Oxygen transfer is a key limitation in aerobic fermentations that directly impacts cell growth, productivity, and bioprocess efficiency.

Figure 1. H.E.L Group’s BioXplorer 400P. Image Credit: H.E.L Group
Introducing more pressure to a bioreactor facilitates an increase in the Oxygen Transfer Rate (OTR), which in turn improves gas solubility in the fermentation broth. More bioprocess scientists are switching to pressurized systems to improve the availability of dissolved gases and expedite gas-dependent bioprocesses.
This article discusses how operating at increased pressure has a considerable impact on the availability of dissolved oxygen and OTR (kLa), using the Bioxplorer 400P high-pressure parallel bioreactor system.
There is an improvement in oxygen solubility in the fermentation broth with an increase in pressure, facilitating faster and more efficient bioprocesses. The Bioxplorer 400P enables precision control of dissolved oxygen by pressure, as it provides a powerful alternative to conventional agitation-based strategies.
Key benefits
- Accelerates fermentation by enhancing oxygen availability
- Enables higher productivity without influencing shear stress
- Provides a scalable route for the optimization of oxygen transfer
- Allows precise dissolved oxygen control via pressure
- Reduces reliance on agitation for oxygen transfer, reducing shear stress and protecting sensitive cultures
What is kLa: Why is it important in fermentations?
kLa refers to the volumetric oxygen mass transfer coefficient and is used to measure the efficiency of oxygen transfer from bubbles to liquid in the bioreactor.
Effectively, kLa clarifies the speed at which oxygen is made available to cells, making it a key parameter for aerobic fermentation performance, productivity, and scale-up. Greater operating pressures raise oxygen solubility in the fermentation broth, which can improve oxygen transfer and boost process efficiency all round.
Previous studies conducted with the H.E.L bench-top bioreactor systems (BioXplorer 100 & 400) have demonstrated that a constant kLa can be leveraged as a basis for scale-up, as the values acquired from the instrument can be reproduced accurately in traditional-scale bioreactors (Gill et al., 2008).
In these studies, kLa is used as a measure of how effective the oxygen transfer is for liquid under pressure.
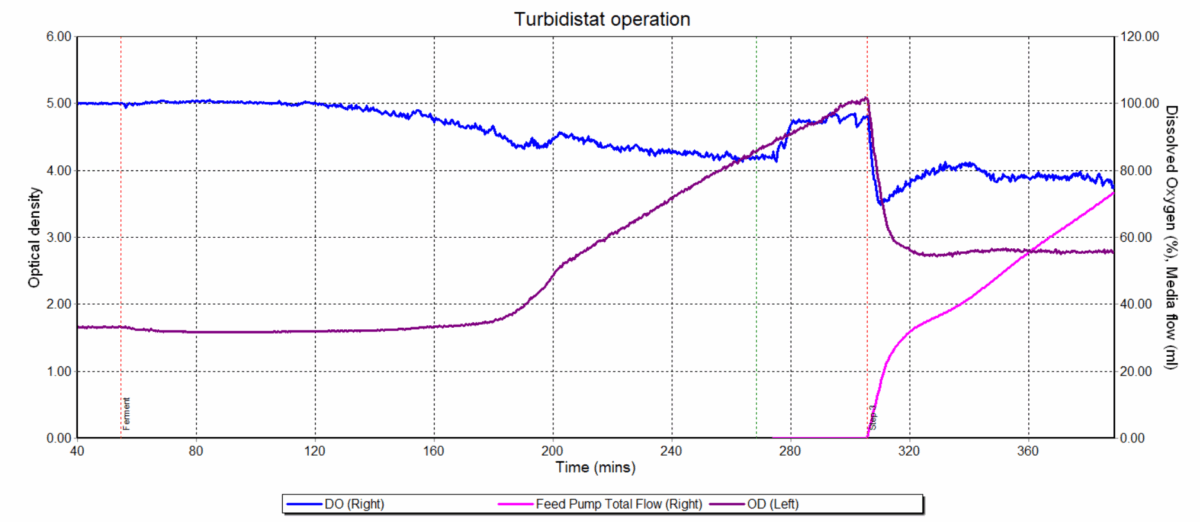
Figure 2. Turbidostat control profile of BioXplorer 100. Image Credit: H.E.L Group
The BioXplorer 400P
The BioXplorer 400P is an automated four-zone parallel pressure bioreactor system developed for the discovery and optimization of novel bioprocesses.
Designed for applications ranging from syngas fermentation to cell-free enzymatically catalyzed processes, the BioXplorer 400P leverages pressure capabilities in combination with precise gas feed control to accelerate bioprocesses that are dependent on the availability of dissolved gases.
Materials and methods
Experiments were designed to assess the influence that pressure has on oxygen transfer under controlled conditions:
- Reactor: 500 mL stainless steel reactor
- Working volume: 300 mL w/v dilute solution of sodium sulphite.
- Gas: 1 vvm (300 sccm) air gas blend containing H2.
- Temperature: 37 °C.
- Stirring: 1500 rpm.
The BioXplorer 400P stainless steel pressure vessel was equipped with a gas sparger, pH probe, DO probe, overhead motor, and magnetic drive with Ruston impeller (Figure 2). The system was subjected to increased pressure cycling between gassing with air and scrubbing with nitrogen.
Results and discussion
Figure 3 exhibits the dissolved oxygen (DO) profile of a reactor as it is cycled between gassing with air and nitrogen. As pressure increases, the DO in the liquid increases within the reactor.
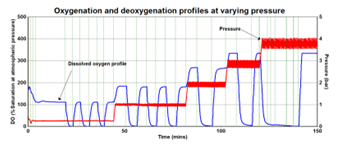
Figure 3. Oxygenation and deoxygenation profile at various pressures over time. Image Credit: H.E.L Group
As displayed in Figure 4, the gradient of the DO profile depicts the rate of change of oxygen concentration, which is a measure of oxygen flux. Applying the fact that 100 % saturation aligns with an oxygen concentration of ∼9 mg/L (at room temperature), the analogous oxygen flux profile (in grams per liter per minute) can be generated.
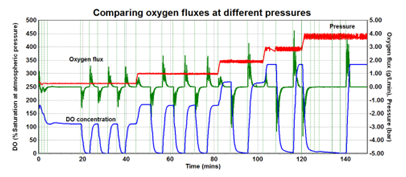
Figure 4. Comparison of O2 flux at different pressures. Image Credit: H.E.L Group
Pressure can be used to manipulate the oxygenation level. This would typically be achieved through a manipulation of the stirrer speed. However, this is not possible when handling more fragile organisms such as algae and amoeba due to the negative influence of shear forces on cell integrity.
The data in Figure 5 shows a control loop that was created in this experiment to link a requested DO set point with the control output to a back-pressure regulator (BPR).
The system’s pressure will change periodically as a result of automatic regulation dictated by BPR, which is not, however, manipulated explicitly.
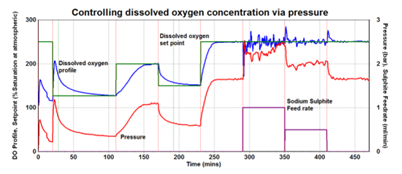
Figure 5. Control of dissolved oxygen via pressure over time using an automatically controlled BPR. Oxygen concentration increases stepwise with pressure showing clear correlation between pressure and oxygen transfer rate. Image Credit: H.E.L Group
Data was processed using H.E.L analysis tools. Each of the colored sections represent a separate curve-fitting region, and the markers represent measured data points.
Each fitted curve yields a different kLa estimate. The kLa values demonstrate a high level of reproducibility, with kLa = 2.0 min -1 +/- 10 %, i.e., independent of pressure.
During the first 250 minutes of the experiment, the DO set point fluctuated at a range between 120 and 250 %. The reactor DO intervals were easily attained and automatically maintained using a BPR assist, which regulated pressures between 0.4 and 2.5 bar.
The set point of the DO was stabilized at around 250 minutes, and the DO stability was evaluated by introducing sodium sulphite, which extracts oxygen from the liquid in an oxygen scavenging reaction.
After sodium sulphite has been nourished, the BPR demonstrated a notable rise in the reactor’s pressure to ensure the DO remained at the set point until the experiment concluded.
Conclusion
In agreement with Henry’s Law, the DO concentration (but not kLa) scales approximately linearly with oxygen partial pressure. In these high-pressure experiments, the DO value underwent a significant improvement and demonstrated a near-proportional relationship with applied pressure.
Operating bioprocesses at elevated pressures (e.g., up to 10 bar) using the BioXplorer 400P can lead to significant improvements in oxygen transfer. Achieving comparable gains via kLa optimization alone would present a considerable challenge, due to additional factors such as gas–liquid interfacial area, bubble size, and agitation.
By leveraging pressure rather than increased agitation, the risk of shear stress is minimized, preserving and maintaining sensitive cultures.
The results show that applying pressure in the BioXplorer 400P bioreactors boosts DO levels and improves gas transfer rates. This makes pressure-enhanced transfer a power tool to improve yield, productivity, and biomass growth across complex bioprocesses.
About H.E.L Group
H.E.L develops and manufactures innovative scientific instruments and software designed to optimize the efficiency, safety, and productivity of key processes in chemistry and biology applications.
The H.E.L team of 70 includes highly skilled process and software engineers, based at their extensive research and manufacturing facilities in the UK, as well as sales and support offices around the world.
H.E.L has a long history of solving complex challenges for customers. Since 1987, the Company has worked with businesses and laboratories globally, providing proprietary automated solutions for the pharma, biotechnology, chemical, battery, and petrochemical sectors.
We continue to expand the reach of our products and services to further support and enable R&D and process optimization across Europe, the US, China, and India.
H.E.L is accredited with ISO 9001: 2015
Our mission
To help create a healthier, more sustainable, safer world for everyone.
Our vision
We equip scientists with the right tools and knowledge to develop safe, efficient new processes and molecules that benefit the world and its population.
Our values
Insightful through experience. With over 30 years of in-house expertise and experience, we know how to overcome a challenge
Collaborative by design. Dedicated to listening, learning, and working closely with industry experts, we empower others to fulfill potential
Tenacious in spirit. Always looking for new and innovative solutions, we don’t stand still; instead, we are focused on reaching the next success
Proud of progress. Fueled by our ability to make a real difference, while celebrating the achievements of others
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.