This article is based on a poster originally authored by Daniel Schieffer, Karissa Cottier, and David Ash.
Primary hepatocytes are an important cell type in toxicity and drug metabolism workflows. Given the sensitivity of these tests, consistent and precise hepatocyte counts are required to achieve reproducible results in downstream applications.
Automated cell counters were first introduced roughly 20 years ago and are now commonly used to standardize cell counting operations across a variety of laboratory settings.
However, the cell-counting algorithms typically used in these devices struggle to reliably count hepatocytes due to their uneven morphologies, tendency to clump, and multinucleated nature.
To solve this issue, researchers have developed a machine-learning-based model to reliably determine hepatocyte viability on an automated cell counter. This will:
- Help standardize results across laboratories
- Reduce user variability
- Reduce the time needed for counting (Figure 1)
The presented study aims to determine whether an automated machine learning model can achieve the same level of counting accuracy for hepatocyte quantification as the recognized gold standard: manual hemocytometer counts performed by trained scientists.
Datasets were created independently by internal DeNovix scientists and collaborators from a leading commercial hepatocyte source (BioIVT). This study examined the survival and concentration of cryopreserved cells from humans, rats, mice, and dogs, as well as of freshly obtained mouse hepatocytes.
Methods
Machine learning model development
The machine learning hepatocyte counting model was developed using cryopreserved and freshly isolated hepatocytes from four common toxicology research species (human, canine, rat, and mouse).
To develop the unique cell counting model, the training dataset included several hundred full-size images from each of the four species. DeNovix scientists validated the model's performance, which was then confirmed by BioIVT experts.
Hepatocyte preparation
BioIVT (Baltimore, MD) provided cryopreserved hepatocytes from humans, dogs, and rats. Prior to staining, each sample was thawed and resuspended in Invitrogro KHB medium following manufacturer guidance.
Hemocytometer: Hepatocyte counting and viability
Hepatocytes were stained with 0.4 % Trypan Blue (Sigma Aldrich, St. Louis, MO) by combining equal quantities of cells and 0.4 % Trypan Blue (50 µL of each)–or 10 µL of 0.4 % Trypan Blue and 90 µL of cells–and homogenized with mild inversion.
Each sample was counted in triplicate with a Neubauer grid hemocytometer mounted on a 4x brightfield microscope. Each replication received a fresh 10 µL sample volume.
CellDrop automated: Hepatocyte counting and viability
Hepatocyte samples were stained with a dual fluorescent dye containing 12 µm Acridine Orange and 140 µm Propidium Iodide (AO/PI).
Cells and AO/PI were mixed in equal volumes (50 µL each), gently inverted, and incubated for less than two minutes. Each sample was measured in triplicate with a fresh 10 µL volume. To prevent hepatocyte damage, stained samples were mixed gently rather than pipetted.
Common co-cultured Liver-on-a-chip (LOC) non-parenchymal cells: Preparation, counting, and viability
Primary human Stellate and Kupffer cells were thawed in a 37 °C water bath, centrifuged at 250 x g for five minutes at room temperature, and then resuspended in NPC* media. Primary human liver endothelial cells were thawed similarly and maintained in T-75 flasks until approximately 85 % confluence.
All three non-parenchymal cell types were stained using equal parts AO/PI (DeNovix, Wilmington, DE) and cells. After mixing quickly, 10 µL of labeled cells were put onto the CellDrop FLi Automated Cell Counter to calculate "%Viablity" and "Live Cells/mL" counts.
The Hepatocyte App was specifically used to assess and count the vitality of Stellate cells.
The Primary Cell AOPI app was used to analyze and count the vitality of Kupffer and Liver Endothelial cells. Kupffer cells (KC) were counted using the default procedure settings, while liver endothelial cells (LC) were counted with modified protocol settings (maximum diameter = 40 µm).
*Denotes modified recipe without ITS+premix (#354352, Corning)
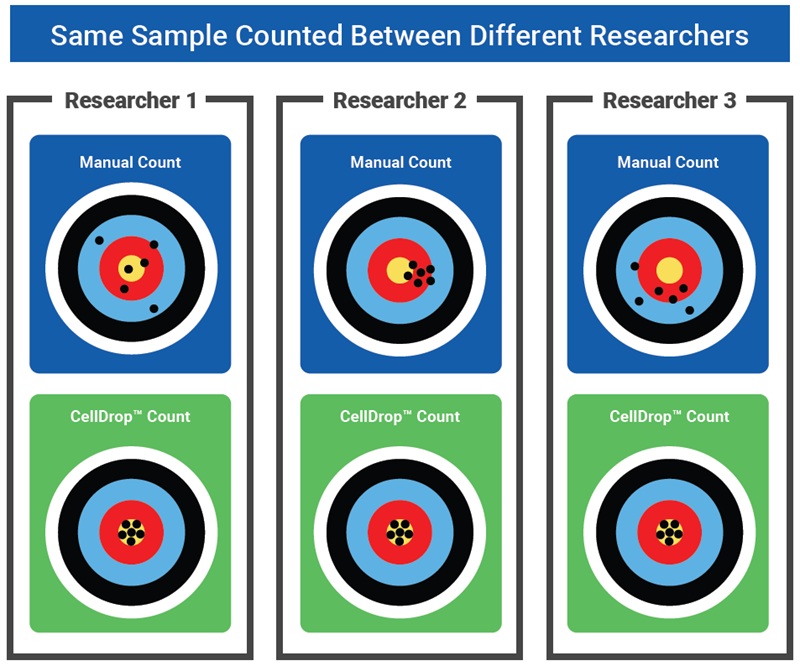
Figure 1. This graphic demonstrates the overarching goal of moving hepatocyte counting from a manual count to an automated count. As different researchers across different labs may have slightly different training, methods, and equipment, accuracy and precision may vary across these manual counts. With all researchers using an automated cell counter with the same optics and counting algorithm, results between researchers, labs, and institutions will become standardized. Image Credit: DeNovix Inc.
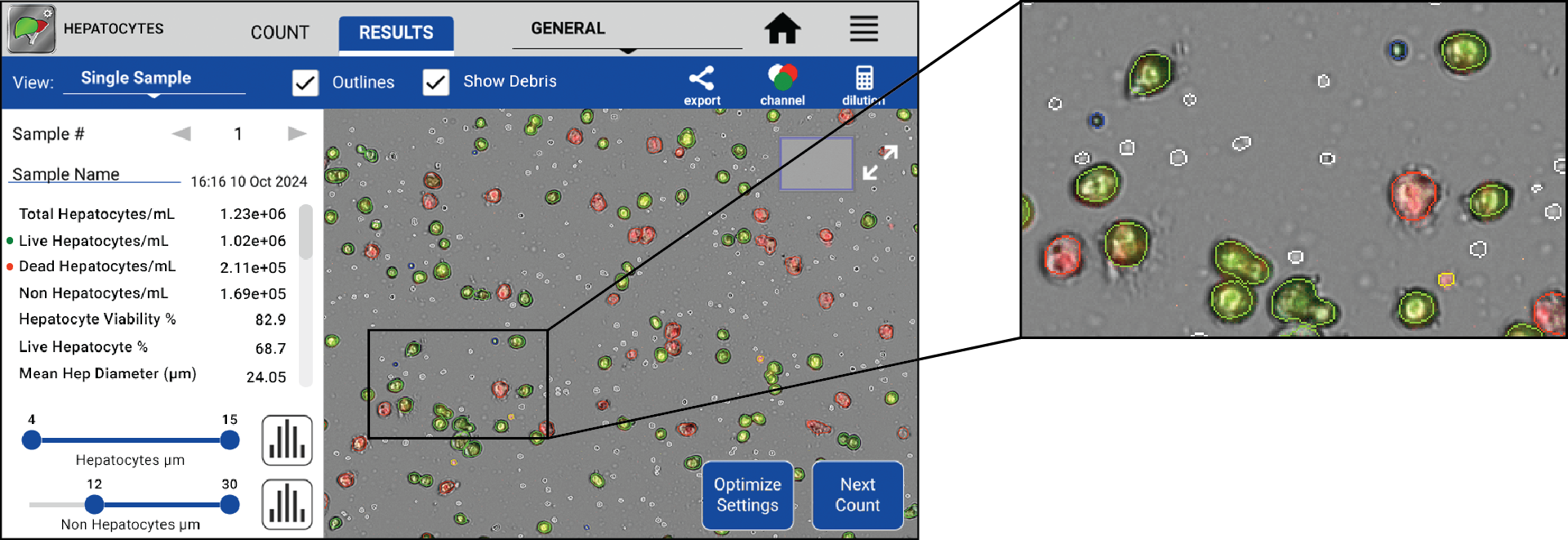
Figure 2. Full-screen view of a freshly isolated mouse hepatocyte sample, with the image showing the counted hepatocyte sample and relevant data. Zoomed in view of image from Figure 2 showing counted live hepatocytes (green), dead hepatocytes (red), live non-parenchymal cells (blue), free nuclei/dead non-parenchymal cells (yellow), and non-cellular debris (white). Image Credit: DeNovix Inc.
Results
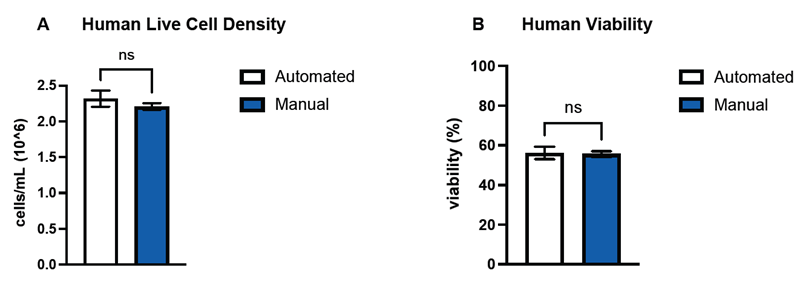
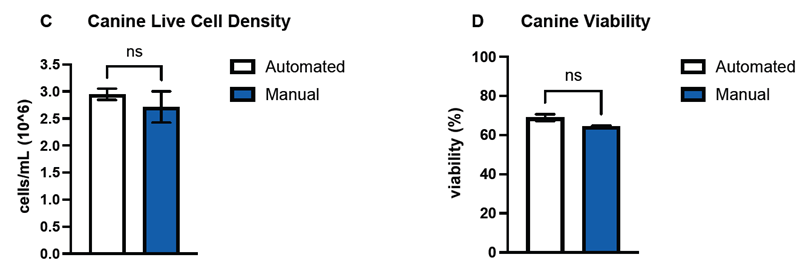
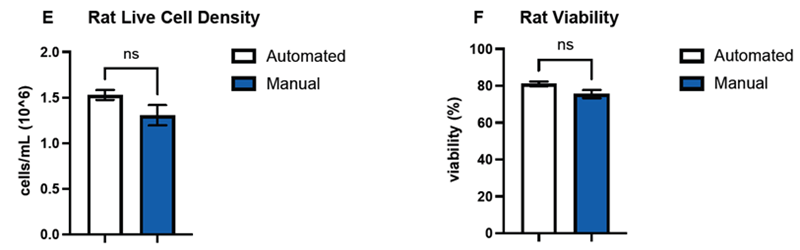
Figure 3. DeNovix confirmed the performance of the machine learning model by counting and determining the viability of hepatocytes of three mammalian species–human (A, B), canine (C, D), and rat (E, F)–and compared the data against hemocytometer counts. Comparisons of the means (n=3) between automated and manual counts for both live cell densities and sample viability were not significant (p > 0.05) across all three species. Error bars represent SEM of the data sets. Image Credit: DeNovix Inc.
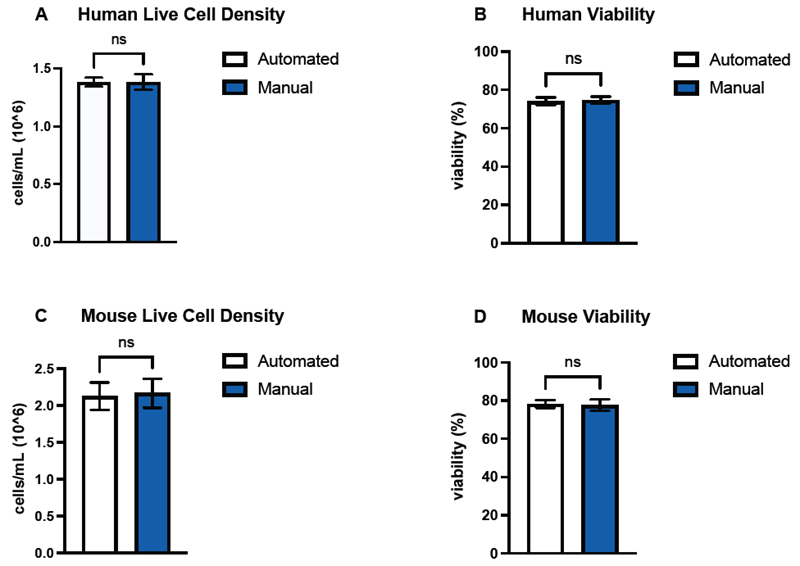
Figure 4. BioIVT verified the performance of the machine learning model by counting and determining the viability of hepatocytes from human (A, B) and mouse (C, D) samples, and comparing the data with hemocytometer counts. Comparisons of the means (n=3) between automated and manual counts for both live cell densities and sample viability were not significant (p > 0.05) across both species. Error bars represent SEM of the data sets. Image Credit: DeNovix Inc.
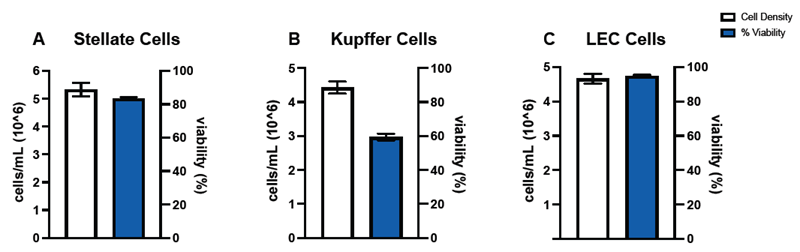
Figure 5. Common co-cultured non-parenchymal cells used on Liver-On-A-Chip platforms were also performance validated on the CellDrop. Cell densities and viability were determined for Stellate (A), Kupffer (B), and Liver Endothelial Cells (C). Across the three cell types, cell density (n=3) showed precision of ≤ 8 % CV and viability precision (n=3) of ≤ 5 % CV. Error bars represent SEM of the data sets. Image Credit: DeNovix Inc.
The results of the sample viability determinations and hepatocyte counts between the counting methods were contrasted using the unpaired two-tailed Welch’s t-test (GraphPad Prism version 10.6.1 for MacOS GraphPad software, Boston, MA).
Calculated p-values for all three species were greater than the alpha value of 0.05. These p-values were also validated by the estimated 95 % confidence intervals for each Welch's t-test performed.
Overall, the hepatocyte count data obtained using the two methodologies revealed no statistically significant differences in mean sample concentration or percent viability.
Conclusion
Automation outperforms human counting in both speed and consistency, particularly when multiple technicians are involved and when there are differences in skill and training within or between labs.
The CellDrop Hepatocytes App also quickly and accurately offers additional information about the sample that manual counting does not, such as the levels of debris and free-floating nuclei, and the ratio of non-parenchymal cells to hepatocytes.
Acknowledgments
Produced using materials originally authored by Daniel Schieffer and David Ash from DeNovix Inc., and Karissa Cottier from BioIVT.
About DeNovix Inc.
Award-Winning Products for Life Science
DeNovix multi-award-winning products include the Reviewers’ Choice Life Science Product of the Year and Platinum Seal awarded- DS-11 Series Spectrophotometers / Fluorometers - with integrated 1 µL UV-Vis, cuvette UV-Vis, and fluorescence - and CellDrop™ Automated Cell Counters for counting cells without slides. Additionally, the DeNovix Squid™ Full Range Pipette is the only electronic pipette that covers the full 1 - 1000 µL range.
These powerful devices integrate patented DeNovix technology with easy-to-use software designed by life scientists for life scientists. Researchers report that they love the industry-leading performance, smartphone-like operation, and the flexible connectivity of the instruments.
When support is needed, the DeNovix team is here to help. DeNovix received the prestigious Life Sciences Customer Service of the Year based on independent reviews posted by scientists worldwide!
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.
Last Updated: May 15, 2026