Firstly, a sample must be transported from the clinic to the laboratory. Simple tests may be undertaken in a small lab within the hospital or clinic, whilst complex testing such as drug susceptibility testing is often done in a large centralized laboratory many miles from the clinic.
Testing is then undertaken using several different large, expensive and slow pieces of equipment.
Analysis is the final time-consuming step before the result can be returned to a health professional. The patient must then return to the clinic to be given their result, and the whole process begins again if further testing is needed.

What has this meant in terms of the number of countries able to benefit from DNA analysis?
Resource-scarce settings in particular do not have access to DNA analysis, as much of the current equipment and reagents require stable electricity, a temperature controlled environment, clean water and cold chain transportation.
Another barrier is the skill set required to run the tests, particularly for rural areas which struggle as skilled workers move to the cities.
How powerful can DNA analysis be?
DNA analysis can answer a range of questions from “what species of plant is this?” to “what is in this water?” to “what hereditary mutations are causing this family’s cancer?”.
We are dedicated to using our handheld molecular lab, Q-POC, to tackle another pressing question: “which drug will fight this infection?”.
Antibiotic resistance has been increasingly hitting the headlines, with British Prime Minister David Cameron warning that if we fail to act, we’ll be back in the dark ages of medicine.
DNA analysis can tell a healthworker which of the drugs in their arsenal will best fight an infection; this enables us to save the most powerful and/or expensive antibiotics for when they’re really needed, and also enables healthworkers and patients to be confident in their use of lower cost frontline antibiotics, when appropriate.
Please can you outline QuantuMDx’s mission to develop a handheld molecular diagnostics device?
In order to make molecular diagnostics accessible to all, the technology needs to be able to be used anywhere by anyone. This means battery-run, this means intuitive to use, this means no clean water requirement and no complex manual sample preparation.
Once we had developed a specification of the device, shaped by my experiences in healthcare in the UK and co-founder Jonathan O’Halloran’s experiences running diagnostics in South Africa, we set about finding or developing the individual component technologies that would make our vision of ‘diagnostics for all’, a reality.
At what stage of development is Q-POCTM currently at?
Q-POC is at prototype stage. Our research teams are optimizing and integrating each component, as well as developing a range of infectious disease detection and drug resistance assays alongside companion diagnostics assays. We anticipate commercialization of Q-POC in 2017/18.
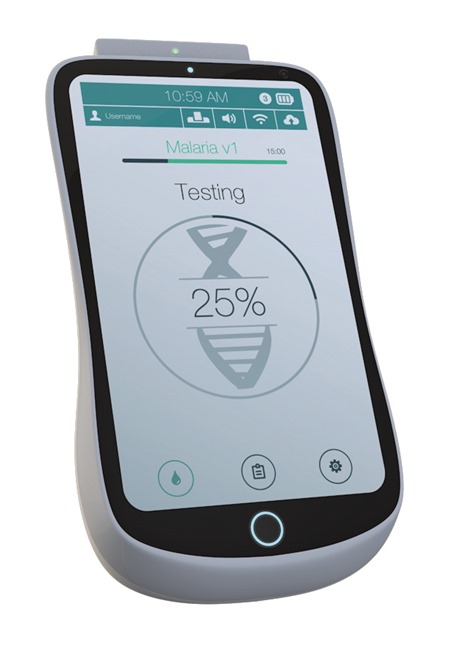
How rapid do you think DNA analysis could potentially be?
We’re currently working to ten to twenty minute turnaround (depending on the complexity of the test) - that’s from inputting the unprocessed clinical sample into the device to reading the onscreen result.
What impact do you think the device will have in terms of addressing humanitarian health challenges?
70% of people with extremely drug resistant tuberculosis (XDR) die within a month of their diagnosis. Rapid and accurate identification of drug resistant infection in the field is crucial in providing proper treatment as well as for containment, for protection of the patient’s friends, family and community.
Q-POC will help to address health inequalities within richer countries too. In the UK and USA, individuals on the anticoagulant drug warfarin have an improved outcome if they are cared for at a better resourced clinic. This is likely due to more careful monitoring of their reaction to treatment.
In clinics with fewer resources, Q-POC could run a pharmacogenomics test to determine the patient’s dosage requirement, enabling them to receive the right dose first time and negating the need for as stringent monitoring.
What do you think the future holds for molecular diagnostics?
Just as we’ve seen an increased uptake in DNA sequencing as the cost decreases, so will we see a democratization and proliferation of DNA analysis as the cost and user skillset requirement decreases.
What are the main hurdles that will need to be overcome?
Obviously the development of new technology comes with its challenges, that’s part of the fun! Regulatory approval is stringent for technologies like ours.
However probably the largest barrier to getting novel diagnostic technologies into clinics is the large body of evidence that must be collected to support its use. We’re working with a range of organizations, academic and NGO, in order to begin this work.
How do QuantuMDx plan to contribute to the future of molecular diagnostics?
We’re looking to lead the way in taking molecular diagnostics out of the laboratory and into the community.
Where can readers find more information?
More information can be found at our website, www.quantumdx.com, or questions can be emailed to [email protected].
About Elaine Warburton, OBE
Elaine is an entrepreneur with over 25 years’ healthcare and biotechnology experience. With an eclectic background in nursing, genetics, accounting and running hospitals, she currently heads up pioneering British life sciences tools and diagnostics company QuantuMDx Group.
QuantuMDx is developing Q-POC, a low cost handheld molecular lab with inbuilt DNA sequencing to address humanitarian health challenges and detect & track emerging pandemics in real-time across developing and developed nations through the Internet of Life™.
The first assays to be commercialized in 2017 are 10-20 minute infectious disease/drug resistance testing and companion diagnostics. Elaine was awarded an OBE in the 2014 Queen’s New Year Honors for services to Innovation in Healthcare.