an equilibrium shift from a tetramer to a dimer occurring during the chromatography run.
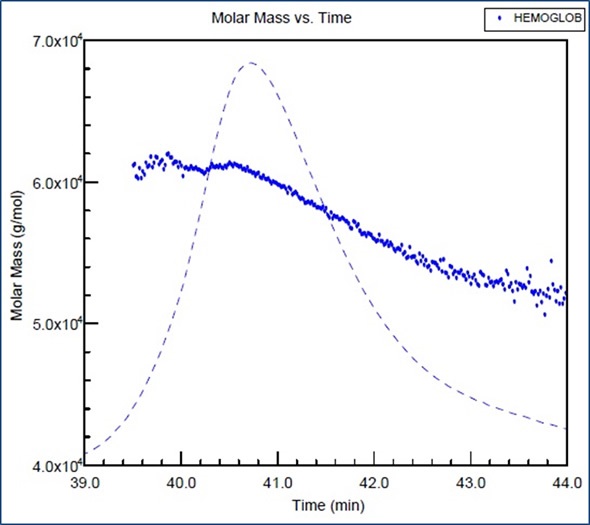
Figure 1. Molar mass versus elution time of hemoglobin obtained from SEC using two columns and combined with MALS detection.
It may prove very challenging to distinguish the regular peak stretching from the tiny polydispersity of the sample using traditional column calibration. However, this issue can be overcome using SEC-MALS measurements.
A standard protein, bovine serum albumin (BSA), is often used for column calibration. The molar mass versus elution time is shown for hemoglobin and BSA in Figure 2. Although the molar masses of the two proteins are nearly in the same range, their shapes and elution times are entirely different. BSA elutes earlier than hemoglobin as its structure is less compact.
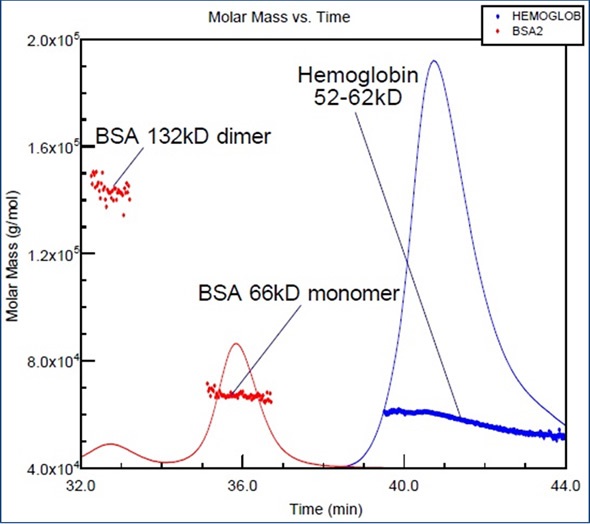
Figure 2. BSA and hemoglobin (proteins with similar molar masses) elute at different volumes, due to the more compact structure of hemoglobin.
Conclusion
When using BSA as a calibration standard (as many laboratories do), the molar mass of hemoglobin could be significantly underestimated. On the other hand, incorrect conclusions can be avoided by combining the chromatography process with a DAWN® or miniDAWN MALS detector.
Reference
Reprinted with permission from “Hemoglobin Characterization” by Wyatt Technology Corp. Graphs and illustrations reprinted with permission from Wyatt Technology.
About Wyatt
 With a long history of excellence in scientific instrumentation, Wyatt Technology is the recognized leader in innovative light scattering instruments, accessories, software and services for determining the properties of macromolecules and nanoparticles in solution. Wyatt provides cutting-edge solutions for in-line multi-angle static light scattering (SEC-MALS), field-flow fractionation (FFF-MALS), composition gradients (CG-MALS), high-throughput and traditional dynamic light scattering (DLS), electrophoretic mobility via phase-analysis light scattering (MP-PALS), differential refractometry and differential viscosity. With a staff composed of 20% Ph.D. scientists and many more dedicated and experienced support personnel, Wyatt's aim is to delight the customer with the best products, training, customer support and service available in the industry.
With a long history of excellence in scientific instrumentation, Wyatt Technology is the recognized leader in innovative light scattering instruments, accessories, software and services for determining the properties of macromolecules and nanoparticles in solution. Wyatt provides cutting-edge solutions for in-line multi-angle static light scattering (SEC-MALS), field-flow fractionation (FFF-MALS), composition gradients (CG-MALS), high-throughput and traditional dynamic light scattering (DLS), electrophoretic mobility via phase-analysis light scattering (MP-PALS), differential refractometry and differential viscosity. With a staff composed of 20% Ph.D. scientists and many more dedicated and experienced support personnel, Wyatt's aim is to delight the customer with the best products, training, customer support and service available in the industry.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.