With the COVID-19 pandemic raging across the world unabated and having claimed over eight hundred thousand lives, the search for a vaccine to prevent the infection is on. Several candidate vaccines are being developed to prevent the dreaded infection caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
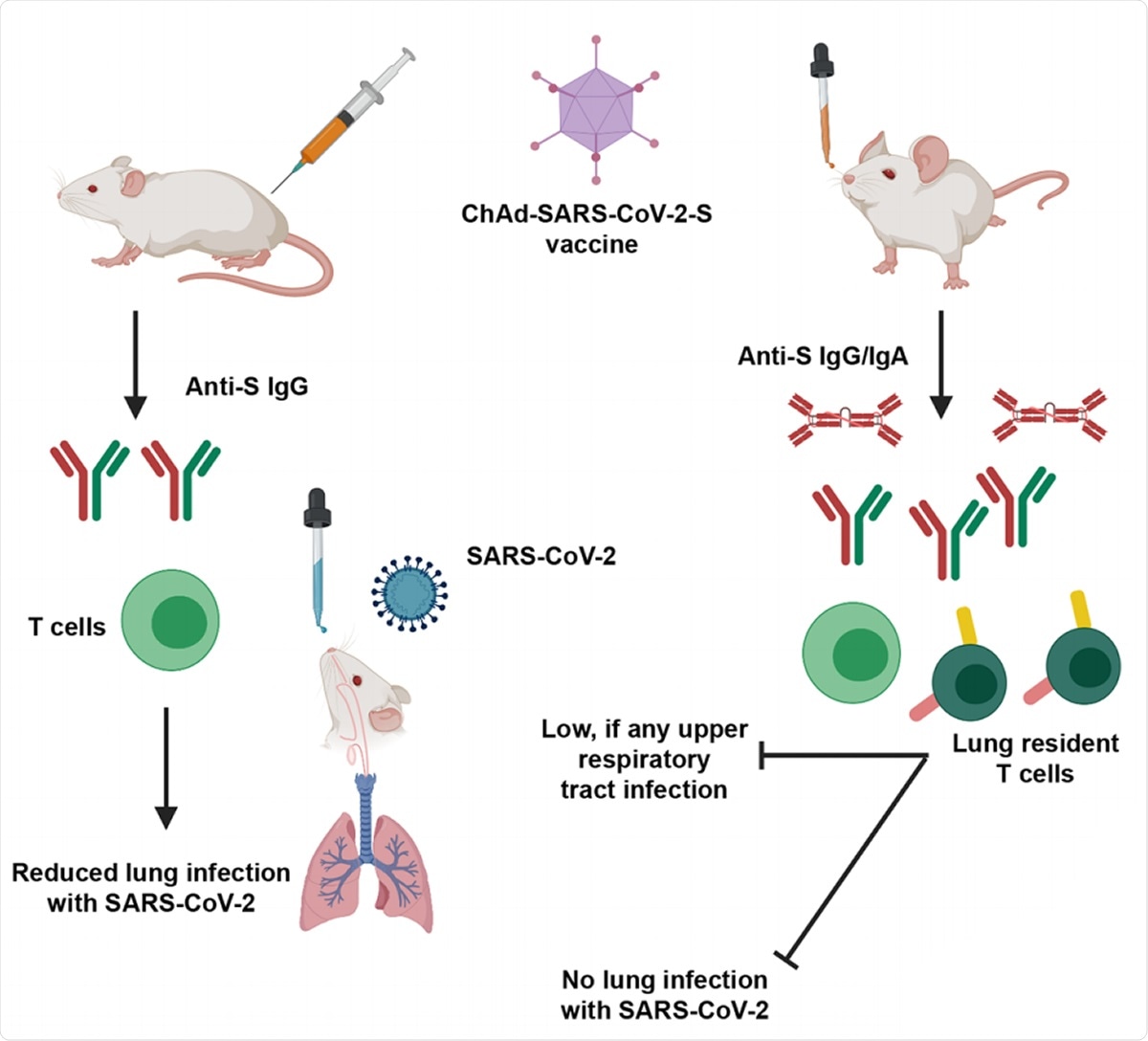
Image Credit: Cell
The study premise
The team writes that the SARS-CoV-2 RNA genome is of around 30,000 nucleotides length and two-thirds of these codes for nonstructural proteins that allow it to replicate and synthesize viral RNA. The remaining third of the genome codes for the proteins such as “spike (S), envelope, membrane, and nucleoprotein (NP).” The S protein that forms the spikes on the virus binds to the cell-surface receptor angiotensin-converting enzyme 2 (ACE2), which allows for the entry of the virus into the host cell. It has two parts, S1 and S2. The S1 protein has the receptor-binding domain (RBD), and the S2 protein allows the fusion of the virus to the cell membrane. The S protein, thus, is vital and is a potential vaccine target, say the researchers.
Animal studies and the new vaccine
The team explains that several vaccines that are being developed have progressed to human trials before having been tested adequately in lab animals for safety and efficacy first. These animal studies are called “pre-clinical studies.” They write, “This circumstance occurred in part because vaccine design and development has outpaced the generation of accessible pre-clinical disease models of SARS-CoV-2 infection and pathogenesis.”
They write that there have been Adenovirus (Ad)-based vaccines against coronaviruses before especially against MERS-CoV. The team thus developed a “chimpanzee adenovirus vectored vaccine encoding a pre-fusion stabilized spike protein (ChAd-SARS-CoV-2-S)” against the SARS CoV-2. This new vaccine is Ad-based and expresses the S protein. One similar vaccine is also being tried on humans as a single intramuscular injection, they wrote. This, they wrote, is a different chimpanzee Ad (simian Ad-36)-based SARS-CoV-2 vaccine (ChAd-SARS-CoV-2-S) that would code for a pre-fusion stabilized spike (S) protein. It was made by introducing two proline substitutions in the S2 subunit.
Explaining the process, the team said that they took adenoviruses, which cause the common cold and inserted the genome that codes for the spike protein on coronaviruses into its genes. This new adenovirus was made incapable of causing disease. This neutralized adenovirus then carried the spike protein into the nose of the test animal. The body of the lab animal, in turn, mounted an immune response without developing an infection, they explained. One of the authors of the study, David T. Curiel, MD, PhD, the Distinguished Professor of Radiation Oncology, said, “Adenoviruses are the basis for many investigational vaccines for COVID-19 and other infectious diseases, such as Ebola virus and tuberculosis, and they have good safety and efficacy records, but not much research has been done with nasal delivery of these vaccines.” He added, “All of the other adenovirus vaccines in development for COVID-19 are delivered by injection into the arm or thigh muscle. The nose is a novel route, so our results are surprising and promising. It’s also important that a single dose produced such a robust immune response. Vaccines that require two doses for full protection are less effective because some people, for various reasons, never receive the second dose.”
What was done?
The team used their candidate vaccine as an intramuscular injection on lab mice that had been exposed to SARS-CoV-2. They noted that it could produce a robust “systemic humoral and cell-mediated immune responses.” A single dose or double doses of this vaccine could protect the animal from lung infection, inflammation, and other pathological features of SARS-CoV-2. However, the injection of the vaccine in one or two doses did not protect the mice against SARS-CoV-2 infection and a high level of the virus was seen in the lungs of the mice.
The mice they used for their tests were genetically modified so that they had human ACE2 (hACE2) receptors, which made them susceptible to SARS CoV-2. They next tried the use of ChAd-SARS-CoV-2-S as an intranasal preparation.
Intranasal vaccine works
A single intranasal dose of the vaccine was capable of inducing high levels of neutralizing antibody and anti-SARS-CoV-2 IgA. The team wrote that this single dose, “conferred virtually complete protection against infection in both the upper and lower respiratory tracts in mice expressing hACE2 receptor after adenoviral vector delivery or as a transgene”.
Conclusions and future direction
The team wrote in conclusion, “Thus, ChAd SARS-CoV-2-S has the potential to control infection at the site of inoculation, which should prevent both virus-induced disease and transmission.”
Study author Michael S. Diamond, MD, PhD, the Herbert S. Gasser Professor of Medicine and a professor of molecular microbiology, and of pathology and immunology, said in a statement, “We were happily surprised to see a strong immune response in the cells of the inner lining of the nose and upper airway — and a profound protection from infection with this virus.” He added, “These mice were well protected from disease. And in some of the mice, we saw evidence of sterilizing immunity, where there is no sign of infection whatsoever after the mouse is challenged with the virus.”
Speaking about the next step, Diamond said, “We will soon begin a study to test this intranasal vaccine in nonhuman primates with a plan to move into human clinical trials as quickly as we can. We’re optimistic, but this needs to continue going through the proper evaluation pipelines. In these mouse models, the vaccine is highly protective. We’re looking forward to beginning the next round of studies and ultimately testing it in people to see if we can induce the type of protective immunity that we think not only will prevent infection but also curb pandemic transmission of this virus.”
Journal reference:
- Hassan, A.O., Kafai, N.M., Dmitriev, I.P., Fox, J.M., Smith, B.K., Harvey, I.B., Chen, R.E., Winkler, E.S., Wessel, A.W., Case, J.B., Kashentseva, E., McCune, B.T., Bailey, A.L., Zhao, H., VanBlargan, L.A., Dai, Y.-N., Ma, M., Adams, L.J., Shrihari, S., Danis, J.E., Gralinski, L.E., Hou, Y.J., Schäfer, A., Kim, A.S., Keeler, S.P., Weiskopf, D., Baric, R.S., Holtzman, M.J., Fremont, D.H., Curiel, D.T., Diamond, M.S., A single-dose intranasal ChAd vaccine protects upper and lower respiratory tracts against SARS-CoV-2, Cell (2020), doi: https://doi.org/10.1016/j.cell.2020.08.026