Amyloid Lateral Sclerosis (ALS), otherwise known as motor neuron disease (MND) or Lou Gehrig’s disease, is a neurodegenerative disorder leading to a loss of motor neurons and death, often caused by respiratory failure.
The majority of cases are sporadic (sALS), however, 5-10% are associated with a family history (fALS). This percentage may be underestimated as some patients will have a limited knowledge of their family histories.1 The risk of developing the disease is 1.2-1.5 times higher for males than for females.2 Age is also considered a risk factor with incident rates reaching their peak between the ages of 60 and 75 years.3
SOD1 Structure and Function
A highly conserved, ubiquitously expressed enzyme, Superoxide Dismutase 1 (SOD1) catalyzes the dismutation of superoxide radicals in order to protect organisms from oxidative damage.4 It contains Cu and Zn centers involved in the reaction and, as such, it is also known as Copper-Zinc Superoxide Dismutase:4
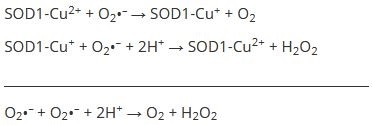
Superoxide radical (O2•–) is more toxic than the oxygen (O2) and hydrogen peroxide (H2O2) products. Another enzyme subsequently converts H2O2 into O2 and H2O.5 In the first step of the reaction the Cu active site is reduced from Cu2+ to Cu+, then oxidized back to Cu2+. A bridging imidazolate ligand6 is used to link the Zn ion to the Cu ion followed by a secondary bridge where Asp 83 is hydrogen-bonded to both a Zn ligand and a Cu ligand.7 The Zn site is not redox active but stabilizes the SOD1 structure.8
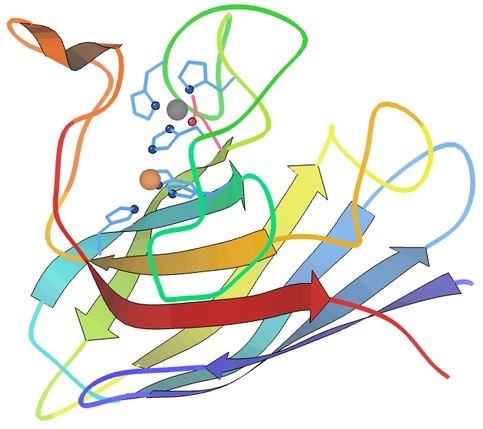
Ribbon diagram showing Cu (orange) and Zn (grey) active sites in SOD1. Made in KiNG from PDB 2C9V. Image Credit: Dcrjsr [CC BY 4.0 (https://creativecommons.org/licenses/by/4.0)]
The SOD1 protein is a dimer and consists of two monomeric subunits. A copper ion, a zinc ion, and a disulfide bond between two cysteine residues are contained in each subunit.9 Of the total soluble protein in the central nervous systems around 1-2% is accounted for with SOD1.10 It is primarily localized in the mitochondria and cytosol10 and has two isoenzymes: SOD2 and SOD3.
Linking SOD1 and ALS
Around 20% of fALS cases are associated with SOD1 mutations11 with over 150 mutations implicated.12 The majority are point mutations.13 It is not clear whether all of these single mutations could cause ALS on their own or whether several would be needed in combination. It is also possible for SOD1 mutations to occur in sALS,14 and these have been associated with variations in fALS survival times.15
It was previously said that SOD1 may lose its ability to scavenge superoxide radicals as a result of these mutations, which would lead to increased oxidative stress. However, it is now believed that SOD1 contributes to ALS by gaining a toxic function as opposed to losing its regular function.16
It is thought that the misfolding of SOD1 and aggregation into oligomers and ultimately larger aggregates is the cause of this toxic gain-of function. This belief is supported by the presence of mutant SOD1–containing aggregates which rapidly increase as disease progresses.17
How SOD1 Mutations Lead to Aggregation
SOD1 usually undergoes the following post-translational modifications:
- Copper Insertion
- Zinc Insertion
- Dimerization
- Disulfide Bond Formation
These post-translational modifications are disrupted by many fALS-associated mutations, which prevents the proper structure and folding of the SOD1 protein.18 Misfolded SOD1 is prone to aggregation into soluble oligomers, through the formation of intermolecular disulfide bonds between the free cysteine residues of different SOD1 molecules and non-covalent interactions (hydrogen bonds) between beta strands of SOD1 subunits.16
Demetallation
Apo, or completely demetallated SOD1, as well as zinc-deficient SOD1, are susceptible to oligomerization.16 It is thought that FALS-associated mutations lower the zinc binding affinity of SOD1 by altering the zinc binding geometry.7 It is possible that solvent is able to access the metal sites through mutations that perturb the electrostatic loop, which prevents metallation with Cu and Zn.19
Dimerization
The surface area accessible to solvent is reduced by dimerization of SOD1, which in turn increases its stability. Unstable monomers are prone to aggregation when dimerization is disrupted. fALS-associated mutations occur on the dimer interphase and may cause dimers to dissociate into monomers which act as aggregation templates.18
Disulfide Bonding
A disulfide bond between two of cysteine residues (Cys 57 and Cys 146) is contained in each SOD1 subunit. SOD1 stability is increased by intrasubunit disulfide, therefore, aggregation of unstable species can result from its interruption. Intrasubunit disulfide bond reduction is required for fibril initiation and promotes faster seeding of aggregates.20
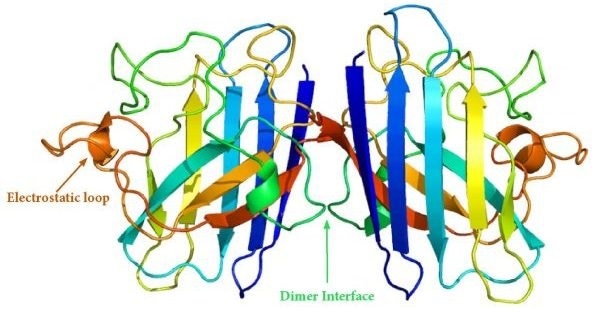
SOD1 Dimer Structure. Image Credit: Adapted from Emw [CC BY-SA 3.0 (https://creativecommons.org/licenses/by-sa/3.0)]
WT SOD1 Aggregation
WT SOD1 aggregation occurs in both fALS and sALS. WT and mutant SOD1 are thought to also aggregate via similar mechanisms.14 In vitro, WT and mutant SOD1 are both self-seeding and cross-seeding.21 Similar effects can be produced through oxidation of WT SOD1 as disease-causing mutations, and SOD1 fibrillation is heavily reliant on redox environment.22
Mechanisms of SOD1 Toxicity
The oligomeric form of SOD1 is usually more toxic than large SOD1 aggregates just as in other neurodegenerative diseases.23 It is hypothesized by some that aggregation is a protective mechanism against these toxic oligomers.24 It is thought that these oligomers contain antiparallel, out-of-register β-sheet structures involving segment 28–38.25
There are a number of proposed mechanisms of toxicity for SOD1 oligomers:
Excitotoxicity
An influx of calcium into the postsynaptic neuron is caused by an increase in the extracellular glutamate concentration. This may result in mitochondrial damage and ultimately apoptosis.26 One drug that extends survival for ALS patients by 2-3 months is Riluzole.27 It is believed to negate excitotoxicity.28
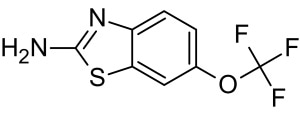
Riluzole Molecular Structure. Image Credit: StressMarq Biosciences
Endoplasmic Reticulum Stress
Mutant SOD1 interacts with ER-associated degradation machinery (ERAD) and leads to it malfunctioning.29 This may also result in apoptosis.
Axonal Transport Disruption
Axonal transport defects may be induced by mutant SOD1 through the reduction of microtubule stability and the disruption mitochondrial transport.30
Oxidative Stress and Mitochondrial Damage
A variety of disease states are associated with oxidative stress. It is also thought to contribute to the pathogenesis of sporadic ALS.31 The responsible ROS could potentially be a product of mitochondrial dysfunction as a result of the accumulation of aggregated SOD1.32 The release of mitochondrial Ca2+ may also contribute to apoptosis.33
Non-Cell Autonomous Toxicity
ALS is considered to be non-cell autonomous. This means that other non-neuronal cells, for example astrocytes and microglia, also contribute to pathogenesis and disease progression.34
“Prion-Like” Cell-to-Cell Transmission
The ability of a protein to initiate self-propagating protein misfolding is referred to as “prion-like”. Unlike PrPSc, SOD1 is not infectious and so would not survive transmission between organisms. Aggregated SOD1 can spread from cell to cell.35 When neurons die, misfolded SOD1 may be released into the extracellular space.35 This can also occur through exocytosis.36 Other cells can then take up this SOD1 through fluid phase endocytosis.36
Interactions with Other Proteins
Alpha synuclein can cross-seed SOD137 and increases its oligomerization rate in mouse models.38 It is also thought that TDP-43 and FUS trigger SOD1 misfolding.39
SOD1 PFFs
StressMarq just launched SOD1 preformed fibrils (PFFs) for ALS research.
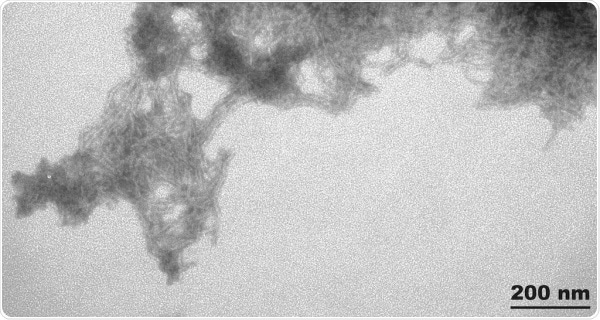
TEM of Human Recombinant Superoxide dismutase Protein Preformed Fibrils (SPR-470). Image Credit: StressMarq Biosciences
References and Further Reading
- Andersen, P.M. and A. Al-Chalabi, Clinical genetics of amyotrophic lateral sclerosis: what do we really know? Nat Rev Neurol, 2011. 7(11): p. 603-15.
- Manjaly, Z.R., K.M. Scott, K. Abhinav, L. Wijesekera, J. Ganesalingam, L.H. Goldstein, A. Janssen, A. Dougherty, E. Willey, B.R. Stanton, M.R. Turner, et al., The sex ratio in amyotrophic lateral sclerosis: A population based study. Amyotrophic Lateral Sclerosis, 2010. 11(5): p. 439-442.
- Chio, A., G. Logroscino, B.J. Traynor, J. Collins, J.C. Simeone, L.A. Goldstein, and L.A. White, Global Epidemiology of Amyotrophic Lateral Sclerosis: A Systematic Review of the Published Literature. Neuroepidemiology, 2013. 41(2): p. 118-130.
- Bergh, J. Structural investigation of SOD1 aggregates in ALS : identification of prion strains using anti-peptide antibodies. 2018.
- Bosco, D. A. (2015) The Role of SOD1 in Amyotrophic Lateral Sclerosis. Nature Education 8(3):4
- Bertini, Ivano and Gray, Harry B. and Lippard, Stephen J. and Valentine, Joan Selverstone (1994) Bioinorganic Chemistry. University Science Books , Mill Valley, CA. 5. Dioxygen Reactions
- Rakhit, R., Chakrabartty, A. (2006). Structure, folding, and misfolding of Cu,Zn superoxide dismutase in amyotrophic lateral sclerosis. Biochim et Biophys Acta. 1762: 1025-1037.
- Messerschmidt, A. Comprehensive Natural Products II: Chemistry and Biology. 8.14 Copper Metalloenzymes. p. 489-545.
- Valentine, J. S., Doucette, P. A., and Zittin Potter, S. (2005) Copper-zinc superoxide dismutase and amyotrophic lateral sclerosis. Rev. Biochem. 74, 563–593
- Pardo CA, Xu Z, Borchelt DR, Price DL, Sisodia SS, Cleveland DW. 1995. Superoxide dismutase is an abundant component in cell bodies, dendrites, and axons of motor neurons and in a subset of other neurons. Proc Natl Acad Sci USA 92:954–8.
- Rosen, D.R., Siddique, T., Patterson, D. et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature, 362 (1993), p. 59-62
- Brasil, A.A., Magalhaes, R.S.S., De Carvalho, M.D.C., Paival, I., Gerhardt, E., Pereira, M.D., Outeiro, T.F., Eleutherio, E.C.A. Implications of fALS Mutations on Sod1 Function and Oligomerization in Cell Models. Mol Neurobiol (2018) 55:5269-5281.
- Guegan C, Przedborski S. Programmed cell death in amyotrophic lateral sclerosis (2003) J Clin Invest 111:153–161.
- Gruzman A, et al. Common molecular signature in SOD1 for both sporadic and familial amyotrophic lateral sclerosis. Proc Natl Acad Sci USA. 2007; 104:12524–12529.
- Alemasov, N.A., Ivanisenko, N.V., Medvedev, S.P., Zakian, S.M, Kolchanov, N.A., Ivanisenko, V.A. Dynamic properties of SOD1 mutants can predict survival time of patients carrying familial amyotrophic lateral sclerosis. J Biomol Struct Dyn. (2017). 35(3):645-656.
- Banci L, Bertini I, Boca M, Girotto S, Martinelli M, et al (2008) SOD1 and Amyotrophic Lateral Sclerosis: Mutations and Oligomerization. PLoS ONE 3(2):e1677.
- Bruijn LI, Houseweart MK, Kato S, Anderson KL, Anderson SD, et al. (1998) Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1. Science 281: 1851–1854.
- McAlary, L., Aquilina, J.A., Yerbury, J.J. (2016). Susceptibility of Mutant SOD1 to Form a Destabilized Monomer Predicts Cellular Aggregation and Toxicity but Not In vitro Aggregation Propensity. Front Neurosci. 10:499.
- Schmitt, N.D., Agar, J.N. Parsing Disease-relevant Protein Modifications from Epiphenoma: Perspectives on the Structural Basis of SOD1-Mediated ALS. (2017). J Mass Spectrom. 52(7): 480–491
- Chattopadhyay, M., Nwadibia, E., Strong, C.D., Gralla, E.B., Valentine, J.S., Whitelegge, J.P. The Disulfide Bond, but Not Zinc or Dimerization, Controls Initiation and Seeded Growth in Amyotrophic Lateral Sclerosis-linked Cu,Zn Superoxide Dismutase (SOD1) Fibrillation. J Biol Chem. (2015) 290(51):30624-36.
- Bunton-Stasyshyn, R.K.A., Saccon, R.A., Fratta, P., Fisher, E.M.C. SOD1 function and its implications for ALS. The Neuroscientist 2015, Vol. 21(5) 519–529
- Alvarez-Zaldiernas, C., Lu, J., Zheng, Y., Yang, H., Blasi, J., Solsona, C., Holmgren, A. Cellular Redox Systems Impact the Aggregation of Cu,Zn Superoxide Dismutase Linked to Familial Amyotrophic Lateral Sclerosis. J Biol Chem. (2016). 291(33):17197-17208.
- Ross CA, Poirier MA (2006) Protein Aggregation and Neurodegenerative Disease. Nat Med 10: S10–17.
- Zhu, C., Beck, M.V., Griffith, J.D., Deshmukh, M., Dokholyan, N.V. (2018). Large SOD1 aggregates, unlike trimeric SOD1, do not impact cell viability in a model of amyotrophic lateral sclerosis. Proc Natl Acad Sci USA. 115(18):4661-4665.
- Sangwan, S. et al. (2017). Atomic structure of a toxic, oligomeric segment of SOD1 linked to amyotrophic lateral sclerosis (ALS). PNAS 114(33):8770-8775.
- Van Den Bosch, L., Van Damme, P., Bogaert, E., Robberecht, W. (2006). The role of excitotoxicity in the pathogenesis of amyotrophic lateral sclerosis. BBA – Molecular Basis of Disease. 1762(11-12):1068-1082.
- Miller, R., Mitchell, J., Moore, D. Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND). Cochrane Database Syst Rev. 2012; 3
- Bensimon, G., L. Lacomblez, and V. Meininger, A controlled trial of riluzole in amyotrophic lateral sclerosis. ALS/Riluzole Study Group. N Engl J Med, 1994. 330(9): p. 585-91.
- Nishitoh, H., Kadowaki, H., Nagai, A., Maruyama, T., Yokota, T., Fukutomi, H., Noguchi, T., Matsuzawa, A., Takeda, K., Ichijo, H. (2008). ALS-linked mutant SOD1 induces ER stress- and ASK1-dependent motor neuron death by targeting Derlin-1. Genes & Dev. 22:1451-1464.
- De Vos, K.J., Hafezparast, M. (2017). Neurobiology of axonal transport defects in motor neuron diseases: Opportunities for translational research? 105: 283-299.
- D’Amico E., Factor-Litvak P., Santella R. M., Mitsumoto H. (2013). Clinical perspective on oxidative stress in sporadic amyotrophic lateral sclerosis. Free Radic. Biol. Med. 65, 509–527.
- Ferri A., Cozzolino M., Crosio C., Nencini M., Casciati A., Gralla E. B., et al. . (2006). Familial ALS-superoxide dismutases associate with mitochondria and shift their redox potentials. Natl. Acad. Sci. USA 103, 13860–13865
- Chinnery PF, Elliott HR, Hudson G, et al. Epigenetics, epidemiology and mitochondrial DNA diseases. Int J Epidemiol. 2012;41(1):177–187.
- Ilieva, H., Polymenidou, M., Cleveland, D.W. Non–cell autonomous toxicity in neurodegenerative disorders: ALS and beyond. (2009) J Cell Biol. 187(6):761-772.
- Grad, L.I., Cashman, N.R. (2014). Prion-like activity of Cu/Zn superoxide dismutase, Prion, 8:1, 33-41.
- Lee, S., Kim, H. (2015) Prion-like Mechanism in Amyotrophic Lateral Sclerosis: are Protein Aggregates the Key? Exptl Neurobiol. 24(1):1-7.
- Helferich, A.M, McLean, P.J., Weishaupt, J.H., Danzer, K.M. (2016), Alpha-synuclein interacts with SOD1 and promotes its oligomerization. J Neurol & Neuromed. 1(7):28-30.
- Koch, Y., Helferich, A.M., Steinacker, P., Oeckl, P., Walther, P., Weishaupt, J.H., Danzer, K.M., Otto, M. Aggregated a-Synuclein Increases SOD1 Oligomerization in a Mouse Model of Amyotrophic Lateral Sclerosis (2016). 186(8):2152-2161.
- Pokrishevsky, E., L.I. Grad, and N.R. Cashman, TDP-43 or FUS-induced misfolded human wild-type SOD1 can propagate intercellularly in a prion-like fashion. Scientific Reports, 2016. 6: p. 22155.
Acknowledgments
Produced from materials originally authored by Patricia Thomson from StressMarq Biosciences Inc.
About StressMarq Biosciences
Established in 2007, StressMarq Biosciences Inc. is a supplier of life science products that operates out of Victoria, Canada with a small, but dedicated group of scientists. Headed by our CEO and President Dr. Ariel Louwrier, StressMarq provides the research community with high-quality reagents backed with rigorous quality control data, expert scientific support, and fast international delivery.
“Discovery through partnership, Excellence through quality”
With over 7,000 products, our growth can be attributed to the continual production of cutting edge research products. Our diverse portfolio of primary antibodies, antibody conjugates, proteins, immunoassay kits and small molecules bridges across the life sciences, including products for cancer research, cardiovascular disease, cell signaling and neuroscience. To aid research worldwide, StressMarq has an extensive network of international distributors that allow us to supply reagents to over 50 countries.
In the years to come, StressMarq will continue to aid life science research by providing “Discovery through partnership, and Excellence through quality”.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.