Brain concussion occurs when the soft cottage cheese-like brain tissue is slammed against the hard skull surface, sustaining transient and reversible injury which causes brain dysfunction for a period. A new study shows that the mechanism of brain injury in these patients is vascular, rather than related to the nerve cells themselves. This could change the way such patients are treated in the future.
The brain may be injured in many traumatic situations, such as sports collisions, vehicular accidents or in fights. The sequelae of brain injuries may be both severe and chronic. One such type of traumatic brain injury (TBI) is the concussion. When a severe concussion occurs, the patient may well suffer cognitive dysfunction, that is, may not be able to think lucidly, remember some things, move freely or regulate emotional states. The reason for such changes is unknown.
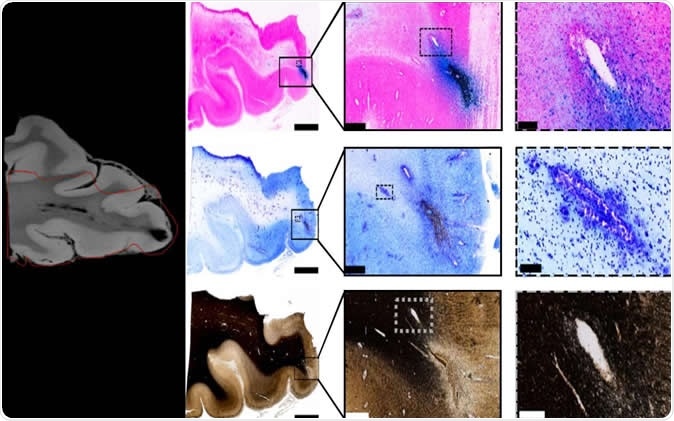
Tissue imaging confirms blood vessel damage in traumatic brain injury patients. Image Credit: Mitra lab/CSHL, 2019
The study – and what it shows
The current study aimed to explore the theory that concussion is caused by damage to the nerve axons because of blunt injury force to the nerve cells. The researchers performed imaging studies using magnetic resonance imaging scanning (MRI scanning), which relies on the pattern of energy release by excited protons within a strong magnetic field when exposed to and then released from a radiofrequency current. They followed this up with postmortem examinations of the same brains, using a high-throughput strategy to analyze the histological profile of the brain tissue. This workflow uses multiple techniques to produce images that label virtual cross-sections of the brain, or brain ‘slices’, and was originally developed to study mouse brain connections.
They found that the predominant damage caused by TBI was to blood vessels rather than to the nerve cells themselves. The post mortem examination confirmed this finding.
An MRI scan produces detailed 3-D images of soft tissue, based on proton excitation and de-excitation, with consequent energy release. The body is first subjected to a strong magnetic field that aligns each one of the trillions of protons to the field. The next step is to use a radiofrequency field to excite the protons, which are forced out of alignment and rotate through 90 or 180 degrees. When the RF field is withdrawn, the protons swing back into their aligned state in accordance with the magnetic field. As they do so they release varying amounts of energy, depending on the surroundings and the molecules in the field. These bursts of energy are interpreted to produce images of what kind of tissue the scanner is viewing.
However, MRI scanning only allows structures a few hundred microns apart to be seen separately, and this resolution is not high enough to distinguish between injuries to nerve fibers or axons and injuries to blood vessels. To solve this, the researchers imaged physical slices of the same brain after the patient died, and digitally analyzed them at resolutions as fine as a single micron. The MRI and digital analysis images were compared to detect vascular injury on the MRI scan more accurately.
The MRI results showed small dark patches, looking like “black blobs”, around any physical damage that had occurred as a result of the trauma. These were examined by the scientists using iron stain, to detect blood, as well as a myelin stain, to detect broken-up axonal tissue, on the physical slices of the postmortem brain. They observed that the area of the physical damage, or lesion, was crisscrossed by iron-containing stained areas, representing bleeding, and hence vascular injury. They found that this was due to microbleeds from breakages in the microscopic blood vessels in the brain, and these occurred along the course of the vessel. The myelin stain failed to show up any significant damage to the nerve fibers in the area of the lesion.
Of course, this doesn’t mean that there is no axonal injury in cases of TBI. However, the researchers did conclude that vascular injury as a result of brain trauma is characteristic in these patients, and is shown in the form of traumatic microbleeds.
Traumatic microbleeds were also found to be associated with a higher chance of development of worse future outcomes including disability in these patients. However, there are two possible explanations: either the patient with microbleeds suffered more severe TBI accounting for the worse outcome, or the microbleed itself is a factor that causes increased brain damage compared to cases where these are absent.
Implications
This indicates that more study is required to find out which of these hypotheses fits the observations best, to find out what actually causes TBI, and how it produces future adverse health outcomes in this patient subset. This could serve to focus future attempts to mitigate brain damage in these patients by appropriate prevention and treatment of vascular injury. The benefits of such experiments would help treat patients with TBI in a more focused way, and to increase the extent of recovery in such patients.
Researcher Partha Mitra says, “Nerve damage following traumatic brain injuries has been a majority point of view, and therapy as well as drug development has been targeted towards that. The idea is that if the mechanism is actually different, therapeutic intervention may also be different.”
Journal reference:
Allison D Griffin, L Christine Turtzo, Gunjan Y Parikh, Alexander Tolpygo, Zachary Lodato, Anita D Moses, Govind Nair, Daniel P Perl, Nancy A Edwards, Bernard J Dardzinski, Regina C Armstrong, Abhik Ray-Chaudhury, Partha P Mitra, Lawrence L Latour, Traumatic microbleeds suggest vascular injury and predict disability in traumatic brain injury, Brain, , awz290, https://doi.org/10.1093/brain/awz290