Copper is inherently antimicrobial. When cleaned regularly, frequently touched surfaces manufactured from uncoated copper alloy materials will continuously kill bacteria that cause infections. The EPA (Environmental Protection Agency) has approved hundreds of copper alloys for their disinfectant properties. However, it takes up to 4 hours to fully deactivate microbes.
Currently, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has spread all over the world infecting over 56 million individuals and caused over 1.34 million deaths. Complex host-virus interaction and infection process, high transmission rate, and poor mitigation methods have led to the continuing of this crisis.
To contain the coronavirus disease 2019 pandemic (COVID-19) caused by SARS-CoV-2, we need agents better than masks, soap and alcohol-based sterilizers. Specifically, antiviral agents need to act rapidly. This will disinfect before any possible transmission of the virus, effectively arresting the spread of infection.
In a recent bioRxiv* preprint paper, a team of researchers from Kuprion, Inc., Integrated Pharma Services, Virginia-Maryland College of Veterinary Medicine, Virginia Polytechnic Institute and State University, and George Mason University shows the possibility of using copper, in a new conformation, for effectively killing microbes immediately.
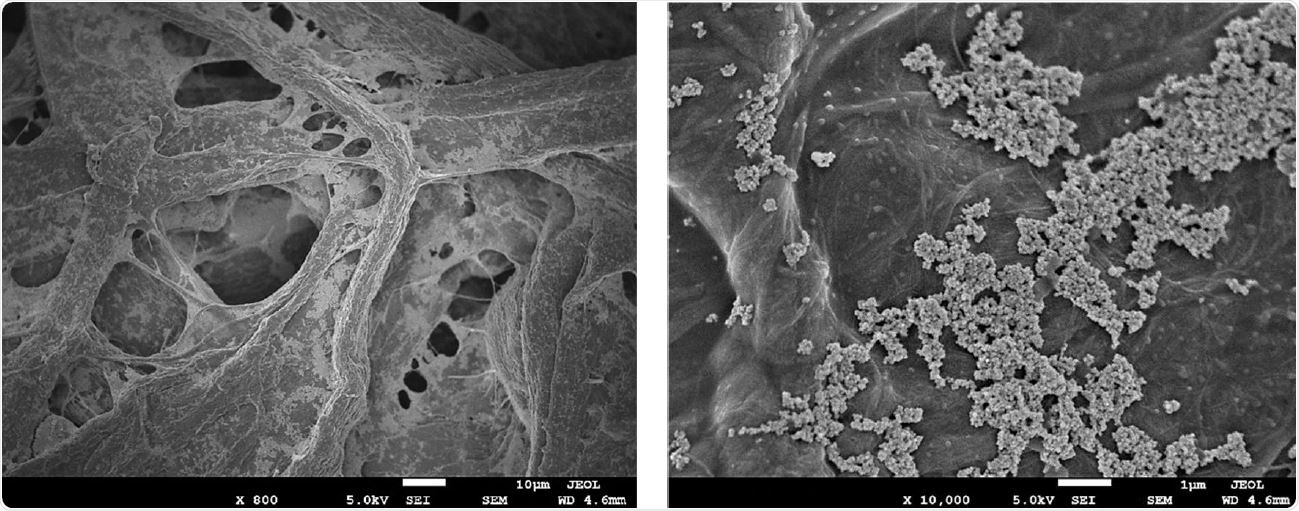
High magnification scanning electron microscopy images of aCu on 45/55 cellulose/polyester fabric. (A) fibers of the cellulose/polyester fabric coated with aCu. (B) 10,000x magnification of aCu on fabric, showing copper agglomerates in the 3-7 micrometer range.
Here, Alfred A. Zinn et al. have created a rapid-acting, self-sterilizing copper configuration, called ‘active copper,’ and have quantified its activity against a host of viruses and bacteria with the eventual goal to test against the SARS-CoV-2 virus. While the effect of copper against microbes is well known, this study aims to achieve prolonged effects of antimicrobial activity.
In this study, active copper (aCu) is a large interconnected mesostructured metal network consisting of polymeric gel-like copper material with mesoscale porosity and surface roughness. It is manufactured using a bottom-up synthesis approach: Anhydrous copper (II) chloride is reduced with sodium borohydride in the presence of amine surfactants to form the raw aCu material. It behaves like a typical non-Newtonian “liquid” with high thixotropy.

aCu after being dispensed from a tube. The dense thixotropic copper gel is paste-like and stable. It behaves like a typical non-Newtonian “liquid”. A layer of amine surfactants exerts strong cohesion via hydrophobic interactions and organic end-chain entanglement.
Its structure is dense but flowable metallic gel-like paste with an interconnected mesoscale network, porosity, and surface roughness that enables safe handling and processing. The authors have characterized the material in detail.
In this study, the researchers perform a time-dependent antimicrobial activity (against Salmonella enterica-specific bacteriophage Ø32) to determine how quickly aCu acts against microbes. They formulated water-based paints to coat different fabrics and porous filter materials that could be used in face masks and air purification units such as HVAC systems in airplanes, hospitals, or public transportation. All active samples showed 100% viral kill rates.
The researchers determined the aCu activity against enveloped (influenza) and non-enveloped (calicivirus) viruses on textile products. They found that the aCu textile reduced the concentrations of all three viruses by more than 99%.
A viral load of approximately 106 PFU/ml was applied to the fabric; after 30 seconds 99.9% of the virus had been killed. And this effect is shown to have a prolonged use as opposed to the need for repeated application of sterilizers and other methods.
“This dramatic efficacy makes aCu a very promising candidate for the development of self-cleaning PPE,” say the researchers.
They also tested the sustained antimicrobial efficacy according to EPA protocol, demonstrating a high-efficacy of aCu. They also show that aCu maintains vigorous antimicrobial activity independent of its substrate.
The team conducted tests under simulated breathing conditions to ensure zero respiratory exposure upon using an aCu-coated insert in a face mask. They found a maximal copper “dose” of approximately 2 ng/hour for a wearer - this is significantly below OSHA’s maximum Permissible Exposure Limit (PEL) of 1mg/m3 for copper dust in the workplace.
This study is funded exclusively by Kuprion Inc. The authors declare that they own an extensive patent portfolio that protects the foundations of this technology platform. For those who may be interested, the authors inform that the pre-production phase is currently manufacturing 40-50 kg of base ActiveCopper per month; and the material is commercially available.
This study reports the excellent rapid antimicrobial activity of a copper configuration, presenting far-exceeding results in killing SARS-CoV-2 and other microbes in seconds. Because of its broad range of activity, it is also possible that mutations in the virus will also bear no change in its antiviral efficacy. Also, the authors speculate on the possible mechanisms responsible for aCu’s non-specific eradication of microbes.
The authors propose that a long-lasting protective mask integrated with aCu offers individuals an immediate and reliable defense against COVID-19. Not only on masks, but this may also be applied on all high-traffic virus hosting sites to effectively curb the SARS-CoV-2 infectivity.
Journal reference:
- A continuously self-sterilizing form of copper capable of 99% SARS-CoV-2 deactivation in 30 seconds. Alfred A. Zinn, Mina Izadjoo, Hosan Kim, Kylene Kehn-Hall, Caitlin Lehman, Rachel L. Brody, Robert R. Roth, Augustin Vega, Khanh K. Nguyen, Nhi T. Ngo, Hannah T. Zinn, Lauren Panny, Rafaela Flor, Nicholas Antonopoulos, Randy M. Stoltenberg bioRxiv 2020.11.16.384040; doi: https://doi.org/10.1101/2020.11.16.384040, https://www.biorxiv.org/content/10.1101/2020.11.16.384040v1