Neurological diseases like Alzheimer's and Parkinson's are a serious global health challenge. Despite enormous investment in drug discovery and development pipelines, progress has been hindered by many obstacles.
One major problem was the lack of dependable in vitro and in vivo models for accurate chemical screening and lead optimization. Traditional processes using cell lines and animal experiments often fail to capture the complexity of the human brain and its pathologies.
This changed in 2014, when Lancaster et al.1 created the first brain organoid model. These organoids are intricate 3D miniature replicas of sections of the human brain grown from human induced pluripotent stem cells.
Unlike standard 2D cultures, brain organoids comprise a variety of cell types that interact with one another to recreate the architecture and structure of genuine tissue. Their complexity makes them ideal models for studying brain development and illnesses.
Brain organoid generation protocols have evolved dramatically in recent years, making these models valuable tools in drug discovery, disease modeling, and personalized medicine.2
Despite this progress, translating brain organoids into drug discovery tools remains challenging due to low reproducibility and costly, labor-intensive processes that impede scalability.
Although differentiation techniques from induced pluripotent stem cells (iPSCs) into brain organoids vary across model systems, they often involve medium exchange over several months, as well as constant monitoring. This protracted culture procedure causes substantial variability between wells and plates, which remains a significant restriction.
The CellXpress.ai® Automated Cell Culture System is designed to address these issues. The system integrates a liquid handler, imager, and incubator into a single platform that can seed, feed, passage, and monitor both 2D and 3D cell cultures (Figures 1 and 2).
This article describes an automated brain organoid creation workflow using the CellXpress.ai system, beginning with iPSCs and progressing to differentiation and maturation of complex brain organoids. Single-cell or fragment passaging techniques have been developed for stem cell culture.
The starting material is crucial for stem cell development into brain organoids. Stem cells should form healthy, undifferentiated colonies.
To control stem cell quality, the researchers used IN Carta image analysis software, which uses advanced artificial intelligence to discern between healthy and differentiated colonies.
The researchers implemented a workflow to create brain organoids using a rocker rather than the traditional orbital shaker. Organoid quality was determined using functional tests and whole-mount staining.
The researchers also demonstrated how to seamlessly integrate other devices, such as the ImageXpress® Confocal HT.ai High Content Imaging System, for advanced monitoring using the CellXpress.ai system's back port.
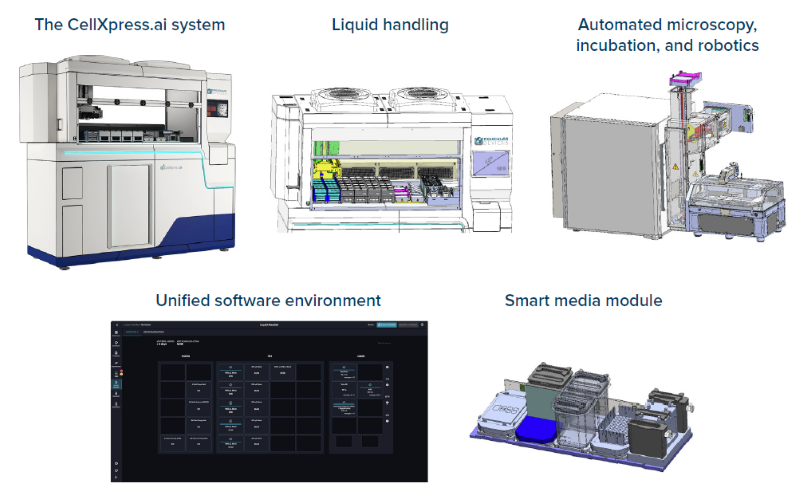
Figure 1. The CellXpress.ai system's built-in hardware and software. Image Credit: Molecular Devices UK Ltd
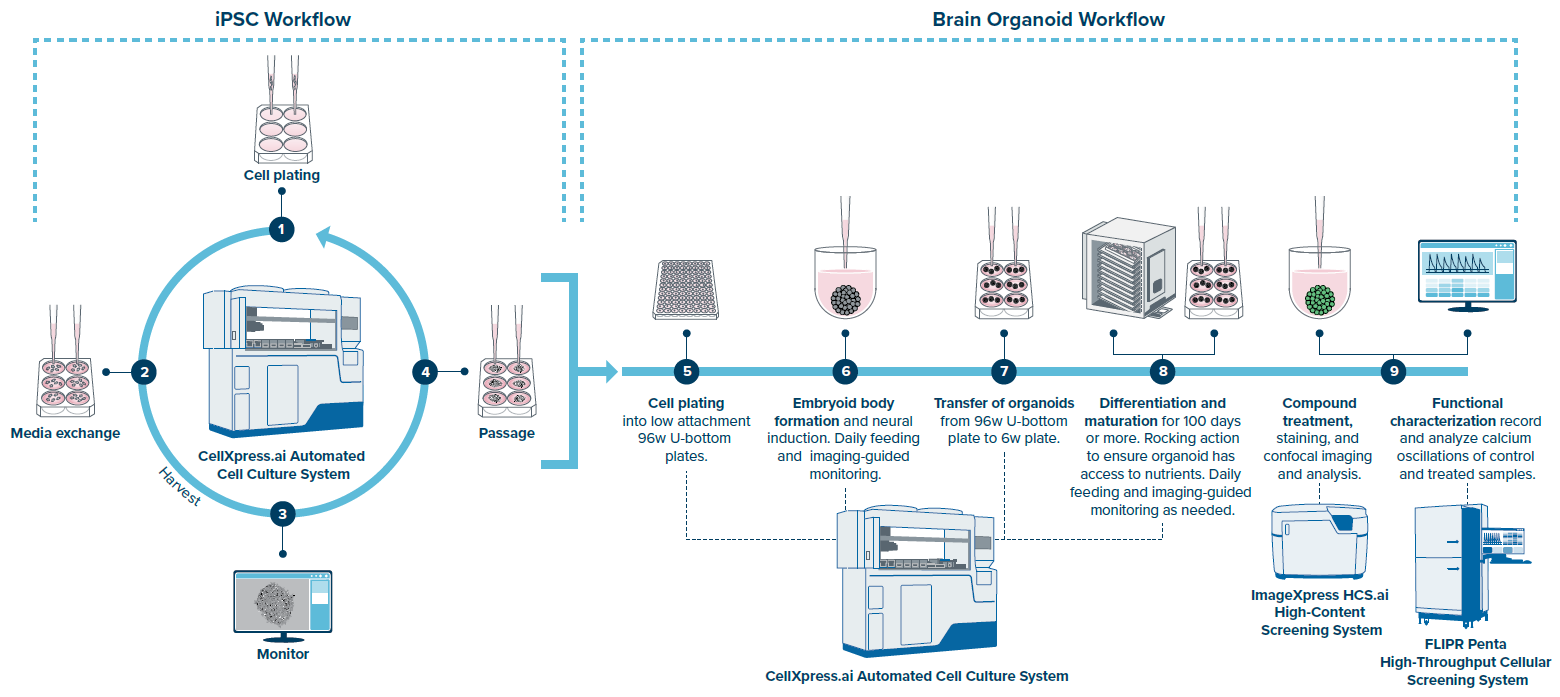
Figure 2. The CellXpress.ai system supports the cultivation of both human iPSCs and brain organoids. Image Credit: Molecular Devices UK Ltd
Variability can be decreased by improving CellXpress.ai's liquid handling procedures, which reduces contamination risk and minimizes organoid loss or damage during media aspiration and dispensing.
The system's "Smart Media Module" increases hands-off time dramatically by allowing for multiple days of on-deck reagent storage. In addition, those media modules will preheat the media to ensure it is at the appropriate temperature when feeding begins and remains at that temperature until all plates are processed.
Following the feeding procedure, media is automatically cooled for on-deck storage (Figure 1). In addition to media containers, reservoir plates can be kept on deck.
The CellXpress.ai Automated Cell Culture System can include a rocking incubator to allow for the culture of free-floating organoids, such as brain organoids, by ensuring constant media agitation during the maturation process.
This rocking incubator can accommodate up to six racks and features a mix-and-match arrangement that allows for both static and rocking conditions. Thus, stem cells can be grown in the same incubator as brain organoids. This functionality is critical for offering a complete workflow for brain organoid production.
The CellXpress.ai system enables continuous culture monitoring via its built-in imager and employs machine learning-assisted image analysis through the use of the IN Carta® Image Analysis Software.
Acknowledgments
Produced from materials originally authored by Felix Spira and Sandra Grund-Gröschke from Molecular Devices.
References
- Lancaster, M.A. and Knoblich, J.A. (2014). Generation of cerebral organoids from human pluripotent stem cells. Nature Protocols, 9(10), pp.2329–2340. DOI: 10.1038/nprot.2014.158. https://www.nature.com/articles/nprot.2014.158.
- Birtele, M., Lancaster, M. and Giorgia Quadrato (2024). Modelling human brain development and disease with organoids. Nature Reviews Molecular Cell Biology. (online) DOI: 10.1038/s41580-024-00804-1. https://www.nature.com/articles/s41580-024-00804-1.
About Molecular Devices UK Ltd 
Molecular Devices is one of the world’s leading providers of high-performance bioanalytical measurement systems, software, and consumables for life science research, pharmaceutical, and biotherapeutic development. Included within a broad product portfolio are platforms for high-throughput screening, genomic and cellular analysis, colony selection, and microplate detection. These leading-edge products enable scientists to improve productivity and effectiveness, ultimately accelerating research and the discovery of new therapeutics. Molecular Devices is committed to the continual development of innovative solutions for life science applications. The company is headquartered in Silicon Valley, California, with offices around the globe. For more information, please visit www.moleculardevices.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.