.jpg)
Novel Coronavirus SARS-CoV-2 Transmission electron micrograph of SARS-CoV-2 virus particles, isolated from a patient. Image captured and color-enhanced at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID
Coronavirus disease (COVID-19) pandemic, which is caused by SARS-CoV-2, swiftly advanced from a local outbreak to a pandemic. As of July 8, 2020, there were approximately 12 million confirmed cases and 550,000 deaths around the globe.
Since the disease is wreaking havoc in health care systems and societies worldwide, a myriad of strategies to combat COVID-19 are currently being tested. And although contact tracing, convalescent plasma treatment, and antiviral drugs are important targets, many argue that without an effective vaccine, we cannot expect the return to normalcy.
Luckily, several promising vaccine candidates are already in clinical trials, but multiple vaccines will be likely needed. Consequently, the ones with a rapid and uncomplicated manufacturing process will be especially useful.
SARS-CoV-2 has many structural components that are promising target candidates – including the spike protein, nucleocapsid protein, and receptor-binding domain. The spike protein is particularly interesting, as previous studies have demonstrated its propensity to induce robust neutralizing antibodies and T-cell response against SARS-CoV-2.
A group of scientists from the Wyss Institute for Biologically Inspired Engineering at Harvard University, and the Center for Virology and Vaccine Research at Beth Israel Deaconess Medical Center, Harvard Medical School, as well as from Massachusetts Consortium on Pathogen Readiness, leveraged a biomaterial vaccine technology (comprised of mesoporous silica rods) that enable a sustained release of granulocyte-macrophage colony-stimulating factor (GM-CSF) and adjuvants to concentrate and mature antigen-presenting cells at the vaccine site.
Plug-and-play nature of mesoporous silica rods
The researchers decided to use this material as synthetic amorphous silica is already known to have quite reliable biocompatibility, which supports its development as an adaptable platform for a plethora of clinical applications.
Furthermore, the GM-CSF and monophosphoryl lipid A (MPLA) used in this biomaterial vaccine are both safe for use in humans. At the same time, the local release of these agents allows the use of small quantities – minimizing any concerns regarding potential toxicity or systemic side-effects.
In this study, the researchers appraised the humoral responses stemming from the use of MPLA as the adjuvant on one side, and SARS-CoV-2 spike protein, the nucleocapsid protein, and receptor binding domain as the target antigens on the other side. Antigen dose and impact of pre-manufacturing of vaccines versus loading antigen just-in-time was additionally explored.
The undemanding manufacturing process of vaccines based on mesoporous silica rods, together with their plug-and-play nature, allowed vaccine fabrication and experimental mice vaccination within seven days upon obtaining the protein antigens. For the purposes of this study, SARS-CoV-2 pseudovirus expressing a luciferase reporter gene has been utilized.
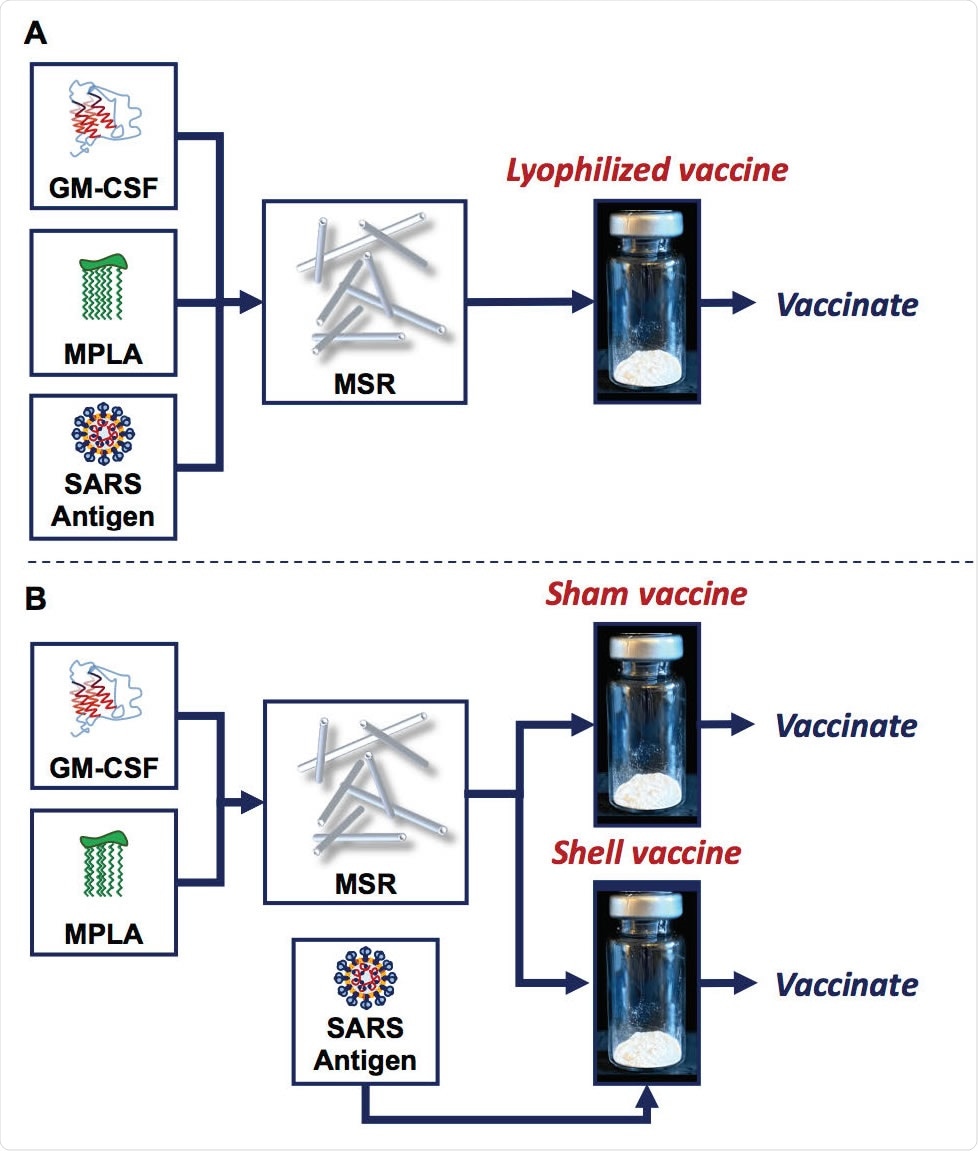
MSR vaccines can be manufactured with antigen added before lyophilization or added after, prior to vaccination. (A) Granulocyte-macrophage colony-stimulating factor (GM-CSF), monophosphoryl lipid A (MPLA), and the SARS antigens were adsorbed onto the biomaterial scaffold, mesoporous silica rods (MSR). The mixture was then frozen and lyophilized (lyophilized vaccine) until ready for vaccination. (B) GM-CSF and MPLA were adsorbed onto MSR, which was then frozen and lyophilized (sham and shell vaccine). Prior to vaccination, the SARS antigens were added just-in-time and adsorbed on shell vaccines.
Functional saturation even at lower doses
"Our results demonstrate the development of rapid and robust antibody titers against all three SARS-CoV-2 proteins that were tested, in all vaccine configurations", study authors delineate their main findings.
More specifically, the addition of SARS-CoV-2 antigens to shell vaccines before immunization (i.e., add-in-time) was shown to induce a vigorous humoral response, similar over time as those seen with the use of pre-manufactured lyophilized mesoporous silica rods vaccines.
Moreover, this study has also shown that the antibodies induced by administered vaccines indeed confer functional neutralization against a SARS-CoV-2 pseudovirus. Two antigen doses tested in this study prompted analogous humoral responses, suggesting that the lower dose of 1 microgram was already saturating.
Still, it has to be noted that the IgG antibody response kinetics was slightly slower with the add-in-time version of the vaccine, which could be a result of inadequate times allowed for antigen adsorption before vaccination.
Quick and scalable solution to tackle COVID-19 pandemic
In a nutshell, this research has utilized recombinant protein subunit vaccines, since they allow ample control over the epitopes and antigen amounts, but also enable fine-tuning of immune responses with adjuvants.
"The sustained presentation of antigen and adjuvant made possible by the MSR vaccine, along with the accumulation of large numbers of dendritic cells at the vaccine site, has been previously demonstrated to yield robust and durable immune response in the context of oncology, as well as infectious diseases", explain study authors.
One of the advantages of this technology is scalability and modularity that enables plug-and-play versatility to quickly incorporate desired antigen. And this is definitely the way forward in our quest towards potent and protective humoral response to SARS-CoV-2.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources