Advanced Cell Diagnostics Inc. (ACD), a technology and market leader in the field of molecular pathology and developer of cell and tissue-based RNA analysis tools, is celebrating a significant milestone. Over 300 peer-reviewed research papers have now been published that feature ACD’s RNAscope technology as a core part of the research methodology, reflecting its facility in advancing the latest biological research in both academia and industry.
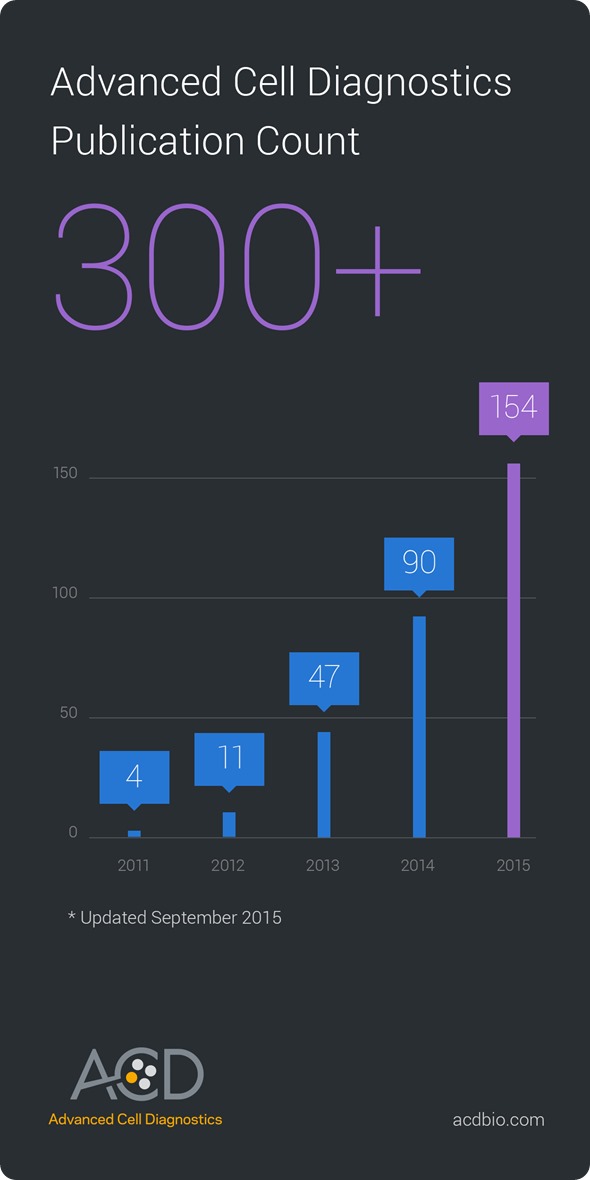
RNA in situ hybridization (ISH) is a valuable tool for studying spatial and cell-specific gene expression, while preserving tissue architecture. ACD’s RNAscope represents a breakthrough advance in RNA ISH, implementing a proprietary signal amplification system to enhance both sensitivity and specificity, producing a high-performance assay that is also quick and easy to use. As awareness has spread of how RNAscope ISH can accelerate a variety of studies, its rise in uptake has translated to a rapid increase in published research. Thus far in 2015, the number of new publications has already exceeded 150, more than the total number of publications from all the previous years combined, signaling acceleration in technology adoption.
Chief Scientific Officer, Dr. Xiao-Jun Ma, commented:
This rapid increase in publications speaks directly to the value of RNAscope technology in many fields of research. Spatially mapped single-cell gene expression data are crucial to understanding the development, structure and function of complex tissues at the molecular and cellular level. However, obtaining such data has been a huge bottleneck due to technical difficulties of traditional RNA ISH methods and the loss of contextual information with grind-and-bind assays. With RNAscope, such data can be generated within weeks from a concept to publication quality images.”
The papers range from basic research in developmental biology, neuroscience, stem cells and ophthalmology, to translational research and diagnostic development of biomarkers in cancer and infectious diseases, in respected journals such as Nature, Science, Cell, New England Journal of Medicine, PNAS, PLoS One, and Clinical Cancer Research.