As the COVID-19 pandemic continues to threaten the world with a second wave even in areas that had achieved control, scientists are working frantically to understand how the host produces antibodies against the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). A full understanding of this process is required to develop mitigation measures to reduce the severity of the outbreak. Now, a new study by researchers at New York University and published on the preprint server bioRxiv* in August 2020 reports on the type and intensity of anti-SARS-CoV-2 antibodies in recovered COVID-19 patients and the implications of these findings.
One of the most important questions is to understand whether the immune response to this virus produces durable protection against reinfection. This will also determine the usefulness of a vaccine.
Most studies rely on antibody testing to distinguish the infected from the non-infected in a community. However, these may provide a partial seroprevalence picture through a binary answer mode to the question of whether an infection has occurred or not. They do not help to understand whether the detected antibodies are neutralizing and prevent reinfection.
.jpg)
Study: High titers of multiple antibody isotypes against the SARS-CoV-2 spike receptor-binding domain and nucleoprotein associate with better neutralization. Image Credit: Kateryna Kon / Shutterstock

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
In the current study, the researchers used convalescent serum obtained from 101 healthcare workers who had tested positive for the virus. They analyzed the different classes of antibody as well as the neutralizing potency of the serum on both the wildtype virus and a lentiviral pseudovirus.
Pseudotyped viruses were used because they are not pathogenic, unlike the native virus, and are therefore employed on a much broader scale in research. On the other hand, they do have differences relative to the wildtype virus, as shown by the variation in their neutralization by convalescent serum.
They looked at the separate Immunoglobulin G (IgG), Immunoglobulin M (IgM), and Immunoglobulin A (IgA) isotypes to the SARS-CoV-2 receptor-binding domain (RBD) on its spike glycoprotein, as well as to the nucleoprotein. The minimum dilution at which convalescent serum provided complete protection against cell infection by the virus, termed the minimum inhibitory concentration (MIC), was calculated for each neutralization assay using the virus. When the pseudovirus was used, the researchers employed a luciferase assay to calculate the IC50 and the IC90.
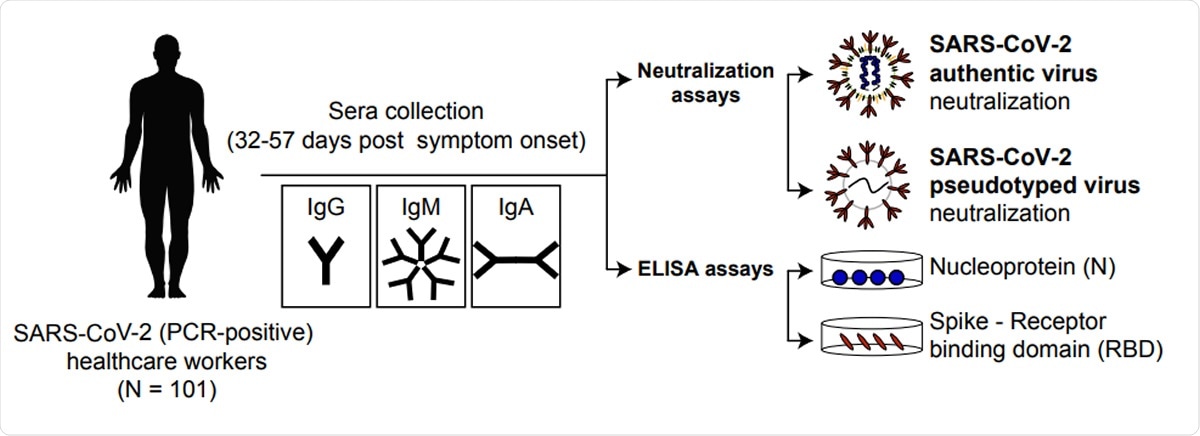
SARS-CoV-2 neutralizing antibody response. Schematic representation of the experimental design.
Low Neutralizing Response
The study shows that neutralization assays using these two systems are positively correlated. The researchers also found three categories of antibody response:
- Most individuals, about 75%, had a low neutralizing antibody capacity, as measured by MIC and IC90, against the wildtype virus and the pseudovirus.
- About 20% had intermediate neutralization capacity against both.
- Only about 6% had high neutralizing capacity against the wildtype virus, but this was zero when pseudotyped virus neutralization was considered. Thus, there was a significant difference in the proportion of high neutralizers found using a pseudotyped virus.
The researchers say that based on this, pseudovirus neutralization is an efficient option to the natural virus for such testing, but caution, “Authentic SARS-CoV-2 virus may be better able to detect potent neutralizing serums, which has implications for the selection of donors for passive immunization therapy.”
The study used samples from individuals at different times from symptom onset, which may account for the difference in neutralizing capacity. However, no timeline could be observed concerning this group. That is to say, low, high, and intermediate neutralization capacity was found at various time points between 32 and 57 days.
One important finding is that at this timeline, ‘serum SARS-CoV-2 neutralizing antibody capacity is low in most recovered individuals.”
Specific and Multi-Isotype Response
Secondly, the researchers examined the isotypes against two different viral antigens and the range of antibody isotypes. They found high IgG, IgM, and IgA titers against both antigens, corresponding to the neutralization capacity. This is important to know since many a time, antibodies are overproduced in response to one or a few viral proteins, and if so, using an antigen other than these may result in a falsely low antibody titer.
The current study showed a uniform antibody response to both spike and N proteins, with the strongest correlation being with IgG, followed by IgA and IgM. However, most patients who developed anti-RBD IgA were negative for anti-N IgA, unlike IgG and IgM agreement for the RBD and N proteins. This would mean that the test used to detect IgA to the N protein, as compared to the RBD, would be negative in many cases.
Again, the study failed to show any relationship between the time of symptom onset and the titer of each isotype. Very interestingly, IgM was detected at 50 days from the date of infection.
Thus, they conclude, “The antibody isotype response to SARS-CoV-2 may be antigen-specific, with IgA skewed towards the RBD.”
Neutralization Correlates with RBD Antibody Response
The researchers found that neutralization capacity could be predicted by the antibody titers obtained from ELISA assays. Comparing each ELISA antibody titer with virus neutralization capacity, the anti-RBD antibody titer for all isotypes bore a strong correlation to the ability to neutralize the wildtype virus. Anti-N IgG also showed significant correlation but not other isotypes.
However, the pseudovirus neutralization capacity correlated best with the IgG titer rather than the other isotypes. For both types of viral particles, the correlation with neutralizing capacity was weaker for anti-N IgA and IgM compared to anti-N IgG.
The researchers postulate that this may be due to misfolding of the folding of the RBD in the purified form, in the wildtype virus, and in the pseudovirus, causing the antibody isotypes to recognize them differently. IgM antibodies are generally thought to signify recent infection, with declining titers over time as antibody production switches to IgG and IgA. Instead, this may indicate that all the isotypes have to be evaluated to understand the true timeline of the immune response in this infection.
The researchers comment, “These data suggest that ELISA methods based on the RBD may benefit from detection of additional isotypes, rendering them better suited as predictors of sera neutralization.”
Multi-Isotype High Antibody Titer Indicates Strong Neutralizing Response
The researchers then compared the titers for each isotype and each antigen, finding that the highest neutralizing sera had higher antibody isotype titers against both antigens. Within each serum sample, the isotypes targeting the RBD agreed better than those against the N protein.
The researchers say, “Mounting a robust antibody response, consisting of diverse isotypes, leads to efficient neutralization for all isotypes.”
Implications
The researchers classified the patients into 21 different clusters depending on the combination of antibody isotypes, from those who were positive for all six to being negative for all. Of the 6 individuals with high neutralizing titers, four were positive for all isotypes against both antigens, while two had a combination common to medium and low neutralizers as well. In fact, one cluster which is positive for all except anti-RBD IgM has low neutralizing capacity. This may mean that certain combinations are needed for the highest neutralization to occur.
Again, most individuals were positive for IgG and IgA against the RBD, and for IgG against N protein, suggesting that IgG alone may not suffice for good neutralization.
The researchers muse, “It remains to be elucidated how these clusters are generated and why certain clusters elicit potent SARS-CoV-2 neutralization, and others do not. Since we use a targeted approach focusing on only two antigens and specific epitopes, it is possible that there are other anti-SARS-CoV-2 antibodies that bind viral particles and impact neutralization.”
Since disease severity is well known to correlate with antibody production, in high neutralizers, the multiple isotypes and high neutralizing titer could be driven by severe disease. In contrast, three individuals did not seroconvert, as shown in some earlier studies too. Perhaps they really did not produce neutralizing antibodies, or the response declined to undetectable levels by the time of this study.
Follow-up studies over longer periods are beginning to show how antibody titer is related to protection against reinfection. An important caveat is that laboratory neutralization assays show just that – but do not necessarily convey the protective capacity of antibodies in the clinical setting.
The researchers say, “Detailed studies monitoring initial and possible reinfections along with the antibody response are crucial to understanding immunity to SARS-CoV-2.”

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Noval, M. G. et al. (2020). High Titers of Multiple Antibody Isotypes Against The SARS-Cov-2 Spike Receptor-Binding Domain and Nucleoprotein Associate with Better Neutralization. bioRxiv preprint. doi: https://www.biorxiv.org/content/10.1101/2020.08.15.252353v1
- Peer reviewed and published scientific report.
Tada, Takuya, Belinda M. Dcosta, Marie I. Samanovic, Ramin S. Herati, Amber Cornelius, Hao Zhou, Ada Vaill, Wes Kazmierski, Mark J. Mulligan, and Nathaniel R. Landau. 2021. “Convalescent-Phase Sera and Vaccine-Elicited Antibodies Largely Maintain Neutralizing Titer against Global SARS-CoV-2 Variant Spikes.” Edited by Stephen P. Goff. MBio 12 (3). https://doi.org/10.1128/mbio.00696-21. https://journals.asm.org/doi/10.1128/mBio.00696-21.