Sponsored Content by TissueGnosticsReviewed by Maria OsipovaMar 1 2023
Glioma, a prevailing type of malignant brain tumor, poses a significant threat to public health due to its high morbidity and mortality rates.1
Glioblastoma, the most common and invasive glioma subtype, has a median survival rate of approximately 15 months.1 Despite substantial research efforts, the treatment of glioma has made very little progress over the past ten years.1
In the realm of cancer treatment, immunotherapy has surfaced as a viable strategy for attaining lasting remission by directing and controlling the immune system.2
This methodology has proved to be a breakthrough in managing several forms of solid tumors, primarily through immune checkpoint inhibition.2,3
Therapies that impede the tumor immune microenvironment (TIME) can disrupt the mechanisms through which tumor cells can maneuver to evade the immune system.3
Despite this, clinical trials investigating the efficacy of therapies directed at immune checkpoint proteins PD-1 (or its ligand) and CTLA-4 in glioma yielded negative outcomes.2
Acquiring a better understanding of the operations of immune checkpoints within glioma, and how they interrelate, is crucial in the pursuit of developing efficacious immune checkpoint therapy for the disease.
A team of researchers from Sichuan University in Chengdu, China, delved into the connection between RNA expression and protein expression and the co-localization of a set of cell markers and immune checkpoints in glioma.3
RNA sequencing
The research team scrutinized glioma samples from two genome atlases and examined the correlation between clinical features and RNA expression of three checkpoint inhibitors (PD-L1, VISTA, and B7-H3), as well as three cell markers (GFAP, IBA-1, and CD3, representing glioma cells, tumor-associated macrophages (TAMs), and T-cells, respectively).3
The results of the study revealed that, at the RNA level, all markers, except for PD-L1, exhibited elevated levels of expression in glioma tissue relative to normal tissues.
Augmented expression of PD-L1, B7-H1, CD3, and IBA-1 was significantly linked with increasing tumor grade and predicted inferior overall survival.3
These findings confirm the significance of immune checkpoints, T-cells, and TAMs in the development of glioma.
Multiplex immunofluorescence
To obtain a more comprehensive understanding of the association between phenotype and the aforementioned alterations at the transcript level, the team examined protein expression in glioma samples from 168 patients.3
The researchers utilized seven-color multiplex immunofluorescence (mIF) staining in conjunction with multispectral tissue cytometry (TissueFAXS SPECTRA and StrataQuest) to quantify the expression levels and examine the co-expression of immune checkpoints and cell markers in varying grades of glioma.
The researchers correlated these findings with clinicopathological features, such as age, sex, grade, and recurrence.
Out of all the immune checkpoints and cell markers examined, only B7-H3 demonstrated a statistically significant association with tumor grade, overall survival, recurrence status, and age. The expression of CD3 also decreased as the tumor grade increased.
According to the team, these findings emphasize the potential of B7-H3 as a molecular target for immunotherapy and demonstrate its utility as a prognostic biomarker in glioma, as confirmed by both RNA-seq and mIF analyses.
The team also made a novel discovery, as they found that VISTA, which is known to downregulate T cell-mediated immune responses, was primarily expressed in tumor cells and tumor-associated macrophages (TAMs). This finding underscores the strong correlation between VISTA and TAMs in glioma.
In contrast to their findings regarding B7-H3, PD-L1 expression, which was mainly observed in T cells, was not universally present.
The team suggests that the absence of a relationship between PD-L1 expression and tumor grade may help explain the previous lack of success in using anti-PD-L1 therapy for glioma treatment.
Regarding the interrelationship between the three immune checkpoints, the team found that both B7-H3 and VISTA expression showed a positive correlation with PD-L1 expression.
Image Credit: TissueGnostics
Immune checkpoint insights
As per the researchers' collective suggestion, their findings imply that the immune system's evasion by glioma tumor cells is facilitated by the collaborative function of the three immune checkpoints.
The team states in their findings:
“The correlation of PD-L1, VISTA, and B7-H3 was perhaps consistent with a mutually exclusive expression pattern, which led to immunosuppression of glioma[.]”3
The researchers propose that the co-expression of PD-L1 with T cells, the co-expression of VISTA with TAMs, and the interrelation among B7-H3, VISTA, and PD-L1 in glioma cells may collaboratively generate an immunosuppressive environment for glioma.
This observation has the potential to provide valuable information for the advancement of immunotherapy for glioma.
“We evaluated the clinical and expression characteristics of these immune checkpoints in the sample by mIF and transcriptome data and provided new insights into immunotherapy and the correlation discovery of immune checkpoints,”3 the team concluded in Clinical Immunology.
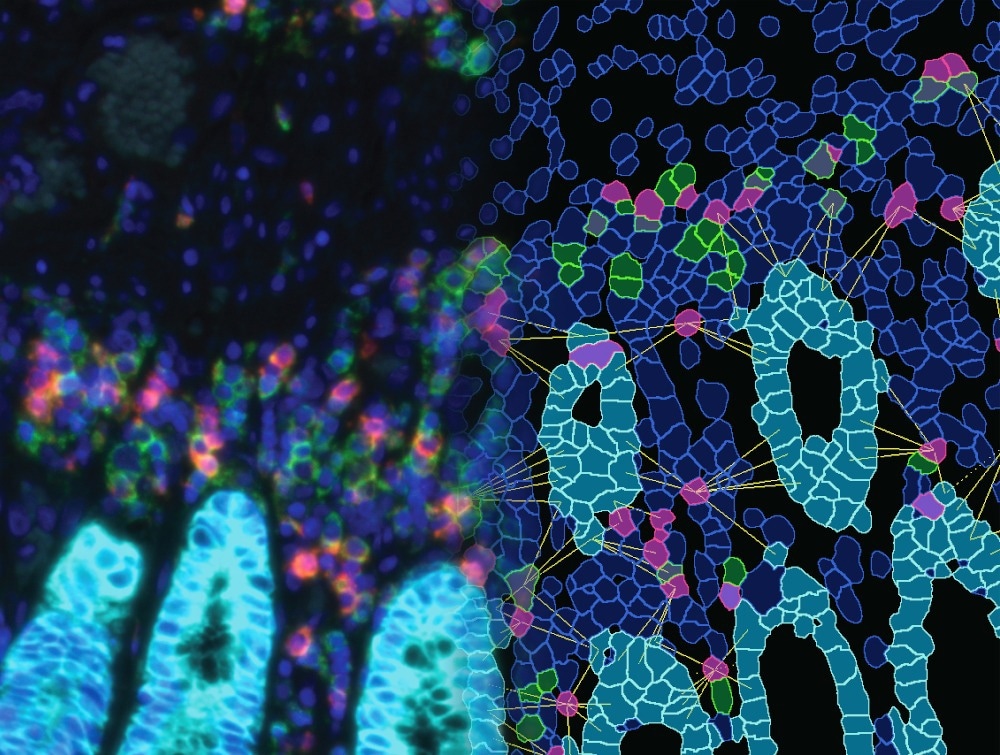
Image Credit: TissueGnostics
Multispectral tissue cytometry
The utilization of the TissueFAXS SPECTRA and StrataQuest analysis software (TissueGnostics) facilitated the acquisition of certain results through whole-slide multispectral imaging and image analysis.
The TissueFAXS SPECTRA system permits the scanning of up to eight markers simultaneously in one run, which makes it an excellent tool for comprehensive phenotyping.
This system employs a liquid crystal tunable filter (LCTF) in conjunction with an inbuilt spectral unmixing algorithm to remove any instances of bleed-through and autofluorescence.
TissueFAXS systems are modular, adaptable, and upgradable to meet specific requirements. They can be customized to perform high-throughput multispectral, widefield fluorescence, confocal, and brightfield whole-slide imaging, enabling users to tailor the system's capabilities to their specific needs.
StrataQuest provides a sophisticated and potent image-processing solution that includes more than 100 apps to aid automation, as well as an Advanced Mode that allows experienced users to design their solutions.
References and further reading
- Ghouzlani A, Kandoussi S, Tall M, et al. (2021) Immune Checkpoint Inhibitors in Human Glioma Microenvironment. Front. Immunol.,12, p. 679425. doi: 10.3389/fimmu.2021.679425.
- Ghouzlani A, Rafii S, Karkouri M, et al. (2021) The Promising IgSF11 Immune Checkpoint Is Highly Expressed in Advanced Human Gliomas and Associates to Poor Prognosis. Front. Oncol., 10, p. 608609. doi: 10.3389/fonc.2020.608609.
- Wang L, Wang Y, He B, et al. (2022) Expression and clinical significance of VISTA, B7-H3, and PD-L1 in glioma. Clin Immunol., 245, p. 109178. Doi: 10.1016/j.clim.2022.109178.

About TissueGnostics
TissueGnostics (TG) is an Austrian company focusing on integrated solutions for high content and/or high throughput scanning and analysis of biomedical, veterinary, natural sciences and technical microscopy samples.
TG was founded by scientists from the Vienna University Hospital (AKH) in 2003. It is now a globally active company with subsidiaries in the EU, the USA and China and customers in 28 countries.
TG systems offer integrated workflows, i.e. scan and analysis, for digital slides or images of tissue sections, Tissue Microarrays (TMA), cell culture monolayers, smears and of other samples on slides and oversized slides, in Microtiter plates, Petri dishes and specialized sample containers.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.