Serological studies help understand the proportion of pathogen exposure in a population, which helps identify what point in the epidemic a given region or country is at. Case reporting in the current coronavirus disease 2019 (COVID-19) pandemic has been found to identify only a fraction of actual infections. Thus, seroprevalence appears to be a more accurate method of assessing population exposure.
However, an important new realization came to the fore, namely, the apparent rapid waning of specific antibodies against the virus to undetectable levels, or seroreversion. Researchers have found that antibody titers are inferior to cellular immunity as indicators of long-term immunity, preventing reinfection, but it is more difficult and expensive to measure the latter.
As a result, most countries still rely on seroprevalence surveys, including England, which carried out one such survey from June 20 to July 13, 2020. This resulted in a 13% estimated prevalence of infection in London, and 6% in England, overall. On this basis, the infection fatality ratio (IFR) was calculated to be 0.90%. The estimate failed to take declining antibody levels into account, which may have resulted in an underestimation of the true infection levels.
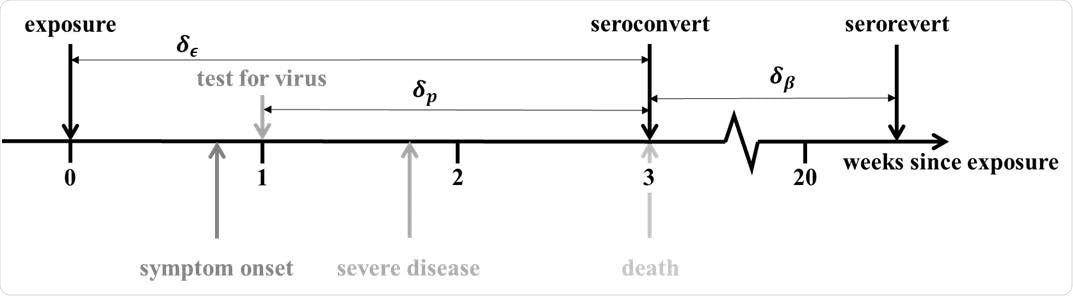
Progression of exposed individuals through the various clinical (below the timeline), and diagnostic (above the timeline) stages of infection and recovery. Stages marked in grey represent events that may happen, with a probability consistent with the darkness of the shade of grey.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Seroreversion affects seroprevalence
Ongoing research shows that antibodies are detectable for about six months following infection. Once they fall below the limit of detection of a conventional assay, the test is interpreted as negative. In other words, a negative serological test may denote either the absence of antibodies or a decay in antibody titers with time. This may be the reason for the fall in seroprevalence in England after its peak towards the end of May 2020, rather than the expected steady rise as more people become infected over time.
This seroreversion is defined as “the inverse of the average time taken following seroconversion for antibody levels to decline below the cut-off for testing seropositive.” The extent of underestimation of true epidemic size could therefore depend on the time at which the serologic study is taking place, or in other words, the time elapsed since the first peak of the epidemic.
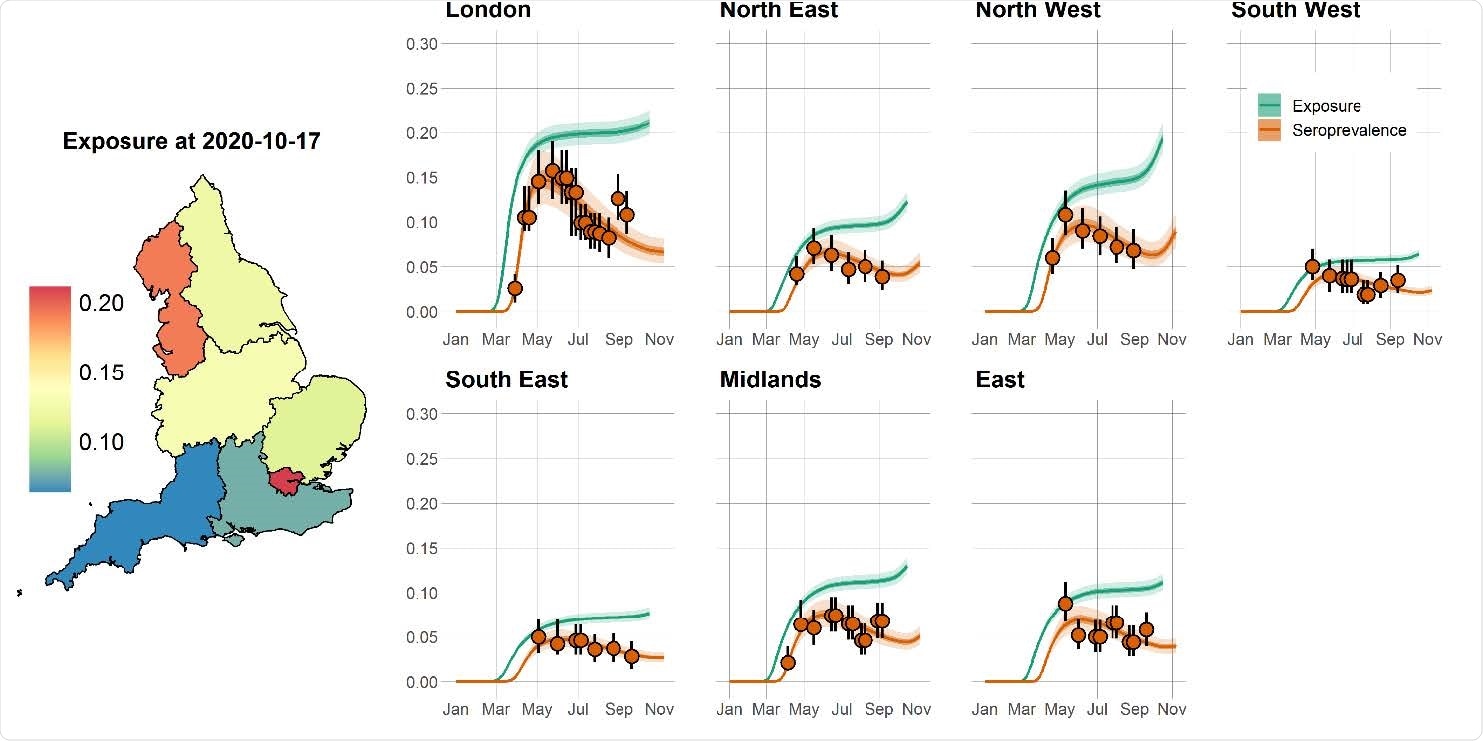
Time course of the SARS-CoV-2 pandemic up to 7 November 2020 for seven regions in England. The solid orange circles and black error bars in each regional panel represent the observed seroprevalence data and their confidence interval, respectively, after adjusting for the sensitivity and specificity of the antibody test. The green and orange lines show the model predictions of median exposure and seroprevalence, respectively, while the shaded areas correspond to 95% CrI. The regional predicted exposure levels (expressed as the proportion of the population that has been infected) as of 17 October 2020 are shown on the map of England.
Correcting serology-based estimates
The current study proposes a data triangulation method to find the number of people infected over time. The researchers used the mortality and seroconversion rate for this measure. The dates on which a given death count could be used in connection with seropositivity were calculated on the basis of the median period between infection and symptom onset, symptom onset and testing, and symptom onset to IgG production. This gave rise to a period of 21 days from exposure to seroconversion, assuming that all infections give rise to antibodies at about the same time.
The method thus focused on the IFR in various regions of England, as the anchoring variable, using this to find the number of people infected at three weeks before the date of the measured IFR. This has been used by other scientific groups, but the current study uses data at regional level within a country, on the basis of data that has been validated by another independent study carried out at the same time.
Infections could be twice the number of seropositives
The results show that seroconversion is followed by seropositivity for about 161 days, after which they serorevert. This means that as the epidemic progresses, the total population exposure will move progressively farther away from the seroprevalence measurements. “Ultimately, this may mean that more than twice as many people have been exposed to the virus relative to the number of people who are seropositive.” If so, seroprevalence data would be an unreliable parameter on which to base prospective containment policies.
The investigators also considered the fluctuating country-wide IFR over time. This could be due to different age structures in the population in different regions of the country. Alternatively, it could be due to a selection for the less frail sections of society as the epidemic progresses because in the initial phase of the outbreak, the frailest are most likely to die. Thus, over time, the IFR is likely to go down.
They found that the test positivity ratio out-performs both the case fatality ratio (CFR) or the hospitalization fatality ratio (HFR), in terms of predicting the mortality incidence graph. They thus used this to find the time-varying IFR.
Virus selects for less frail people over time
The study shows that only London may have shown a significant variation in IFR with epidemic progression, which has declined substantially. This could be due to the age structure of the city, which is pronounced, unlike other regions. Exposure levels do not appear to drive this variation since the Northwest of England also has similar levels of predicted exposure but no corresponding change in the IFR. Instead, younger populations with fewer very frail people show a clear selection pressure compared to other more mixed populations.
What are the implications?
The researchers emphasize that these findings do not mean that herd immunity cannot be achieved because of rapid seroreversion. The fact is that neutralizing antibodies are not synonymous with total antibodies, and an effective titer of the first may well coexist with a declining level of the latter. Also, T cell immunity has been found in seronegative individuals and is protective against disease. This method is useful only to reliably evaluate population prevalence and thus may help shape containment policies for the future.
They also point out that the study does not mean that herd immunity has almost been achieved in London because of the high exposure. Rather, this could be because London has a higher proportion of younger people compared to the rest of England, which resulted in a higher initial IFR as frailer people succumbed to COVID-19 at an increased rate, followed by a drop due to selection processes.
Using this method that is based on seroprevalence but accounts for seroreversion, using mortality data, the researchers estimated that the actual cumulative population exposure in the seven regions they studied is twofold or more greater than the estimate arrived at by seroprevalence studies, notably, the REACT study. Such estimates would make it easier to assess the potential role of vaccination and other interventions to break the chain of transmission in this pandemic.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources