The insulin gene produces the polypeptide hormone insulin, which is required for normal glucose homeostasis and regulation of blood sugar levels. The lack of insulin or insulin insufficiency can lead to diabetes mellitus, which affects up to 5% of the human population.
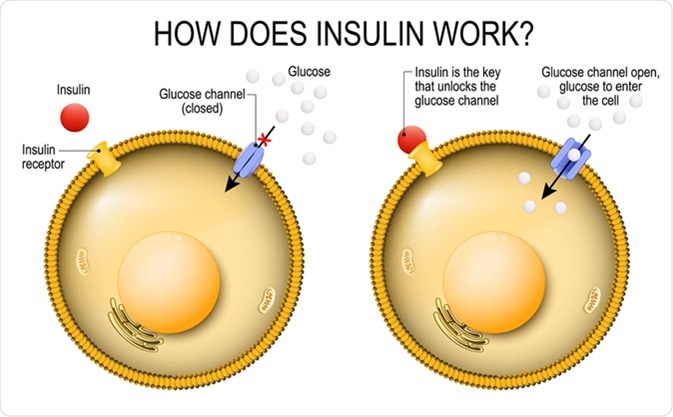
Insulin regulates the metabolism and is the key that unlocks the cell's glucose channel. how does insulin work. Image Credit: Designua / Shutterstock
The Genetics of the Insulin Gene
Insulin is formed as a precursor protein pre-proinsulin. This is encoded by a 14kb sequence on the INS gene. In most animals including humans, a single gene for insulin is found. The human gene is located on the short arm of chromosome 11 at position 15.5 (11p15.5).
The insulin gene has been recently de-coded in its complete form in genomic studies. Human and rat insulin genes have been cloned and the DNA has been sequenced. It has been shown that mouse and rat insulins are identical and they have similar gene sequences and organization, which are similar in genetic sequences to humans.
Insulin injection. Image Credit: JPC-PROD / Shutterstock
Gene Activation and Inhibition
The insulin gene is expressed almost exclusively in pancreatic β-cells (Islets of Langerhans). Glucose in the blood is the major stimulant that regulates the insulin gene expression and enables the beta cells to produce insulin and maintain an adequate store of intracellular insulin to sustain the metabolic demand.
Glucose in blood acts via transcription factors such as pancreatic/duodenal homeobox-1 (PDX-1, mammalian homologue of avian MafA/L-Maf (MafA), Beta2/Neuro D (B2)), and controls the rate of transcription, and the stability of insulin mRNA. This functions at the level of synthesis and secretion of insulin.
Low insulin production in diabetes may occur if there are continued high levels of glucose or lipids in the blood. This can lead to glucotoxicity (sugar related toxicity) or lipotoxicity (fat related toxicity) respectively. Consequentially, there is worsening of β-cell function in type 2 diabetes, in part via inhibition of insulin gene expression.
This glucotoxicity involves decreased binding activities of PDX-1 and MafA and increased activity of C/EBPβ. High levels of glucose also leads to cellular damage as a result of the generation of reactive oxygen species (ROS) leading to oxidative stress. Lipotoxicity also leads to de novo ceramide synthesis and involves the inhibition of PDX-1 nuclear translocation and MafA gene expression.
Genetic Variants and Diabetes
There are several genetic mutants of the INS gene. There is a read-through gene, INS-IGF2 that can overlap with the INS gene at the 5' region and with the IGF2 gene at the 3' region.
For example, key genetic variants within INS such as rs689 and within INSR (rs1799816) have been shown to be correlated with the onset of Type-2 diabetes in 3 distinct ethnic groups.
Understanding how different genetic variants and polymorphisms that alter insulin or glucose metabolism is key to preventing and treating diabetes in the future.
Using Gene-Editing to Cure Type 1 Diabetes
Sources
- https://ghr.nlm.nih.gov/gene/INS
- Owerbach et al, 1980. Nature 286:82-84 https://www.nature.com/articles/286082a0
- Joshi et al, 2007. Supplement of Japi 55:19-25 https://www.japi.org/
- Sokhi et al, 2016. Mol Genet Genomics 291(1):205-16 https://www.ncbi.nlm.nih.gov/pubmed/26251103
Further Reading
Last Updated: Jun 20, 2023