Carrying out exact movements requires the precise exchange of information between the central nervous system (CNS) and the muscles in the body. In order to achieve a desired movement, limb, motor behavior and neural mechanisms must all be accurately coordinated.
In spite of all of the complex processes that must be in sync with each other, movements still tend to be smooth and fluid. For example, when a hand moves to catch an object it does so via a relatively straight path with the hand’s velocity having a regular bell-shaped profile.
Shutterstock | Alextype
Whilst the nerves in the arm and hand are well known and have been studied in detail, the division between motor neural input and sensory neural input in the movement of the upper limbs has not been fully characterized.
Motor Behavior
It has already been established that the complex interactions between motor and sensory nerves result in precise and fluid motion. This has been shown by studies on patients with CNS injuries, if the patient has a functional motor neuron system but a damaged sensory neuron system, the patient will present impaired movement without simultaneous visual feedback.2
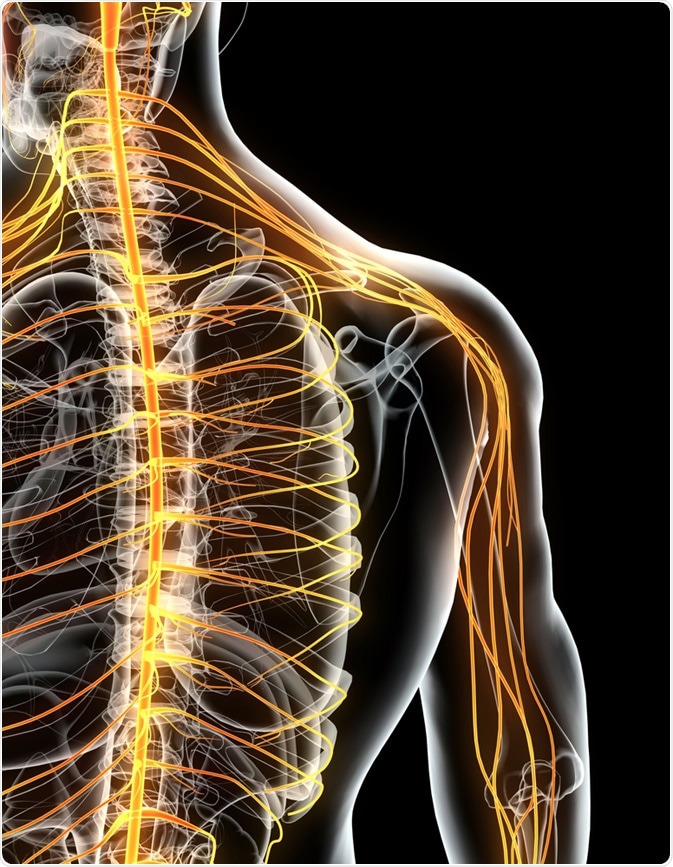
Skilled motor behavior is regulated by CNS control circuits; which includes the spinal cord, cerebral cortex and the brain stem. The CNS is connected to the limbs and other bodily organs via the peripheral nervous system.
The peripheral nervous system’s nerves connect the brain and spinal cord (CNS) to the rest of the body and allow signals to be received and sent, enabling the sensing of, and responding to, stimuli. The peripheral motor system accepts signals from the peripheral nervous system and converts these signals into the muscular motions that result in movement.
The nerves in the peripheral nervous system consist of bundles of axons from neuron cells, with the main components of this system being efferent and afferent somatic axons.3
The efferent axons are responsible for the transmission of signals to muscles to activate them, whereas the afferent axons transmit sensory information from the skin and internal stimuli in joints and muscles. Whilst the two different axon types are separated from one another in the spinal cord, they merge together at one point to form the spinal nerve.
The brachial plexus holds the peripheral nerves used for movement of the arm and the hand. The majority of these nerves are either motor or sensory neurons with information flowing in both directions to provide precise motion control.
Quantification of Nerve Activation
Whilst the route of the different nerves and axons in the brachial plexus has already been established there is little information available on the distribution of motor and sensory axons and in what volumes they are present.4 An important piece of missing knowledge is the number of motor neurons that are responsible for muscle control in this region and if this number is correlated to the accuracy of muscle movement that can be achieved.
This knowledge gap is a result of the lack of appropriate techniques that can be used to determine the number of sensory and motor contributions to the peripheral nervous system. This is because current techniques such as histomorphometric analysis are incapable of differentiating between type I afferent fibers and alpha motor neurons.
Whilst this problem has been solved in animal research, using selective denervation, it had not (until now) been fully solved in humans. Whilst afferent and efferent axons can be distinguished on the basis of their acetylcholinesterase levels this cannot be used to determine if a neuron has a sensory or motor function.5
It has been shown that the enzyme ChAT (choline acetyltransferase) is only expressed in efferent neurons meaning that different types of motor neurons can be distinguished from one another using a monoclonal anti-ChAT antibody.6 Recent research has produced a polyclonal anti-ChAT that selects for motor axons in peripheral nerves,7 providing a method of imaging motor neurons using in regions where only small volumes of ChAT are present.
Study of Neurons Involved in Human Arm Movement
Neurofilament (NF) immunofluorescence and ChAT were used to quantitatively analyze the axonal components of the upper limb nerves of humans. The labeled polyclonal anti-ChAT antibody specifically bound to molecular features found in motor neurons from the spinal cord level down to the terminal nerves found in the wrist.
Transverse portions of a whole nerve, collected from nine human heart-beating organ donors, were analyzed using a complete imaging system – the TissueGnostics TissueFAXS™. The TissueQuest™ and StrataQuest™ software, which can be used to easily process the data included in images of whole-slide samples, were used to automatically determine the number of double-positive and NF-positive (ChAT and NF).
The software used a custom adjusted profile and predefined limits to highlight axons and determine the neuron numbers. The NF label was used as a control, with only ChAT-positive axons that had a reference overlap defined as positive.
To make sure that the research provided repeatable results all of the automatically found positives were visualized as an encircled event. This data was then manually combed through and corrected, and the accuracy of the software was checked independently to ensure deviation was less than 1%.
The research found that approximately only 10% of the axons that innervate the upper limb from the spinal cord are motor neurons.7 In every sample analyzed it was found that there were more sensory axons than motor axons by a ratio of at least 9 to 1. It was also found that there were increasing numbers of sensory axons towards the hand, with only 1,700 motor axons shown to innervate the muscles found in the hand.
This is the first example of research where the proportions and number of motor and sensory axons in the brachial plexus and its branches have been determined. The research showed that an unanticipatedly low number of motor neurons are used for dexterity and movement of the upper limbs. In contrast to this there is a huge number (9 times more) of neurons involved in afferent nervous feedback control for these processes.
These results indicate that movements of high complexity, such as the fine motor control involved in dexterity does not require more efferent axons. Instead, the research suggests that these movements require a higher number of sensory axons.
The researchers have proposed that the most probable central motor mechanism for complex motions, such as hand movements, is the grouping of motor neurons (in order to achieve movements in time with one another) rather than using a greater volume of efferent axons.
References
- Scott SH. Nature Reviews Neuroscience. 2004;5:532–546.
- Cole JD, Sedgwick EM. J Physiol 1992;449:503–515.
- Brushart TM. Nerve repair. New York, NY: Oxford University Press, 2011.
- Farina D, et al. J Physiol 2014;592:3427–3441.
- Gruber H, Zenker W. Brain Res 1973;51:207–214.
- Zimmermann L, et al. J Neurosci 2013;33:2784–2793.
- Gesslbauer B, et al. Ann Neurol 2017;82:396–408.
About TissueGnostics
TissueGnostics (TG) is an Austrian company focusing on integrated solutions for high content and/or high throughput scanning and analysis of biomedical, veterinary, natural sciences and technical microscopy samples.
TG has been founded by scientists from the Vienna University Hospital (AKH) in 2003. It is now a globally active company with subsidiaries in the EU, the USA and China and customers in 30 countries.

TissueGnostics Portfolio
TG scanning systems are currently based on versatile automated microscopy systems with or without image analysis capabilities. We strive to provide cutting-edge technology solutions, such as multispectral imaging and context-based image analysis as well as established features like Z-Stacking and Extended Focus. This is combined with a strong emphasis on automation, ease of use of all solutions and the production of publication-ready data.
The TG systems offer integrated workflows, i.e. scan and analysis, for digital slides or images of tissue sections, Tissue Microarrays (TMA), cell culture monolayers, smears and of other samples on slides and oversized slides, in Microtiter plates, Petri dishes and specialized sample containers. TG also provides dedicated workflows for FISH, CISH and other dot structures.
TG analysis software, apart from being integrated into full systems is fully standalone capable and supports a wide variety of scanner image formats as well as digital images taken with any microscope.
TG also provides routine hematology scanning and analysis systems for peripheral blood, bone marrow and body fluids.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.