Introduction
How can genetic variation affect drug metabolism in IBS?
How can genetics influence how IBS treatments work?
Can genetics help predict which treatments will work best for IBS patients?
How close is pharmacogenomics to being used in routine IBS care?
What does the future look like for personalized IBS treatment?
References
Further Reading
Although genetic and microbiome research is beginning to uncover biological drivers of treatment variability in IBS, current evidence highlights that most applications remain exploratory, with limited integration into everyday clinical practice.
 Image credit: Premreuthai/Shutterstockj.com
Image credit: Premreuthai/Shutterstockj.com
Despite decades of research in search of a cure, irritable bowel syndrome (IBS) remains classified as a complex clinical condition. Therefore, pharmacological interventions have primarily adopted a "one-size-fits-all" therapeutic approach, which is often reported to lead to a cycle of trial-and-error prescribing.
Patients are forced to navigate a frustrating number of inconsistent drug efficacy and unexpected adverse events, often at a high personal and economic cost. Recent advances in pharmacogenomics seek to address these past limitations by directly decoding the molecular drivers of inter-individual variability.
The present article synthesizes the latest results from these cutting-edge investigations to show how host genetics and the gut microbiome are being integrated into a precision medicine framework.
It provides evidence that the identification of specific genetic signatures is increasingly being leveraged to develop predictive models of treatment response. However, it is important to note that most genetic associations in IBS remain exploratory and are not yet routinely used to guide clinical decision-making.9
Introduction
Irritable bowel syndrome (IBS) is a mechanistically complex and increasingly prevalent disorder of gut-brain interaction (DGBI), currently estimated to affect between 7 % and 21 % of the global population.1
Since its discovery in 1820, the condition has traditionally been managed through symptom-based exclusion. Unfortunately, studies show placebo response rates range from 30-35 % (empirical trial data), highlighting that IBS treatment has historically been a "one-size-fits-all" cycle of trial-and-error.2
Recent technological advances, particularly in computational power and next-generation “multi-omics” assays, have driven the emergence of precision medicine. This approach in patient care aims to mitigate traditional therapeutic limitations by sub-grouping patients based on their molecular traits, thereby biologically guiding intervention selection and overall treatment efficacy.
What is IBS? Symptoms and treatment | NHS
Video credit: NHS/Youtube.com
Recent research has established that the efficacy of pharmacological interventions in IBS is partly influenced by the cytochrome P450 (CYP) enzyme system, particularly for centrally acting neuromodulators.10
Within the CYP system, CYP2D6 is considered a critically important enzyme as it metabolizes tricyclic antidepressants (TCAs) such as amitriptyline and nortriptyline, which are often prescribed for the management of visceral pain. Genetic association studies (GWAS) have identified four distinct CYP2D6 phenotypes: poor, intermediate, normal, and ultrarapid metabolizers.4
Poor metabolizers (5-10 % of the White population; ~1 % of the Asian population) have been found to lack functional enzyme activity. This results in elevated drug plasma levels and increased risks of dose-dependent toxicities like cardiac arrhythmias or panic attacks.2
Conversely, ultrarapid metabolizers have been observed to metabolize these pharmacological agents too quickly, often leading to therapeutic failure unless dosage is substantially increased.5
Clinical Pharmacogenetics Implementation Consortium (CPIC) guidelines support genotype-informed dosing adjustments for TCAs based on CYP2D6 and CYP2C19 status, although such testing is not IBS-specific and is not yet standard in routine IBS care.4
How Can Genetics Influence How IBS Treatments Work?
Research reveals that genetic polymorphisms can significantly alter pharmacodynamic responses, particularly in pathways such as the serotonin transporter (SERT) and receptor pathways, which directly influence drug targets.2,4,5
For example, the SLC6A4 gene, which encodes SERT, contains a functional polymorphism known as 5-HTTLPR. This functional polymorphism consists of two main variants: 1. the "long" (L) variant is associated with more efficient serotonin reuptake, and 2. the "short" (S) variant, which is linked to reduced SERT synthesis and increased serotonergic activation.2 However, evidence linking SLC6A4 polymorphisms to treatment response remains heterogeneous and has not been consistently replicated across large clinical trials.9
Reviews have analysed clinical data that indicate that patients with the L/L genotype exhibit a superior response to alosetron, a 5-HT3 receptor antagonist used in diarrhea-predominant IBS (IBS-D) to slow colonic transit.2 In contrast, the S/S genotype is associated with a diminished response to 5-HT3 antagonists but a potentially enhanced response to 5-HT4 agonists, such as tegaserod, used in constipation-predominant IBS.2
Need to save this article to read later? Download your free PDF version by clicking here.
Can Genetics Help Predict Which Treatments Will Work Best for IBS Patients?
Current research aims to identify specific genetic markers that can stratify patients into responders for subsequent pharmacological or dietary interventions.6 Balsiger and colleagues’ (2022) pilot study of 390 IBS patients found that the rs2020934G allele in the SLC6A4 gene was a significant predictor of response to the antispasmodic drug otilonium bromide (p = 9.1 x 10-5). This finding is derived from a relatively small, exploratory cohort and requires external validation before clinical implementation.6
The study further reported that responders to a low FODMAP diet were more likely to carry alleles in immune-related genes, such as the CCR3 rs7617872A variant (p = 2.2 x 10-6), which was associated with increased blood-based chemokine receptor expression. At present, there are no validated genetic tests routinely recommended to guide dietary therapy selection in IBS.6
Together, these findings suggest that a patient's genetic predisposition can objectively guide clinicians’ intervention selection between medication and restrictive dieting.6
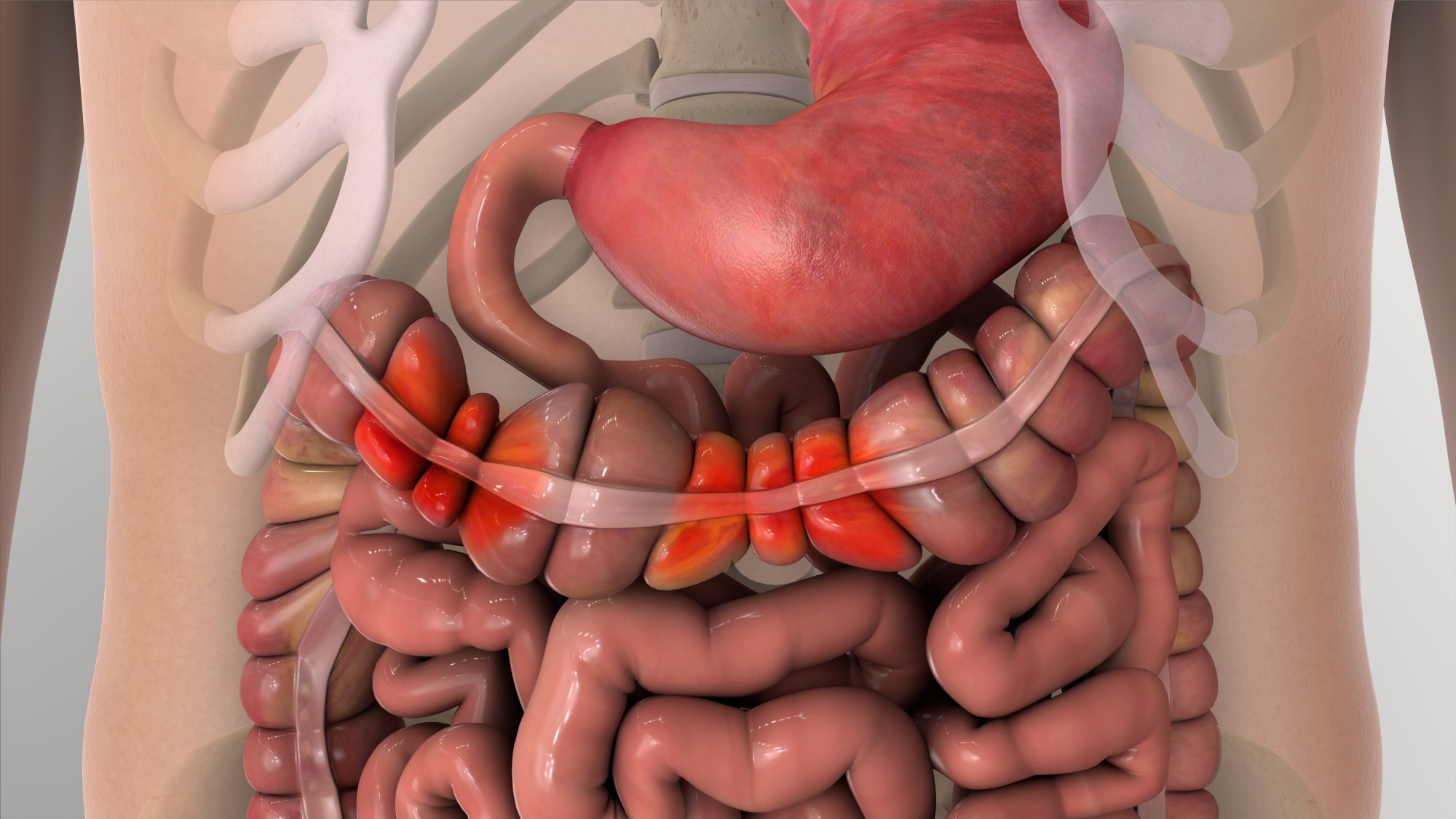 Image credit: Nathan Devery/Shutterstock.com
Image credit: Nathan Devery/Shutterstock.com
How Close is Pharmacogenomics to Being Used in Routine IBS Care?
Singh and colleagues (2025) conducted a multicenter, double-blind trial involving 238 participants to investigate whether individualized IgG antibody profiles could guide dietary elimination. The study found that 59.6 % of patients in the guided elimination group achieved the FDA-standard target for abdominal pain reduction, significantly outperforming the 42.1 % observed in the sham diet group. However, major gastroenterology societies currently do not recommend IgG-based food sensitivity testing for IBS due to insufficient evidence and concerns about biological plausibility.7
The study highlighted that diagnostic-guided strategies may offer a more sustainable alternative to broad dietary restrictions, as patients were typically required to eliminate only 2 to 4 specific trigger foods to achieve significant symptom relief.7,8
Despite this growing body of physiological evidence underscoring the benefits of pharmacogenomics in precision medicine, the transition of these techniques into routine IBS care has been hindered by significant regulatory and economic hurdles. Shekhani and colleagues’ (2019) comparative analysis of the FDA and EMA product labels for 54 drugs with known actionable gene-drug interactions found that these agencies agreed on actionable labeling in only 54 % of investigated cases.7,9
Furthermore, systematic reviews have shown that clinicians often encounter challenges with "number needed to harm" (NNH) metrics. For example, the NNH for alosetron is 14 due to the risk of ischemic colitis, while tenapanor has an NNH of 16, primarily due to the drug's association with severe diarrhea.2
These safety risks, combined with uncertain insurance reimbursement and a lack of rapid point-of-care genotyping, are currently cited as the primary limitation to the "preemptive" use of genetic testing in primary care settings.3
What Does the Future Look Like for Personalized IBS Treatment?
Multi-omics approaches combining genomics, microbiome profiling, and environmental exposures are increasingly recognized as essential for understanding IBS heterogeneity and improving treatment stratification.1
Research by the Korean Society for Laboratory Medicine suggests that the next frontier of personalized IBS care lies in integrating host genetics with the gut microbiome. Metagenomic profiling has already identified that lower baseline abundances of Roseburia and Subdoligranulum (metabolic signatures) may predict favorable responses to fecal microbiota transplantation (FMT) in moderate to severe IBS.10
Consequently, the goal of current research is to leverage advances in artificial intelligence (AI) to compute the vast datasets generated through multi-omics sequencing approaches, thereby shifting IBS interventions from reactive symptom management to proactive, molecularly targeted treatments.2
Nonetheless, these approaches remain largely investigational, and significant validation, standardization, and cost-effectiveness analyses are required before widespread clinical adoption.1
References
- Zhao, X., Yang, Y., Liu, Z., Sun, R., & Suo, A. (2026). Identifying chemicals associated with irritable bowel syndrome by integrating a transcriptome-wide association study with chemical-gene-interaction analysis. PLOS One, 21(2), e0340666. DOI:10.1371/journal.pone.0340666, https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0340666
- Lacy, B. E., Saleh, H., & Cangemi, D. J. (2025). Precision medicine in disorders of gut-brain interaction. Gastroenterology & Hepatology, 20(10). https://www.gastroenterologyandhepatology.net/archives/october-2025/precision-medicine-in-disorders-of-gut-brain-interaction/
- Soufan, F., Ghosson, A., Jaber, R., Ghandour, A., & Uwishema, O. (2025). The Gut‐Brain Axis in Irritable Bowel Syndrome: Implementing the Role of Microbiota and Neuroimmune Interaction in Personalized Prevention - A Narrative Review. Health Science Reports, 8(4). DOI:10.1002/hsr2.70660, https://onlinelibrary.wiley.com/doi/10.1002/hsr2.70660
- Wang, X. J., & Camilleri, M. (2019). Personalized medicine in functional gastrointestinal disorders: Understanding pathogenesis to increase diagnostic and treatment efficacy. World Journal of Gastroenterology, 25(10), 1185–1196. DOI:10.3748/wjg.v25.i10.1185, https://www.wjgnet.com/1007-9327/full/v25/i10/1185.htm
- Hicks, J., et al. (2017). Clinical pharmacogenetics implementation consortium guideline (CPIC) for CYP2D6 and CYP2C19 genotypes and dosing of tricyclic antidepressants: 2016 update. Clinical Pharmacology & Therapeutics, 102(1), 37–44. DOI:10.1002/cpt.597, https://ascpt.onlinelibrary.wiley.com/doi/10.1002/cpt.597
- Balsiger, L. M., et al. (2022). Genetic analyses of treatment response in primary care IBS, a pilot study. Gut, 72(9), 1803–1804. DOI:10.1136/gutjnl-2022-328430, https://gut.bmj.com/content/72/9/1803
- Singh, P., et al. (2025). A Novel, IBS-Specific IgG ELISA-Based Elimination Diet in Irritable Bowel Syndrome: A Randomized, Sham-Controlled Trial. Gastroenterology, 168(6), 1128-1136.e4. DOI:10.1053/j.gastro.2025.01.223, https://doi.org/10.1053/j.gastro.2025.01.223
- Biomerica. (2025, November 25). Biomerica’s inFoods IBS product featured in Biotherapeutic’s Quarterly highlighting a new precision approach for treating IBS. https://investors.biomerica.com/news/news-details/2025/Biomericas-inFoods-IBS-Product-Featured-in-Biotherapeutics-Quarterly-a-Henry-Schein-Publication-Highlighting-a-New-Precision-Approach-for-Treating-IBS/default.aspx. Accessed 20th March 2026
- Shekhani, R., Steinacher, L., Swen, J. J., & Ingelman‐Sundberg, M. (2019). Evaluation of Current Regulation and Guidelines of Pharmacogenomic Drug Labels: Opportunities for Improvements. Clinical Pharmacology & Therapeutics, 107(5), 1240–1255. DOI:10.1002/cpt.1720, https://ascpt.onlinelibrary.wiley.com/doi/10.1002/cpt.1720
- Rim, J. H., et al. (2024). Clinical Pharmacogenetic Testing and Application: 2024 Updated Guidelines by the Korean Society for Laboratory Medicine. Annals of Laboratory Medicine, 45(2), 121–132. DOI:10.3343/alm.2024.0572, https://www.annlabmed.org/journal/view.html?doi=10.3343/alm.2024.0572
Further Reading
Last Updated: Mar 23, 2026