What environmental toxin did your recent research focus on and what prompted you to study whether dietary exposure to this toxin could trigger neurofibrillary tangles (NFT) and beta-amyloid plaques in the brain?
Many Chamorro villagers on the island of Guam perished from a puzzling paralytic disease that combines aspects of ALS, Alzheimer's, and Parkinson's disease. The brain tissues of those who die are replete with neurofibrillary tangles and beta- amyloid plaques, similar in structure to those of Alzheimer's patients.
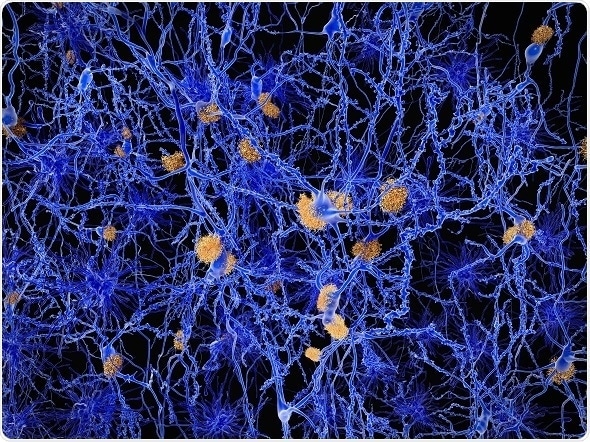
Alzheimer disease neuron network with amylod plaques. Image credit: Juan Gaertner / Shutterstock.com
Some traditional Chamorro food items contain the cyanobacterial toxin BMAA which has been linked to the disease. We sought to gauge the effects of chronic dietary exposure to BMAA.
What diseases are NFTs and beta-amyloid plaques associated with?
Guam ALS/PDC and Alzheimer's disease.
Why were vervets chosen as an animal model for this research?
Vervets are an exotic species on the Caribbean island of St. Kitts introduced over a century ago. Previous researchers have found all of the St. Kitts vervets to be homozygous for the ApoE4 gene. In humans ApoE4 is associated with an increased risk of developing cerebral Aβ-amyloid angiopathy associated with Alzheimer's disease.
Which regions of the vervets’ brains were investigated for neuropathology and why?
Fourteen regions of each vervet’s brain were investigated for neuropathology including:
- The Superior Frontal Gyrus
- Anterior Cingulate Gyrus
- Insula Temporopolar Cortex (Dorsal)
- Temporopolar Cortex (Ventral)
- Entorhinal Cortex (Anterior)
- Entorhinal Cortex (Posterior)
- Perirhinal Cortex
- Occipital Cortex
- Primary Motor Cortex
- Paralaminar Nucleus of the Amygdala
- Dentate Gyrus
- Caudate Nucleus
- Substantia Nigra
These regions were selected for study by Dr. David Davis and Dr. Deborah Mash of the Miami Brain Endowment Bank because of the presence or absence of neuropathology in similar regions of brains of human neurodegenerative patients.

Image credit: Huron Digital Pathology.
What techniques were used to study the brain hemispheres?
Using the multibrain services of NeuroScience Associates in Tennessee each hemisphere was freeze-sectioned at 40 microns and stained with AT8 IHC stain with a Nissl counterstain for hyperphosphorylated tau or with beta-amyloid (1-42) IHC stain for amyloid deposits. Thioflavine-S with a thinonine Nissl counterstain was used to confirm the presence of neurofibrillary tangles (NFT) and amyloid deposits.
In the first experiment, NFT and amyloid deposits, identified in a review of blinded sections, were hand counted in three sections in series from non-overlapping brain regions.
Why did you choose to use the Tissue Scope LE and how did this aid your research?
The manual counts in the tissues from the first experiment was a very laborious procedure, so for the replication experiment, our neuroanatomist, Dr. David Davis and neuroscientist Dr. Deborah Mash suggested that we use an automated system to quantitate NFT. Given the importance of our results, this would also help to remove subjectivity.
They recommended the Tissue Scope LE. Huron Digital Pathology was very helpful in getting the system to the Miami Brain Endowment Bank and in getting it up and running for us.
There are many advantages to this system, and we will continue to use it for our future publications. We think it offers a lot of advantages in quantitation over manual counts.
Huron Digital Pathology Innovative Slide Holder
What were the main findings of your research?
We found that chronic dietary exposure to BMAA triggered the formation of NFT and amyloid deposits in the brain. This tends to confirm the hypothesis that BMAA in the Chamorro diet causes the Guam disease.
Both of these neuropathological features had never been before seen in a single animal model. To our knowledge, this is the first time that a tauopathy has ever been triggered in an animal model.
Were you surprised by your results and what impact do you think they will have?
We were surprised to visualize both NFT and amyloid plaques in the brain tissues of vervets fed fruit dosed with BMAA. Not only do these results support the BMAA theory of Guam ALS/PDC, this new model may also lead to a new way to rapidly screen candidate drugs for Alzheimer's disease at a fraction of the time and price it currently takes,
What further research is needed to understand the role of environmental toxins in triggering NFTs and amyloid plaques in the brain?
We would like to better understand the mechanism by which BMAA triggers such a major tauopathy. We also found that supplementing the vervet diet with the dietary amino acid L-serine significantly reduces the density of NFT.
We would like to better understand the mechanisms by which L-serine is neuroprotective. This could help us in our current human clinical trials of L-serine for ALS patients.
Where can readers find more information?
- Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain. Paul Alan Cox, David A. Davis, Deborah C. Mash, James S. Metcalf, Sandra Anne Banack. Proc. Royl. Soc. B 2016 283 20152397; DOI: 10.1098/rspb.2015.2397. Published 20 January 2016
- https://brainchemistrylabs.org/
- http://brainbank.med.miami.edu
About Dr Paul Alan Cox
After receiving his Ph.D. from Harvard, Paul Alan Cox was appointed a Miller Fellow at the Miller Institute for Basic Research in Science at the University of California, Berkeley and subsequently as Melbourne University Research Fellow at the University of Melbourne in Australia. He served as Professor and Dean At Brigham Young University in Utah, as the King Carl XVI Gustaf Professor at Uppsala and the Swedish Agricultural University, and as Director of the National Tropical Botanical Gardens in the USA.
Cox was named by TIME magazine as one of eleven "Heroes of Medicine" and was awarded the Goldman Environmental Prize for rain forest conservation. At the Brain Chemistry Labs of the Institute for Ethnomedicine in Jackson Hole, Wyoming, Cox and his colleagues search for cures for progressive neurodegenerative illnesses.