The advancement in the field of immunotherapeutics has led to a new drive to comprehend the tumor microenvironment. Immunohistochemistry is a robust tool for the investigation of the expression, distribution and activation of proteins in situ.
Antibodies linked to an antigen of interest are utilized to identify the antigen in thin sections of flash-frozen or formalin-fixed paraffin-embedded tissue.
The antigen is visualized by making use of fluorescent reporters or an enzymatic reaction that induces chromogen precipitation at the site of antibody-antigen binding.
Fluorescent reporters might be conjugated directly to the primary antibody that helps detect the antigen of interest (direct immunofluorescence), or may be fixed to a secondary antibody that detects the species-specific primary antibody (indirect immunofluorescence).
The second one is more common, as it is capable of achieving more sensitive antigen detection.
Multiplex immunohistochemistry and its benefits
Historically, this method has been carried out separately for each marker of interest. But recently, molecular histopathology has been moving from single-marker immunohistochemistry towards multiplexed detection of biomarkers.
Multiplex immunohistochemistry, also known as multiplex immunostaining or multiple immunolabeling, can increase the amount of data obtained from a single sample. This is crucial in cases where the sample is less, for example, a tumor biopsy or other clinical specimen.
In contrast to next generation sequencing and mass spectrometry, multiplex immunohistochemistry also enables the investigation of spatial arrangement of proteins of interest, as well as protein interaction or co-localization.
Although chromogenic detection is compatible with multiplex immunohistochemistry to some extent, indirect labeling with tyramide-based fluorescent detection offers various advantages.
In the tyramide-based technique, fluorophore-conjugated tyramide is deposited at the site of the target antigen, thereby amplifying the signal efficiently.
Tyramide signal amplification (TSA) allows the identification of targets of specifically low abundance, apart from improving the fluorescence signal. Most significantly, TSA also enables unlabeled primary antibodies to be used, with the flexibility to use several antibodies cultured in the same species.
One more important advantage of tyramide-based fluorescent detection is the permanent nature of the tyramide-antigen bond, which is achieved via its covalent bonding with tyrosine residues on or next to the target antigen.
The stability of this binding enables heat-mediated elimination of antibodies, while preserving the fluorescence signal linked to the antigen of interest.
Thus, sequential applications of targeted antibodies can be used without any problem for the cross-reactivity that would be anticipated otherwise when multiple antibodies from the same host species are used.
Using fluorophores with clear emission wavelengths and non-overlapping excitation simplifies visualization of every special target protein. Multispectral imaging enables separation of fluorophores with partially overlapping spectra and inhibition of tissue autofluorescense.
The latest advances in multiplex immunohistochemistry and multispectral imaging allow a precise, concurrent analysis of multiple tissue markers. For instance, detection of markers of both autophagy and proliferation in intestinal tissue can support precise evaluation of epithelial turnover.
Clinically, gaining insights into the tumor microenvironment and further selection of suitably targeted therapies can be supplemented by localizing and profiling immune checkpoint proteins. Multiplex immunohistochemistry finds various applications covering basic, translational, and clinical research applications.
Bethyl provides many multiplexing panels and a wide range of immuno-oncology targets.
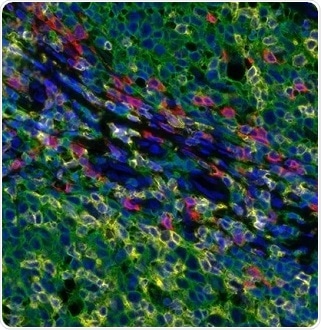
Figure 2. Detection of human CD3 (yellow), CD8 (red), and CD20 (green) in FFPE tonsil by IHC-IF. Rabbit anti-CD3e recombinant monoclonal [BL-298-5D12] (A700-016), rabbit anti-CD8a recombinant monoclonal [BLR044F] (A700-044), mouse anti-CD20 monoclonal [L26] (A500-017A). Secondary: HRP-conjugated goat anti-rabbit IgG (A120-501P) and HRP-conjugated goat anti-mouse IgG (A90-116P). Substrate: Opal™ 520, 620, and 690. Counterstain: DAPI (blue). Image Credit: Bethyl Laboratories Inc.
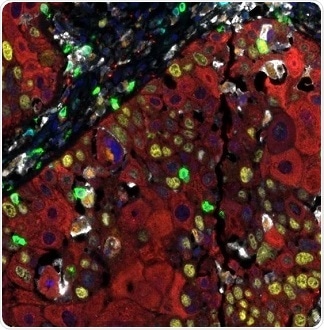
Figure 3. Detection of human CD3 (teal), CD8 (green), CD68 (orange), CK (red), Ki67 (yellow), and PD-L1 (white) in FFPE HNSCC by IHC-IF. Rabbit anti-CD3e recombinant monoclonal [BL-298-5D12] (A700-016), rabbit anti-CD8a recombinant monoclonal [BLR044F] (A700-044), mouse anti-CD68 monoclonal [KP-1] (A500-018A), mouse anti-cytokeratin monoclonal [AE1/AE3] (A500-019A), rabbit anti-Ki67 monoclonal [BLR021E] (A700-021), and rabbit anti-PD-L1 recombinant monoclonal [BLR020E] (A700-020). Secondary: HRP-conjugated goat anti-rabbit IgG (A120-501P) and HRP-conjugated goat anti-mouse IgG (A90-116P). Substrate: Opal™ 480, 520, 570, 620, 690, and 780. Counterstain: DAPI (blue). Image Credit: Bethyl Laboratories Inc.
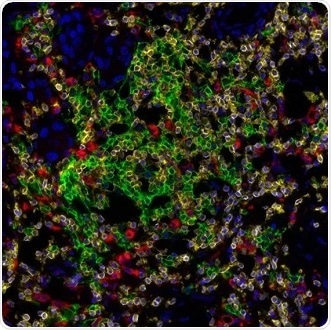
Figure 4. Detection of human CD3 (yellow), CD20 (green), and CD68 (red) in FFPE lung carcinoma by IHC-IF. Rabbit anti-CD3e recombinant monoclonal [BL-298-5D12] (A700-016), mouse anti-CD20 monoclonal [L26] (A500-017A), and mouse anti-CD68 monoclonal [KP-1] (A500-018A). Secondary: HRP-conjugated goat anti-rabbit IgG (A120-501P) and HRP-conjugated goat anti-mouse IgG (A90-116P). Substrate: Opal™ 520, 620, and 690. Counterstain: DAPI (blue). Image Credit: Bethyl Laboratories Inc.
About Bethyl Laboratories, Inc.

Bethyl Laboratories, Inc. has been dedicated to improving lives by supporting scientific discovery through its qualified antibody products and custom polyclonal services since its founding in 1972. Bethyl has a global reputation for quality, consistency and first-class customer care. Every antibody that Bethyl sells is manufactured to exacting standards in Montgomery, Texas, and is validated in-house by a team of scientists. From the veterinary facilities to the development, production, and validation labs, the entire Bethyl team focuses on delivering quality products and delighting customers.
Bethyl Laboratories has been acquired by Fortis Life Sciences. To learn more visit: https://www.fortislife.com/fortis-life-sciences-acquires-bethyl-laboratories.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.