The virus also displays remarkable global and thermal stabilities, says the team.
Miklós Kellermayer (Semmelweis University) and colleagues say the dynamics of the spike proteins found on the surface of the virus may contribute to the unusual infectivity of SARS-CoV-2, while its mechanical properties and ability to self-heal may contribute to its ability to adapt to wide-ranging environmental conditions.
The researchers also say the single-particle approach used in this study could be useful not only for shedding light on the mechanical properties underlying SARS-CoV-2 infection but also for investigating how the virus responds to potential therapies.
A pre-print version of the paper is available on the server bioRxiv*, while the article undergoes peer review.
Structural information so far
SARS-CoV-2 is a single-stranded RNA virus with a layer of corona-shaped spike proteins on its surface that are fundamental to the infection process.
Recent structural information acquired using purified protein crystals or fixed and frozen virions indicate that hinges in the spike protein provide structural flexibility and that the ribonucleoproteins (RNPs) are segregated into spherical, basket-like structures.
However, how these structural features contribute to the dynamic and mechanical behaviors of native SARS-CoV-2 remains unclear.
What did this study reveal?
Now, Kellermayer and colleagues have applied atomic force microscopy (AFM) and molecular force spectroscopy to investigate the topographical structure and nanochemical properties of chemically-fixed native SARS-CoV-2 virions.
The images revealed spherical virions of varied dimensions and a rugged surface with protrusions that the team identified as spike proteins. Using high-resolution AFM, individual spike proteins were identified, and the team calculated that, on average, the viral surface displays 61 of these proteins.
“This number exceeds those reported recently, suggesting that the spike number is highly variable and may be regulated during virus assembly and maturation,” writes the team.
The researchers say the flexural disorder they observed supports previous cryo-electron microscopy data indicating a high degree of spike flexibility, and they suggest this disorder is due to high mobility of the spike proteins in the viral envelope.
To counteract the effects of chemical fixation and investigate spike dynamics in situ, the team explored the topography of unfixed, native virions.
The spikes quickly and dynamically fluctuated within a space that was restricted by the flexible neck of the proteins. The researchers say it is likely that spike motion is dictated by the Brownian dynamics of the receptor-binding domain (RBD), which can be considered as a tethered particle.
Kellermayer and colleagues suspect that the rapid spike motion enables SARS-CoV-2 to carry out an efficient dynamic search of target host cell surfaces, which may explain why it is at least as infective as the influenza virus, despite having far fewer spike proteins (around 60 versus 350).
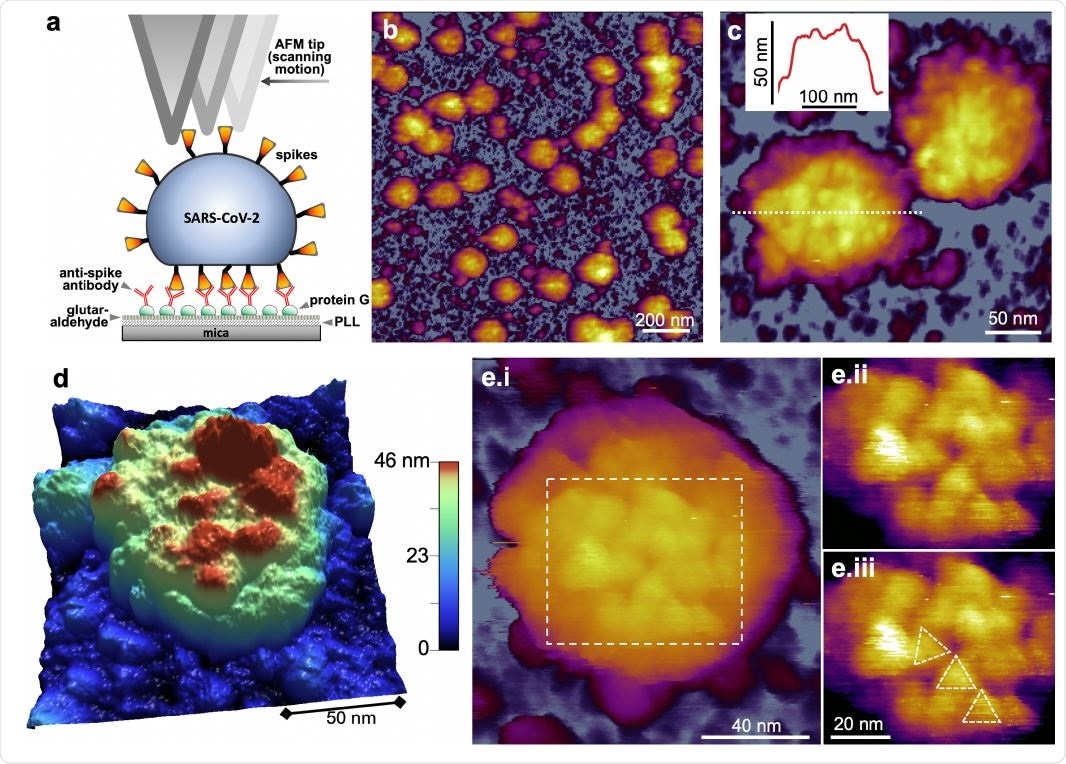
Topographical structure of SARS-CoV-2 virions treated with 5 % glutaraldehyde for structural preservation. a. Schematics of scanning substrate-surface-bound virions with the AFM tip. PLL: pol-L-lysine. b. AFM image of an overview (1.5 x 1.5 μm) sample area. c. Zoomed-in AFM image of SARS-CoV-2 virions. The virion surfaces are covered with protrusions that correspond to spikes (S protein trimers). Inset, topographical profile plot measured along the center of one of the virions (dotted line). The profile plot reveals a rugged surface. d. 3D-rendered image of a SARS-CoV-2 virion. A somewhat flattened virion is observed, pointing at a global flexibility of the virion. e. High-resolution AFM image of a SARS-CoV-2 virion displaying axial view of S trimers. i. AFM image of the entire virion. ii. Enlarged and contrast-enhanced image of the rectangular area. iii. The same AFM image with overlaid triangles indicating S trimer orientation. The spikes apparently display translational, rotational and flexural disorder owing to their flexibility.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Investigating the mechanical properties of SARS-CoV-2
To explore the mechanical properties of SARS-CoV-2, the researchers applied indentation by lowering the cantilever tip on the vertex of individual virions that were selected on the AFM images.
This showed that the virus had a mean stiffness that made it the most compliant virus investigated to date. The virion stiffness, which is lower than that measured for the influenza virus lipid envelope, suggests that the viral envelope dominates the elasticity of SARS-CoV-2, say the researchers.
“Unlike in other viruses, the force did not drop to near-zero values following mechanical yield, indicating that virion collapse or breakage was not evoked despite the drastic mechanical perturbation,” they write.
The team says the mechanical yield may be enabled by the force-induced rearrangement of the basket-like structure of the RNPs.
Following indentation, the team retracted the cantilever tip, which resulted in the generation of forces that suggested the structural recovery was occurring. This continued until the initial virion height was recovered, and the researchers suggest this may have been driven by structural restoration of the initial RNP arrangement.
The indentation-retraction process was carried out up to 100 times, without the virus breaking or collapsing.
“The virions are highly compliant and able to recover from drastic mechanical perturbations,” say Kellermayer and colleagues.
Testing thermal stability
Next, the team assessed thermal stability by exposing virions to temperatures of 60, 80, and 90 ˚C for ten minutes before cooling them back down to 20˚C and performing AFM imaging.
“Remarkably, virions remained on the substrate surface, and their global appearance was only slightly altered,” say the researchers.
Following the heat treatment, far fewer spikes were present, and their trigonal shape was unresolved, indicating that they had been thermally denatured.
Therefore, although SARS-CoV-2 does display remarkable global thermal stability, the conformational response of the spike proteins did eventually lead to their heat-induced inactivation.
“Thus, both the infectivity and thermal sensitivity of SARS-CoV-2 rely on the dynamics and the mechanics of the virus,” writes the team.
A dynamic, compliant and surprisingly resilient virus
The researchers say the study shows that SARS-CoV-2 is a highly dynamic, compliant, and surprisingly resilient virus that displays remarkable mechanical and global thermal stabilities.
“While the dynamics of the surface spikes may play an important role in the unusually high infectivity of the virus, its mechanical and self-healing properties may also ensure adaptation to a wide range of environmental circumstances,” they write.
“Considering its capability of exploring viruses under native conditions, the single-particle approaches employed here may be important in uncovering not only the mechanistic details behind viral infection but the viral response to potential therapies as well,” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources