The ongoing COVID-19 pandemic has cost hundreds of thousands of human lives, besides more than 11 million cases. Caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the disease was initially thought to be more severe in the elderly and those with pre-existing medical conditions. However, it started to manifest in children after the peak of the first wave.
Most cases in the pandemic have occurred among adults. In fact, scientists thought that children would not get the disease or would have a mostly asymptomatic disease. Now, a new study published on the preprint medRxiv* in July 2020 reports clusters of a new hyperinflammatory condition seen among children living in areas with epidemic disease. The study deals with the disease process and the contribution of immunity to the clinical features.
The condition manifests with fever, rash, conjunctivitis., mucocutaneous rash, and cardiac complications. At first, doctors diagnosed it as Kawasaki disease (KD), which is an acute vasculitis syndrome in young children. Later, it was noticed that these features were accompanied by some atypical signs such as shock, gastrointestinal involvement, and coagulation abnormalities.
The current syndrome in children is more common among Black older children, whereas KD is found in Asian children. Thus, the condition was recognized as a new disease, called multisystem inflammatory syndrome in children (MIS-C), also called pediatric inflammatory multisystem syndrome (PIMS).
The disease is found in regions where the virus is rife, but its onset occurs weeks after the peak of the epidemic. Seven of ten patients with MIS-C have negative RT-PCR for the virus, but serum antibodies are present against it. This indicates that it is not a primary consequence of the viral infection but is secondary to immune abnormalities.
The present study aims to uncover the innate and adaptive immune responses to reveal the aberrations that cause this syndrome. The researchers analyzed the clinical and laboratory features in eight children between the second part of April and the first part of May 2020. All had features of MIS-C. Among those who disclosed their racial background, the majority were Hispanic, at about 70%, or Black, at about 30%. Most were healthy children prior to the onset of disease.
The earliest symptoms were fever, with vomiting, diarrhea, and abdominal pain. Other symptoms from the list above were present in one or other of the children. Not one had inflammation of the limbs, a typical feature in KD. All of them had high fibrin degradation products (FDP), especially D-dimer, prothrombin time, and other features of coagulopathy.
None of them had a history of cough or cold. All patients showed abnormalities of heart function, with high troponin and brain natriuretic peptide (BNP) in seven of the patients. Five of them had coronary artery dilation or aneurysm. Respiratory complications like reactive airway disease, pleural effusion, or pneumonia occurred in about 50% of them.
On the first day, all but one patient received intravenous immunoglobulin IVIG, or the IL-6 inhibitor tocilizumab. On admission, a quarter of them had nasopharyngeal swabs that tested positive by RT-PCR, but no other viruses. One of the patients had a history of COVID-19 in the mother, three weeks before the child’s admission. One child had tested positive for the virus four weeks ago, on admission for symptoms of appendicitis. This supports the view that this virus can cause MIS-C after a delay of weeks in children.
Antibody Testing
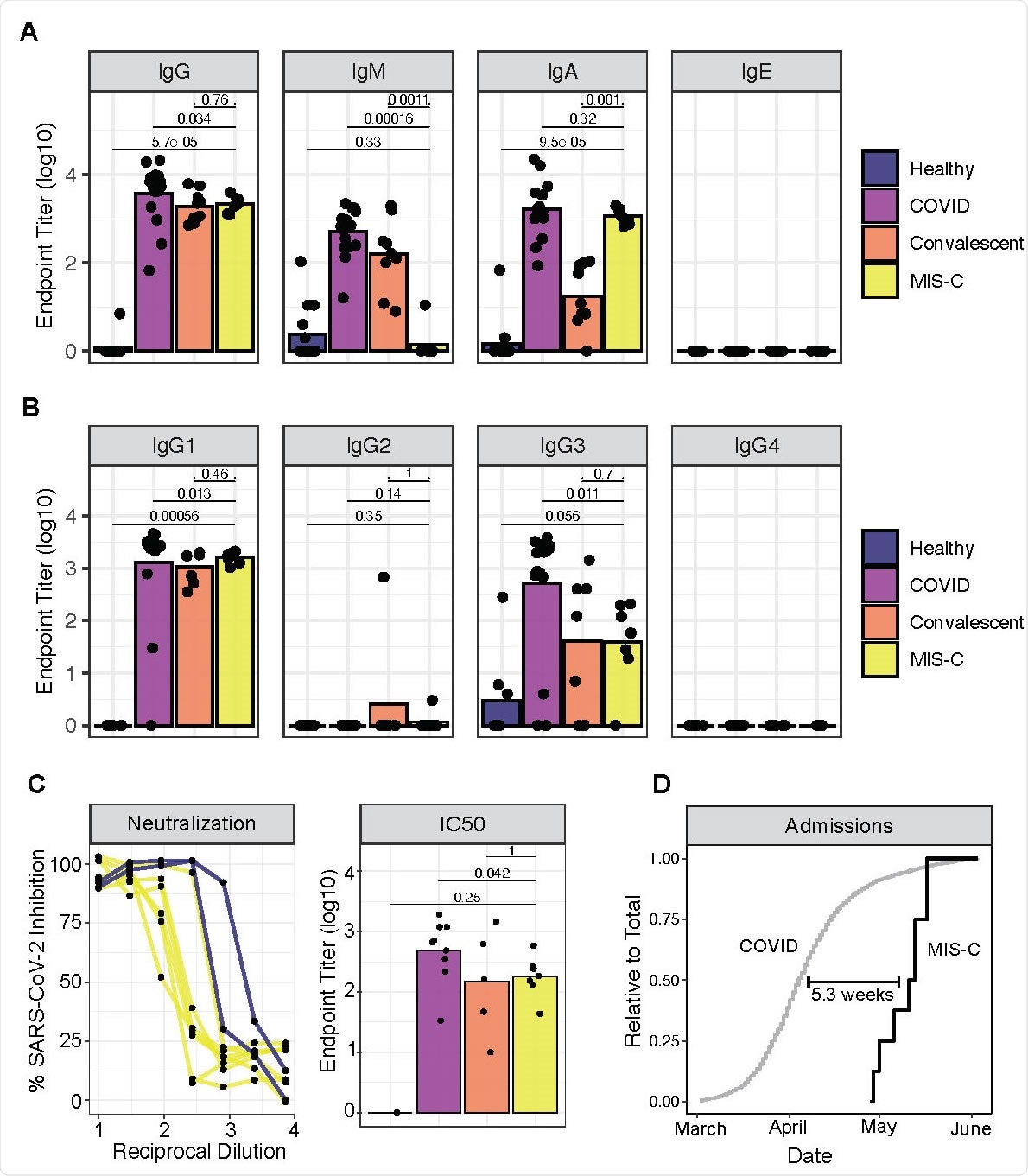
Serologic testing among the children showed high Immunoglobulin G (IgG) and low Immunoglobulin M (IgM) responses as expected in convalescent serum. However, they also had higher Immunoglobulin A (IgA) levels and lower IgM levels than typically found in convalescent serum, similar to the IgA titer in acute infection.
The next step was to look for neutralizing activity against live SARS-CoV-2 infection, using patient plasma. The researchers obtained positive results in all cases, again at levels comparable to convalescent serum, whether or not the children tested positive by RT-PCR. This suggests that positive PCR could simply be the sign that the infection was being overcome. Recent data does show that this test can remain positive for up to three weeks, but infectious particles cannot be recovered.
The delay from infection to the earliest MIS-C sign or symptom was worked out to be about five weeks.
MIS-C disease course and sample collection. (A) Timeline of admission, treatment, sample collection, and discharge. (B) Laboratory markers of inflammation, coagulopathy, and cardiac dysfunction in response to IVIG and/or TCZ treatment. Dotted lines represent normal limits.
Looking for Evidence of Inflammation and Chemokine Elevation
The researchers looked at cytokine profiles, including 92 parameters, in both patients and controls, as well as ten healthy adult donors, or hospitalized children with COVID-19 but without MIS-C.
The children with MIS-C had high levels of many cytokines, some higher than those found in acute infection like OPG and IL-10RA. The most significant quantum of increase was for Interleukin 6 (IL-6), but is partly due to the use of tocilizumab, which prevents its binding to the IL-6 receptor. The levels of cytokines and chemokines that recruit and modulate NK and T cells from the bloodstream were high, including CCL-3 and CCL-4, EN-RAGE, and CSF-1.
The cytokine levels also showed immunosuppression and exhaustion. Soluble PKL1 was very low, which suggests a response to acute inflammation. Cytokines that boost mucosal immunity were much higher, stimulating T-helper cell function and mucosal chemotaxis. High Vascular Endothelial Growth Factor A (VEGF-A) levels suggested that vascular function was also impaired.
Monocytes Elevated, T Cells Activated
The children had low frequencies of non-classical monocytes, natural killer (NK) cells, and T cells. The distribution of B and T cells among their phenotypes was not affected. Antigen-presenting cell activity was sharply increased and the level of CD64, which is commonly elevated in autoimmune and autoinflammatory conditions.
Rather than interferon type 1, however, other pathways seem to be recruited, such as phosphorylated STAT3. The researchers assume that the
Protein Enrichment
Both IgA and IgA autoantigens were found in the children’s samples, including the autoantigenic anti-LA found in Sjogren’s syndrome and in systemic lupus erythematosus. This suggests that MIS-C is a type of autoimmune illness, even though very few of the auto-antigens found in this study bear relation to autoimmune disease.
The autoantigens are most abundant in those organs that bear the brunt of the disease. This includes those found in the gut, the endothelial and cardiac tissue. Immune cell mediators were very high, which could mean that either antibody needs to bind these peptides to regulate immune cell activity, or else that this binding precipitates the formation of immune complexes in the affected organ.
Tocilizumab and IVIG Help Patients Recover
Most patients received tocilizumab, and only one failed to receive IVIG. Inflammatory markers like C-reactive protein, erythrocyte sedimentation rate, IL-6, IL-8, IL1-beta, TNF-alpha, ferritin, and markers of impaired coagulation like D-dimer were raised, as were markers of cardiac damage like troponin and BNP, at admission, and all showed rapid improvement. The median duration of hospital admission was 6 days, and all children recovered fully.
Features of MIS-C
All children in the study had developed antibodies against the virus, and antibody class switching was present. Circulating IgM was low, while effective neutralizing activity was present.
Inflammatory mediators were abundant in plasma, with activation of lymphocytes and myeloid cells and their recruitment to peripheral tissues. Mucosal chemokines were elevated, indicating gut infection. Non-classical monocytes migrated to the periphery, and there were several IgA and IgG autoantibodies against mucosal, endothelial and immune cell antigens.
Both neutrophils and monocytes showed enhanced expression of CD54 and CD64, the latter being known as the high-affinity FcγR1. This binds to autoantibodies and immune complexes, causing a robust inflammatory response and tissue damage.
Future Directions and Concerns
The later age at which MIS-C presents, compared to classical KD, could be due to its association with a novel virus. The genetic background remains to be researched since blacks and Hispanics are disproportionately affected. The future impact of the autoantibodies elicited by the infection also remains to be seen, even though the current episode is transient. Meanwhile, says the study, “MIS-C remains scientifically puzzling but therapeutically manageable.”
The researchers suggest, “Autoreactivity secondary to SARS-CoV-2 infection and the inflammatory innate immune response may be critical to the pathogenesis of MIS-C. These [autoantibodies] may arise by direct cross-reactivity between SARS-CoV-2 and self-antigens. If true, [this] will pose a risk for future vaccination strategies.”

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources