The early diagnosis of most types of skin cancer is directly related to improved survival rates. The DermaSensor device is a revolutionary, non-invasive and spectroscopy-based tool that can assist clinicians in early skin cancer diagnosis.
An overview of skin cancer
As the most common type of cancer to affect individuals within both the United States and around the world, it is estimated that 1 in 5 Americans will develop skin cancer by the time they reach the age of 70. Each day, over 9,500 people in the United States alone are diagnosed with skin cancer.
There are four primary types of skin cancer: basal cell carcinoma (BCC), squamous cell carcinoma (SCC), Merkel cell cancer, and melanoma. Of these four skin cancer types, basal cell carcinoma accounts for approximately 80% of all skin cancer cases.
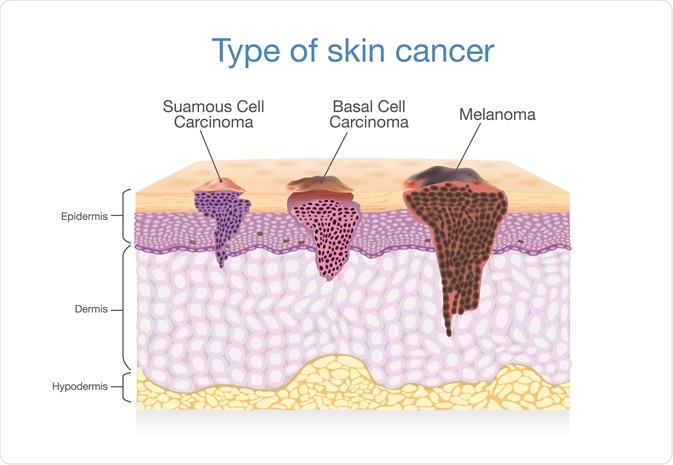 Three of the main types of skin cancer and how they relate to the skin layers. Image Credit: solar22 / Shutterstock.com
Three of the main types of skin cancer and how they relate to the skin layers. Image Credit: solar22 / Shutterstock.com
Current skin cancer detection methods
Skin cancer detection often begins with a visual examination, which can be performed by both the patient and their doctor. Since skin cancer can affect so many people of all ages and ethnicities worldwide, the Skin Cancer Foundation and the American Cancer Society recommend that each person performs a self-examination of their entire skin surface once a month.
This monthly skin self-exam should involve a careful examination of the entire body, during which the individual should note any patterns of moles, blemishes, freckles and other marks present on their skin. Any new, enlarged, changing, crusting, bleeding spots, bumps, patches, and moles can be a sign of skin cancer.
If a patient identifies something suspicious on their skin, it is recommended that they have this spot-checked by a doctor. The patient’s primary care provider (PCP) will examine the suspicious spot and note its size, shape, color and texture, and whether there is any active bleeding or scaling of the lesion. Following his/her examination, the PCP will likely refer the patient to a dermatologist who can perform more specialized tests to make a definitive diagnosis.
Limitations
Out of the nearly 8 million new skin cancer diagnoses that are made each year worldwide, it is estimated that more than 60% of skin concerns are first evaluated by non-specialists such as PCPs. Since many PCPs are not trained to provide a skin cancer diagnosis, these clinicians will typically refer the patient to a dermatologist.
The need to refer patients to these specialists can cause a lapse in time between appointments and dermatologists will often need to perform biopsies of suspicious lesions, which will need to be further examined by a pathologist before making a final diagnosis. An early diagnosis of skin cancer is associated with high curability rates.
Therefore, such delays have the potential to affect patient outcomes. When biopsied tissue samples are found to be benign, many health insurance companies will offer poor reimbursement rates, leaving the patient with a large bill to pay.
 Image Credit: Iryna Kalamurza / Shutterstock.com
Image Credit: Iryna Kalamurza / Shutterstock.com
The future of DermaSensor
The DermaSensor was created to help overcome the challenges associated with conventional skin cancer diagnosis procedures.
The DermaSensor is a handheld objective skin cancer sensing device that utilizes both pulses and light and spectroscopy to non-invasively identify information about a skin lesion at the subcellular level. More specifically, this device uses Elastic Scattering Spectroscopy (ESS), which measures and records photon scattering patterns as they reflect off different cellular structures following the input of quick bursts of light.
ESS technology has been validated in more than 30 clinical publications that have demonstrated this technique's utility to analyze the macroscopic structure of both cellular and subcellular particles. Since malignant lesions scatter light at different intensities, the DermaSensor algorithm, which is derived from thousands of spectral samples of pathologically verified lesions, will immediately categorize a skin lesion as “Higher Risk” or “Lower Risk.”
The DermaSensor technology company has conducted several clinical studies to confirm the efficacy of their device. The most recent of these studies includes the DERM-ASSESS II study, a multi-center study that confirmed both the safety of the DermaSensor device and its sensitivity for detecting skin cancer.
In terms of safety, the DERM-ASSESS II study did not report any adverse effects following the use of the DermaSensor, confirming this non-invasive device's safety.
When used to diagnose melanoma, the DermaSensor was found to have a sensitivity of 100%, comparable to that of the study dermatologists, whose sensitivity for diagnosing melanoma was 90.9%.
When used to diagnose Non-Melanoma Skin Cancers (NMSCs), including BCC and SCC, the DermaSensor reported a sensitivity of 93.3%, as compared to that of the study dermatologists with a sensitivity of 98.3%.
Conclusion
The lack of statistically significant differences between the sensitivity of the study dermatologists and that of the DermaSensor confirms the strong and accurate performance capabilities of this device when compared to that of a trained specialist.
As compared to the high sensitivity rate that is reported for dermatologists in their diagnosis of various types of skin cancers, which is typically in the range of 82% up to 100%, PCPs typically have a much lower sensitivity rate of approximately 71%. This sensitivity rate of PCPs is significantly less than that of the DermaSensor, which was reported to be 94.3% by the DERM-ASSESS II study.
Therefore, while the DermaSensor is not intended to replace a clinician’s decision-making process, it is an adjunctive tool that can provide PCPs with additional information on suspicious lesions that will help them determine whether a referral to a dermatologist is warranted.
It should be noted that the DermaSensor is not equipped with the ability to determine skin cancer conclusively. If a skin lesion is identified as “Higher Risk,” the DermaSensor cannot specify what type of cancer may be present. The DermaSensor allows PCPs to make more confident decisions on suspicious skin spots to protect the patient from unnecessary morbidity and provide more accurate specialist referrals as needed.
References
Further Reading
Last Updated: Jan 22, 2021